
State Operations Manual
Chapter 5 - Complaint Procedures
Table of Contents
(Rev. 212, 02-10-23)
Transmittals for Chapter 5
Sections 5000 to 5080.1 relate to all Medicare/Medicaid-certified
provider/supplier types.
5000 - Management of Complaints and Incidents
5000.1 - Purpose of the Complaint/Incident Process
5000.2 - Overview
5010 - General Intake Process
5010.1 - Information to Collect From Complainant
5010.2 - Information to Provide to Complainant
5010.3 - Notification to the RO
5050 - CMS Regional Office Responsibility for Monitoring SA Management of
Complaints and Incidents
5060 - ASPEN Complaints/Incidents Tracking System (ACTS)
5060.1-Data Entry
5060.2-Reports
5070 - Priority Assignment for Nursing Homes, Deemed and Non-Deemed Non-Long
Term Care Providers/Suppliers, and EMTALA
5075 - Priority Definitions for Nursing Homes, Deemed and Non-Deemed Non-Long
Term Care Providers/Suppliers, and EMTALA
5075.1 - Immediate Jeopardy (for Nursing Homes, Deemed and Non-
Deemed Non-Long Term Care Providers/Suppliers, and EMTALA)
5075.2 - Non-Immediate Jeopardy - High (for Nursing Homes and
Deemed and Non-Deemed Non-Long Term Care Providers/Suppliers, and
EMTALA)
5075.3 - Non-Immediate Jeopardy - Medium (for Nursing Homes and
Deemed and Non-Deemed Non-Long Term Care Providers/Suppliers)
5075.4 - Non-Immediate Jeopardy – Low (for Nursing Homes and
Deemed and Non-Deemed Non-Long Term Care Providers/Suppliers)
5075.5 - Administrative Review/Offsite Investigation (for Nursing Homes
and Deemed and Non-Deemed Non-Long Term Care Providers/Suppliers)
5075.6 - Referral – Immediate (for Nursing Homes, Deemed and Non-
Deemed Non-Long Term Care Providers/Suppliers, and EMTALA)
5075.7 - Referral – Other (for Nursing Homes, Deemed and Non-Deemed
Non-Long Term Care Providers/Suppliers, and EMTALA)
5075.8 - No Action Necessary (for Nursing Homes, Deemed and Non-
Deemed Non-Long Term Care Providers/Suppliers, and EMTALA)
5075.9 - Maximum Timeframes Related to the Federal Onsite
Investigation of Complaints/Incidents
5077 - State Monitoring Visits
5078 - Pre-survey Activities
5079 - Entrance Conference - Non-Long Term Care
5080 - Investigation Findings and Reports
5080.1 - Report to the Complainant
5080.2 - Survey Exit Conference and Report to the Provider/Supplier
Sections 5100 to 5170 relate to deemed providers/suppliers.
5100 - Investigation of Complaints for Deemed Providers/Suppliers
5100.1 - Basis for Investigation
5100.2 - Initial Response to Complainant
5110 - Post-Survey Procedures
5110.1 - Substantial Compliance
5110.2 - Condition-Level, IJ
5110.3 - Condition-Level, Non-IJ
5110.4 - Full Survey after Complaint Survey with Condition-level Deficiencies,
When Authorized by the RO
5120 - Life Safety Code Guidance for Deemed Providers/Suppliers
5130 - Deemed Provider/ Supplier Hospital Refusal of Complaint Investigation Surveys
5140 - Complaints Involving HIV-Infected Individuals
5150 - Investigating Complaints Involving ESRD Services Provided by Deemed
Hospitals or CAHs
5160 - Investigating Complaints Against ESRD Suppliers
Section 5170 Relates to Hospital Restraint/Seclusion Death Reporting
and Investigation
5170 - Hospital Restraints/Seclusion Death Reporting and Investigation
5170.1 - Background
5170.2 - Responsibilities
5170.3 - Process
Sections 5200 to 5240 relate to all non-deemed provider/supplier types,
excluding nursing homes (SNFs/NFs).
5200 - Investigating Complaints for Non-Deemed Providers/Suppliers, Excluding
Nursing Homes (SNFs/NFs)
5200.1 - General Procedures
5200.2 - Special Procedures for Psychiatric Hospitals
5210- Processing of Complaints Originating with or Investigated by the RO
5220 - Investigation Conducted Directly by the RO
5230- Special RO Processing
5240 - Complaints - HHA Hotline
Sections 5300 to 5390 relate to nursing homes.
5300 - Investigation of Complaints for Nursing Homes
5300.1 - Task 1: Offsite Survey Preparation
5300.2 - Task 2: Entrance Conference/Onsite Preparatory Activities
5300.3 - Task 5: Information Gathering
5300.4 - Task 6: Information Analysis
5300.5 - Task 7: Exit Conference
5310 - Action on Allegations of Resident Neglect and Abuse, and Misappropriation of
Resident Property for Nursing Homes
5310.1 - Written Procedures
5310.2 - Review and Triage of Allegation
5310.2 A-Immediate Jeopardy Priority
5310.3 - Investigating Allegations
5310.4 - Factors Beyond the Control of the Individual
5320 - Reporting Findings of Abuse, Neglect, or Misappropriation of Property to the
Nurse Aide Registry
5320.1 - Notification Procedures- Preliminary Determinations
5320.2 - Conduct of Hearing for Nurse Aides
5320.3 - Reporting Findings
5330 - Reporting Abuse to Law Enforcement and the Medicaid Fraud Control Unit for
Nursing Homes
5340 - Post-Survey Certification Actions for Nursing Homes
5350 - Data Entry
5360 - Processing of Complaints Originating with or Investigated by the CMS RO
5370 - Pre-Investigation Actions on Allegations Originating Through the RO
5380 - RO Processing of RO Investigated Complaints
5390 - RO Oversight of Complaint-Related Processes
Sections 5400 to 5480.2 relate to alleged EMTALA violations
5400 - Investigations Involving Alleged EMTALA Violations
5410 - EMTALA and Born-Alive Infants Protection Act of 2002
5410.1 - Interaction of the Born-Alive Infant Protection Act and
EMTALA
5410.3 - Conduct of Investigations
5420 - Basis for Investigation
5430 - RO Direction of Investigation
5430.1 - Evaluation of Allegation
5430.2 - Request for Investigation of Allegations
5440 - Conducting an Investigation
5440.1 - Selecting the Team
5440.2 - Scheduling the Investigation
5440.3 - Guidelines for Surveyors Conducting Investigations
5440.4 - Conducting the Investigation
5440.5 - Exit Conference
5450 - Forwarding Report of Investigation to the RO
5460 - RO Review of Investigation
5460.1 - Hospital Is In Compliance - No Past Violation
5460.2 - Hospital Is In Compliance - Past Violation, No Termination
5460.3 - Hospital Is Not in Compliance - Immediate Jeopardy to Patient
Health and Safety
5460.4 - Hospital Is Not in Compliance - Situation Does Not Pose an
Immediate Jeopardy to Patient Health and Safety
5465 - Procedures for the 5-day QIO Review of Alleged Violations of 42 CFR 489.24
5470 - Termination Procedures for EMTALA Violations
5470.1 - Procedures for Termination when the EMTALA Violation is an
Immediate Jeopardy to Patient Health and Safety
5470.2 - Procedures for Termination When the EMTALA Violation is Not
Immediate Jeopardy to Patient Health and Safety
5480 - Procedures for QIO Review of Confirmed EMTALA Violation
5480.1 - Procedures for Coordinating 60 day QIO Review
5480.2 - EMTALA Case Referral to OIG
5480.3 - Releasing QIO Assessment
Sections 5500 to 5590 relate CLIA.
5500 - Complaints Involving Unaccredited Laboratories
5500.1 - Control
5500.2 - Acknowledgment
5500.3 - Evaluation
5500.4 - Scheduling Investigations
5500.5 - Conducting Investigations
5500.6 - Conducting Investigations in a Laboratory with a Certificate of
Waiver
5500.7 - Conducting Investigations in a Laboratory with a Certificate for
PPM Procedures
5500.8 - Post Investigation Actions
5500.9 - Resolution/Closeout
5510 - CLIA-Exempt Laboratory Complaint Investigations - General
5520 - Review of CLIA-Exempt Laboratory Complaints
5530 - Conducting Complaint Investigations and Surveys for CLIA-Exempt Laboratories
5540 - Complaint Investigations and Surveys of Accredited Laboratories Under CLIA
5550 - RO Direction of Complaint Investigation of an Accredited Laboratory
5560 - Conducting Complaint Survey of an Accredited Laboratory
5570 - Forwarding Investigation Report to RO
5580 - Accredited Laboratory Found in Compliance Following a Complaint Survey
5590 - Accredited Laboratory Found Not in Condition-level Compliance Following a
Complaint Survey
5000 - Management of Complaints and Incidents
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
NOTE: “CMS Regional Office (RO)” is used interchangeably with “CMS location”
throughout this Chapter of the SOM.
5000.1 – Purpose of the Complaint/Incident Process
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
Mission: To protect Medicare/Medicaid beneficiaries from abuse, neglect, exploitation,
inadequate care or supervision.
The goal of the Federal complaint/incident process is to establish a system that will assist
in promoting and protecting the health, safety, and welfare of residents, patients, and
clients receiving health care services. The complaint/incident management system has
three objectives.
1. The first objective and priority for the complaint/incident management system is
protective oversight. This is accomplished by analyzing the complaint allegations
and reported incidents received to identify and respond to those that appear to
pose the greatest potential for harming beneficiaries (has caused or is likely to
cause, serious injury, harm, impairment or death). Complaints/incidents of this
type that allege an immediate threat to the health, safety or welfare of individuals
are investigated immediately.
2. The second objective is prevention. Complaints/incidents that do not allege a
threat of serious harm are investigated to determine if a problem exists that could
have a negative impact on the healthcare services provided. The investigation of
these complaints/incidents is designed to identify and correct less serious
complaints/incident to prevent the escalation of these problems into more serious
situations that would threaten the health, safety and welfare of the individuals
receiving the service. These complaints/incidents are also prioritized and
investigated based on the seriousness of the allegations.
Numerous or more frequent complaints/incidents may indicate systemic problems
and therefore may be assigned a higher priority for investigation.
3. The third objective is to promote efficiency and quality within the health care
delivery system. Complaints/incidents that are not directly related to Federal
requirements are forwarded to the appropriate agency(ies) for follow-up and
investigation. Complaints/incidents in this category may include but are not
limited to Medicare/Medicaid fraud, complaints against individual licensed
practitioners, and billing issues.
5000.2 – Overview
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
All the procedures in this chapter are followed when complaints and reported incidents,
including referrals from public entities, involve Medicare-certified providers/suppliers,
Medicaid-certified providers/suppliers, or CLIA-certified laboratories. The investigation
and resolution of complaints are critical certification activities. The CMS, the State
Medicaid Agency (SMA), and the State survey agency (SA) are responsible for ensuring
that participating providers/suppliers of health care services continually meet Federal
requirements. This requires that the SA promptly reviews complaints/incidents, conducts
unannounced onsite investigations of reports alleging noncompliance, and informs the
CMS Regional Office (RO) and/or the SMA any time certification requirements are
found to be out of compliance.
Since there are multiple activities associated with the management of complaints and
incidents, responsibilities often cut across organizational lines. Thus, the SA must
demonstrate clear-cut accountability for each step of the process and a focal
coordinating/controlling responsibility to assure timely and appropriate action. The SA’s
responsibilities cannot be delegated.
5010 - General Intake Process
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
A complaint is an allegation of noncompliance with Federal and/or State requirements. If
the SA determines that the allegation(s) falls within the authority of the SA, the SA
determines the severity and urgency of the allegations, so that appropriate and timely
action can be pursued. Each SA is expected to have written policies and procedures to
ensure that the appropriate response is taken for all allegations and is consistent with
Federal requirements as well as with procedures in the State Operations Manual. This
structure needs to include response timelines and a process to document actions taken by
the SA in response to allegations. If a State’s time frames for the investigation of a
complaint/incident are more stringent than the Federal time frames, the intake is
prioritized using the State’s timeframes. The SA is expected to be able to share the logic
and rationale that was utilized in prioritizing the complaint/incident for investigation.
The SA response must be designed to protect the health and safety of all residents,
patients, and clients.
Besides the SA, other public entities receive information and/or perform investigations.
These entities include the office of the coroner or medical examiner, end-stage renal
disease (ESRD) networks, quality improvement networks (QINs), law enforcement, the
ombudsman’s office, and protection and advocacy systems. At times, these public
entities will forward information to the SA if there are concerns about the health and
safety of residents, patients, and clients. The SAs are required to manage and investigate
these referrals as complaints.

An allegation is an assertion of noncompliance with Federal health and safety
regulations. The point of receipt of the allegation is a critical fact-finding and decision-
making point. The SA ensures that its complaint telephone number is listed in local
directories. Information regarding the care, treatment and services provided to
beneficiaries can come from a variety of sources, including beneficiaries themselves,
beneficiaries’ family members, health care providers, concerned citizens, public agencies,
or media reports. Report sources may be verbal or written. In some instances, the
complainant may request anonymity.
The SA and RO ensure the privacy and anonymity of every complainant. Generally, the
SA follows the disclosure procedures under chapter 3, §3308. The SA discloses the
complainant’s identity only to those individuals with a need to know who are acting in an
official capacity to investigate the complaint.
In addition to these Federal requirements, the SA abides by any State procedures not in
direct conflict with CMS instructions. The SA notifies the RO if State regulations
conflict directly with any part of these complaint procedures.
See also Section 5310.1 for information related to facility-reported incidents.
5010.1 - Information to Collect From Complainant
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The SA collects information necessary to make important decisions about the allegations.
In instances where written or verbal allegations are received, subsequent communication
may be necessary to obtain additional information.
Comprehensive information should be collected during the intake process to allow for
proper prioritization, including the following:
• Information about the complainant (e.g., name, address, telephone, etc.);
• Individuals involved and affected;
• Narrative/specifics of the complainant’s concerns including the date, and time of
the allegation;
• The complainant’s views about the frequency and pervasiveness of the allegation;
• Name of the provider/supplier including location (e.g., unit, room, floor) of the
allegation, if applicable;
• How/why the complainant believes the alleged event occurred;
• Whether the complainant initiated other courses of action, such as reporting to
other agencies, discussing issues with the provider, and obtaining a
response/resolution; and
• The complainant’s expectation/desire for resolution/remedy, if appropriate.
5010.2 - Information to Provide to Complainant
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The complaint intake process assists the complainant in resolving his/her conflicts. As
part of the intake process the SA provides the following:
• Policies and procedures for handling intakes including the scope of the SA’s
regulatory authority and any considerations pertaining to confidentiality;
• The course of action that the SA or RO will take and the anticipated time frames;
• Information about other appropriate agencies that could provide assistance
including the name and telephone number of a contact person, if available; and
• A SA contact name and number for follow-up by the complainant.
NOTE FOR DEEMED PROVIDERS/SUPPLIERS: If a complaint does not allege
condition-level noncompliance, the SA may: 1) advise the complainant to file the
complaint to the accrediting organization (AO), or 2) ask for the complainant's
permission to release the information to the AO.
5010.3 – Notification to the RO
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
1 – Notification to the RO
The SA immediately forwards allegations involving the following to the RO:
• Deemed providers/suppliers;
• Hospital and psychiatric residential treatment facility (PRTF) restraint/seclusion-
related deaths;
• EMTALA complaints;
• Fires resulting in serious injury or death in a Medicare/Medicaid-certified facility;
• Federal facilities;

• Religious Non-medical Health Care Institutions (RNHCIs)(evaluation performed
by Region I, Boston, only);
• CLIA-certified laboratories holding a certificate of accreditation. (See Chapter 6).
• CLIA-exempt laboratory. (See Chapter 6);
• Blood transfusion-related fatalities (See Chapter 6 and Appendix C);
• Over-utilization or inappropriate utilization of services within the QIO’s
jurisdiction;
• Civil rights violations; or
• Medicare or Medicaid fraud
2 – Special Cases
The SA considers whether notification to the RO is appropriate. If a complaint is
especially significant, sensitive, or attracting broad public or media attention, the SA
informs the RO immediately. Additionally, the SA needs to consider any other early
notice requirements prescribed by other State or Federal policies or interagency
agreements.
5050 - CMS Regional Office Responsibility for Monitoring SA
Management of Complaints and Incidents
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
CMS ROs are responsible for monitoring the SAs’ management of complaints and
incidents to assure that the SAs are complying with the provisions set forth in Federal
regulations, the SOM, and CMS policy memoranda. As part of the monitoring process,
the SAs will be evaluated in accordance with the criteria set forth by the State
Performance Standard Review. Many States have State laws and regulations that specify
how to manage complaints and incidents. Whenever possible, State and Federal
requirements should be integrated to avoid unnecessary duplication. CMS ROs should
accept State requirements that meet or exceed the intent of the Federal requirements. At
a minimum, it is expected that noncompliance with Federal requirements resulting from a
complaint or reported incident will receive follow-up and be documented in the Aspen
Complaints Tracking System (ACTS).
5060 – ASPEN Complaints/Incidents Tracking System (ACTS)
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
The SA collects information related to complaints and facility-reported incidents and
uses a system to track and monitor the receipt and disposition of complaint and incident
intakes.
The ASPEN Complaints/Incidents Tracking System (ACTS) is designed to track,
process, and report on complaints and incidents reported against health care providers
and suppliers regulated by CMS. It is designed to manage all operations associated with
complaint/incident processing, from initial intake and investigation through the final
disposition.
The ACTS must be used for the intake of all allegations against Medicare/Medicaid-
certified providers/suppliers and CLIA. The ACTS is a Federal system and data entered
into ACTS is subject to Federal laws governing disclosure and the protection of an
individual’s right to privacy.
A complaint/incident record is created in ACTS based on how the allegation is received
by the SA or RO. For example, if one person calls with ten allegations about one
provider/supplier, this is counted as one complaint record. If six people call with the
same allegation, this is counted as six telephone calls and is counted as six complaint
records. If one letter is received with one or many allegations and is signed by 20 people,
this is counted as one complaint record.
5060.1 - Data Entry
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
The SAs and ROs are required to enter into ACTS:
• All complaints gathered as part of Federal survey and certification
responsibilities, regardless if an onsite survey is conducted [i.e., complaints
related to noncompliance with the Federal condition(s) of participation (COPs),
condition(s) for coverage (CFCs), condition(s) for certification, requirement(s)
for participation (RFPs), or EMTALA requirement(s)]; and
• For nursing homes, all self-reported incidents that are reported under Federal
law and the requirements for participation [i.e., reporting to law enforcement of
crimes occurring in LTC facilities – §1150B of the Social Security Act and
§483.12(b)(5); alleged violations of abuse, neglect, exploitation or mistreatment,
including injuries of unknown source and misappropriation of resident property –
§483.12(c)(1) and (4)]. For non-long term care providers/suppliers, all self-
reported incidents that require a Federal onsite survey.
Revisions related to the entry of self-reported incidents into ACTS will be implemented no
later than October 1, 2024, in order to provide time for the SAs to prepare given the
resource constraints in the SAs.
The information recorded in ACTS reflects the allegation furnished by the
complainant/provider/supplier at the time of the intake. At a minimum, if the intake
information requires an onsite survey and the allegation may involve both Federal and

State licensure requirements, a Federal onsite survey is completed and entered into
ACTS.
If an investigation finds one or more violations of Federal requirements, the findings
must be cited under the appropriate tags and entered into the Federal system even if the
information is entered into a State licensure data system. Since this information is
essential to the effective management of the survey and certification program, it is
important that SAs complete the required fields in ACTS in a timely manner.
Exhibit 23 defines the required fields in ACTS.
Tracking of Referrals in ACTS
The SAs are required to enter into ACTS all referrals from public entities that allege
noncompliance with the Federal requirements. For reporting purposes, the SAs should
enter these cases as complaints (i.e., Intake Type=Complaint, Intake Subtype=Federal
COPs, CFCs, RFPs, EMTALA). In order to more quickly identify which of these cases
stem from a referral, the SAs are expected to check the appropriate category under the
“Source” field. For example, for referrals from the coroner’s office, states would check
“Coroner” under the “Source” field for the intake.
Tracking of State Monitoring Visits (See Section 5077) in ACTS
When a State Monitoring Visit results in a Federal deficiency, the SA will identify the
survey in ASPEN as “complaint” and create an intake and survey record in ACTS. The
data should be entered into ACTS as follows:
• Intake Type = Complaint;
• Intake Subtype = Federal COPs, CFCs, RFPs, EMTALA;
• Source = State SA;
• Priority = can vary; and
• Allegation Type = State Monitoring.
5060.2 - Reports
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
The ACTS produces a variety of reports that may be used for analysis and evaluation of
provider/supplier performance. Complaint/incident reports are generated and displayed
through menus that can be accessed in ACTS. Reports may be produced for one
provider/supplier, or reports may be combined and present information for multiple
providers/suppliers. Report filtering criteria is available through the Report
Customization window, which allows the user to select criteria for the report to meet the
user’s specifications. Refer to the ACTS Procedures Guide for a list and description of
the reports available in ACTS.

NOTE:
FOR ADDITIONAL INFORMATION ON SPECIFIC POLICIES RELATED TO:
• DEEMED PROVIDERS AND SUPPLIERS, EXCLUDING CLIA, SEE
SECTION 5100
• NON-DEEMED PROVIDERS AND SUPPLIERS, SEE SECTION 5200
• NURSING HOMES, SEE SECTION 5300
• EMTALA, SEE SECTION 5400
• CLIA LABORATORIES, SEE SECTION 5500
• ESRD, SEE SECTION 5160 AND SECTION 5170
5070 - Priority Assignment for Nursing Homes, Deemed and Non-
Deemed Non-Long Term Care Providers/Suppliers, and EMTALA
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
This section does not apply to clinical laboratories subject to the Clinical
Laboratory Improvement Amendments of 1988 (CLIA). See Sections 5500 – 5590
for CLIA information.
An assessment of each complaint or incident intake must be made by an individual who
is professionally qualified to evaluate the nature of the problem based upon his/her
knowledge of Federal requirements and his/her knowledge of current clinical standards of
practice.
From a complainant’s allegation(s) or an allegation from a facility-reported incident, the
SA/CMS Location identifies potential concerns where the provider/supplier may not be in
compliance with Federal requirements. The SA/CMS Location must review the
allegation(s) for all requirements that apply and should be investigated. These
requirements will be specific to each health care entity. The surveyor then investigates
each of those areas of concern and health care entity type.
The role of the surveyor is not to validate whether the events contained in the allegation
had occurred, but it is to determine whether the facility is in compliance with the Federal
requirements for Medicare/Medicaid-certified providers/suppliers. If CMS or the SA
believes that the complaint or facility-reported incident should also be investigated under
the jurisdiction of another entity, referrals should be made as appropriate (e.g., law
enforcement for criminal activity, State licensing boards for health care practitioners, the
Medicare Administrative Contractor (MAC) for billing issues).
In the case of nursing homes, in situations where a determination is made that immediate
jeopardy may be present and ongoing, the SA must start the on-site investigation within
three business days of receipt of the initial complaint or incident report. Receipt of the
initial complaint or incident report means when the report is received by the SA, whether
it is received by the SA directly, or another State agency under arrangement or
contractor that is receiving the report on behalf of the SA from the complainant or
facility. Also, if a complaint or facility-reported incident is received after business hours,
then it is considered to be received on the next business day, for purposes of calculating
the investigation timeframe. For example, if a complaint is received on Saturday and the
SA office is closed during the weekend, then the following Monday will be used to
calculate the investigation timeframe. Revisions related to the timeframes for
investigating nursing home complaints and facility-reported incidents will be
implemented no later than October 1, 2023, to provide time for the SAs to prepare given
the resource constraints in the SAs.
For non-long term care providers/suppliers, in situations where a determination is made
that immediate jeopardy may be present and ongoing, the SA is required to start the on-
site investigation within two business days of receipt of the complaint or incident report,
or, in the case of a deemed provider or supplier, within two business days of RO
authorization for investigation. The same process applies to EMTALA complaints or a
survey related to a report of a hospital or CAH Distinct Part Unit patient death associated
with the use of restraint or seclusion. The SA’s investigation must be initiated within two
business days of RO authorization for investigation.
Generally, an alleged event occurring more than 12 months prior to the intake date would
not require a complaint investigation. However, the SA is not precluded from conducting
a Federal investigation (with appropriate RO authorization, where required) to determine
current compliance status based on the concerns identified in the complaint.
For nursing homes, an onsite survey may not be required if there is sufficient evidence
that the facility does not have continuing noncompliance and the alleged event occurred
before the last standard survey.
For all intakes concerning deemed status providers or suppliers where the intake involves
allegations of substantial noncompliance (in other words, the allegation would result in a
condition-level deficiency citation if found to be true and uncorrected), the SA must
submit a request for RO approval of a complaint validation survey (i.e., substantial
allegation validation survey). The SA must obtain RO approval before conducting a
substantial allegation validation survey. The RO will authorize the SA to conduct the
survey by issuing electronically via ACTS a Form CMS-2802, which will indicate the
specific conditions for which the SA must assess compliance. The RO must authorize
assessment of compliance for a whole condition and not just for particular standards
within a condition, unless the Form CMS-2802 for the applicable provider/supplier type
permits selection of a specific standard, e.g., Life Safety Code.

All allegations of EMTALA violations related to a hospital (which also includes cancer,
children’s, long term care, psychiatric and rehabilitation hospitals) or CAH, regardless of
whether the hospital or CAH is deemed, must be referred to the RO. The RO will
determine whether the SA will conduct an EMTALA investigation.
In cases where the SA or RO has noted a pattern of similar complaints about a specific
provider or supplier, each of which on its own merits would be triaged at a medium or
low level, the SA or RO has the discretion to assign a higher triage level to a current
intake based on the noted pattern, in order to ensure timely investigation of the
provider’s/supplier’s compliance with the applicable requirements or Conditions.
CMS expects SAs to prioritize complaints at the appropriate level that is warranted. The
timeframes in Section 5075 below represent maximum timeframes for investigation
(Note: Revisions related to the timeframes for investigating nursing home complaints and
facility-reported incidents will be implemented no later than October 1, 2023, to provide
time for the SAs to prepare given the resource constraints in the SAs); the SA is not
precluded from investigating complaints and facility-reported incidents within a shorter
timeframe. In addition, the SA is not precluded from taking other factors into
consideration in its triage decision. For example, the SA may identify a trend in
allegations that indicates an increased risk of harm to residents or the SA may receive
corroborating information from other complainants regarding the allegation.
See also Section 5310.2 for requirements for nursing home facility-reported incidents.
5075 - Priority Definitions for Nursing Homes, Deemed and Non-
Deemed Non-Long Term Care Providers/Suppliers, and EMTALA
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
5075.1 - Immediate Jeopardy (for Nursing Homes, Deemed and Non-
Deemed Non-Long Term Care Providers/Suppliers, and EMTALA)
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
General Provisions
The regulations at 42 CFR 489.3 define immediate jeopardy as, “A situation in which the
provider’s noncompliance with one or more requirements of participation has caused, or
is likely to cause, serious injury, harm, impairment, or death to a resident.” Appendix Q
contains the Guidelines for Determining Immediate Jeopardy. Intakes are assigned this
priority if the alleged noncompliance indicates there was serious injury, harm,
impairment or death of a patient or resident, or the likelihood for such, and there
continues to be an immediate risk of serious injury, harm, impairment or death of a
patient or resident unless immediate corrective action is taken. In addition, for nursing
homes, facility-reported incidents are assigned this priority if immediate jeopardy may
have occurred, regardless of whether an immediate risk may continue to exist. Examples
of intakes that are assigned this priority include, but are not limited to, the following:

• All intakes alleging abuse of a resident/patient/client and it is uncertain that they
are adequately protected.
• For nursing homes, all intakes alleging eviction of a resident to an unsafe
location.
• Intakes alleging EMTALA noncompliance may also be assigned this priority.
• Any hospital self-reported incident of patient death associated with use of
restraint or seclusion which the RO determines requires an on-site investigation is
also assigned this priority.
When the SA or RO makes the determination that a complaint or incident report suggests
an immediate jeopardy may be present, the investigation is to be initiated in accordance
with Section 5075.9.
See also Section 5310.2A for additional guidance related to nursing home facility-
reported incidents. (Note: Revisions related to the timeframes for investigating nursing
home complaints and facility-reported incidents will be implemented no later than
October 1, 2023, to provide time for the SAs to prepare given the resource constraints in
the SAs.)
Fires Resulting in Serious Injury or Death
Fires resulting in serious injury or death are prioritized as “immediate jeopardy”. The
following actions are taken when a report of a fire resulting in serious injury or death in a
Medicare/Medicaid certified facility is received from any source:
The SA
• Enters the complaint or self-reported incident into ACTS (Priority = IJ,
Allegation Category = Life Safety Code);
• Informs the appropriate RO of fire resulting in serious injury or death no
later than one working day after receipt of the intake;
• Compiles information as needed to present a comprehensive picture of the
situation surrounding the fire;
• Takes appropriate action necessary to assist the Medicare/Medicaid-
certified provider/supplier to protect and/or relocate residents or patients
from further harm; and
• Performs the Life Safety Code investigation.
The RO
• Informs CMS Central Office (CO) of the fire and planned actions, sending
a copy of the alert to the Life Safety Code specialist;

• Consults with the CO to determine whether there is an indication for CO
participation in the survey for program evaluation purposes;
• Reports any findings and actions taken by the SA to the CO at the end of
the on-site survey; and
• At its discretion, may accompany the SA during the on-site survey.
The CO
• Consults with the RO to determine whether or not issues are present that
indicate further investigation to determine the adequacy of current
standards and their application; and
• In certain cases, CO staff may accompany regional and/or state personnel
on the on-site survey.
5075.2 - Non-Immediate Jeopardy - High Priority (for Nursing Homes
and Deemed and Non-Deemed Non-Long Term Care
Providers/Suppliers, and EMTALA)
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
Nursing Homes:
Intakes are assigned a “high” priority if the alleged noncompliance with one or more
requirements may have caused harm that negatively impacts the individual’s mental,
physical and/or psychosocial status and are of such consequence to the person’s well-
being that a rapid response by the SA is indicated. Usually, specific rather than general
information (such as: descriptive identifiers, individual names, date/time/location of
occurrence, description of harm, etc.) factors into the assignment of this level of priority.
When the SA makes the determination that the alleged noncompliance may have caused
actual physical and/or psychosocial harm to the resident(s), the SA must initiate an onsite
survey within an annual average of 15 business days of receipt of the initial report, not to
exceed 18 business days. Investigating nursing home complaints and facility-reported
incidents according to these timeframes will be implemented no later than October 1,
2023, to provide time for the SAs to prepare given the resource constraints in the SAs.
NOTE: Exhibit 22 provides additional guidance to distinguish between the priorities of
“immediate jeopardy” and “non-immediate jeopardy - high” for nursing home
complaints/incidents.
NOTE: Please refer to Tag F610 in Appendix PP of the State Operations Manual for
information related to facility responses to alleged violations, including facility
investigation, resident protection, and corrective actions.

Non-Long Term Care Providers/Suppliers
Intakes are assigned this priority if the alleged noncompliance with the applicable
Conditions of Participation, Coverage or Certification, or EMTALA requirements, if
found to be true and uncorrected, would not represent an IJ, but would result in a
determination of substantial noncompliance, i.e., at least one condition-level deficiency.
Intakes assigned this priority require an onsite survey to be initiated within 45 calendar
days after intake prioritization for non-deemed providers/suppliers, and within 45
calendar days after authorization of the investigation by the RO for deemed status
providers/suppliers. The RO has the discretion to request the onsite survey be initiated in
less than 45 calendar days.
5075.3 - Non-Immediate Jeopardy - Medium Priority (for Nursing
Homes and Deemed and Non-Deemed Non-Long Term Care
Providers/Suppliers)
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
Nursing Homes:
Complaints are assigned a “medium” priority if the alleged noncompliance with one or
more requirements caused no actual physical and/or psychosocial harm but there is the
potential for more than minimal harm to the resident(s) (Severity Level 2). Facility-
reported incidents are assigned a “medium” priority if the alleged noncompliance with
one or more requirements caused no actual physical and/or psychosocial harm but there
is the potential for more than minimal harm to the resident(s) (Severity Level 2) and the
facility has not provided an adequate response to the allegation or it is not known
whether the facility provided an adequate response. For complaints and facility-reported
incidents that are assigned a “medium” priority, the SA must initiate an onsite survey
within 45 calendar days of receipt of the initial report. Investigating nursing home
complaints and facility-reported incidents according to this timeframe will be
implemented no later than October 1, 2023, to provide time for the SAs to prepare given
the resource constraints in the SAs.
NOTE: Please refer to Tag F610 in Appendix PP of the State Operations Manual for
surveyor guidance related to facility responses to alleged violations, including facility
investigation, resident protection, and corrective actions.
Non-Long Term Care Providers/Suppliers
Intakes are assigned this priority if the alleged noncompliance with one or more standards
within a Condition of Participation, Condition for Coverage or Condition for Certification
is limited in manner and degree and/or caused, or may cause, harm that is of limited
consequence and does not impair the individual’s mental, physical and/or psychosocial
status or function. In other words, the incident or complaint, if found to be true and

uncorrected, would not result in a determination of substantial non-compliance, i.e., there
would not be any condition-level deficiency.
For non-deemed providers/suppliers, intakes assigned this priority are scheduled in
accordance with section 5075.9 for investigation no later than when the next on-site
survey occurs.
For deemed providers/suppliers, the SA (or RO, if the RO handled the intake)
advises the complainant that the allegation does not meet the criteria for a Federal
investigation and refers the complainant to the applicable accrediting
organization(s)(AOs) in accordance with the provisions of section 5100.2.
5075.4 - Non-Immediate Jeopardy – Low Priority (for Nursing Homes
and Deemed and Non-Deemed Non-Long Term Care
Providers/Suppliers)
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
Nursing Homes
Intakes are assigned a “low” priority if the alleged noncompliance with one or more
requirements may have caused no actual harm with a potential for minimal harm
(Severity Level 1). The investigation is to be initiated in accordance with section 5075.9.
Investigating nursing home complaints and facility-reported incidents according to
section 5075.9 will be implemented no later than October 1, 2023, to provide time for the
SAs to prepare given the resource constraints in the SAs.
In addition, facility-reported incidents are assigned a “low” priority if the alleged
noncompliance with one or more requirements may have caused no actual physical
and/or psychosocial harm but there is the potential for more than minimal harm to the
resident(s)(Severity Level 2) and the facility has provided a potentially adequate
response to the allegation.
NOTE: Please refer to Tag F610 in Appendix PP of the State Operations Manual for
information related to facility responses to alleged violations, including facility
investigation, resident protection, and corrective actions.
The SA reviews these intakes for tracking of possible trends in order to determine if there
are common themes that suggest areas for focused attention when the next on-site survey
occurs. If the SA identifies a trend that suggests similar concerns, the SA either
investigates the concerns during the next standard or complaint survey or initiates a
complaint survey.
Non-Long Term Care Providers/Suppliers
Intakes are assigned this priority if the alleged noncompliance with one or more standards
within a Condition of Participation, Coverage or Certification may have caused physical,
mental and/or psychosocial discomfort that does not constitute injury or damage.
For non-deemed providers/suppliers, the SA reviews these intakes for tracking of
possible trends in the nature of complaints in order to determine if there are common
themes that suggest areas for focused attention when the next on-site survey occurs.
Individual investigations of each intake are not required, although the SA has the
discretion to conduct a complaint survey if trending suggests a number of similar
problems that might warrant an on-site investigation.
For deemed providers/suppliers, the SA (or RO, if the RO handled the intake) advises
the complainant that the allegation does not meet the criteria for a Federal investigation
and refers the complainant to the applicable accreditation organization(s)(AOs) in
accordance with the provisions of section 5100.2.
5075.5 - Administrative Review/Offsite Investigation (for Nursing
Homes and Deemed and Non-Deemed Non-Long Term Care
Providers/Suppliers)
(Rev. 120, Issued: 09-19-14, Effective: 09-19-14, Implementation: 09-19-14)
Nursing Homes
Intakes are assigned an “administrative review/offsite investigation” priority if an onsite
investigation is not necessary. However, the SA or RO conducts and documents in the
provider file an offsite administrative review (e.g., written/verbal communication or
documentation) to determine if further action is necessary. Where an administrative
review/offsite investigation is conducted by the SA, the SA may confirm the findings at
the next onsite survey.
Non-long Term Care Providers/Suppliers
For non-long term care providers/suppliers, both deemed and non-deemed, administrative
review or offsite investigation is generally not permitted. Exceptions are usually limited
to the following types of cases:
• RO review of alleged noncompliance with provider agreement requirements found in
42 CFR Part 489, such as:
• Alleged discrimination against Medicare beneficiaries, or
• Failure of a hospital to accept Medicare-like payment rates for treatment
provided to a patient referred by an Indian Health Service or tribal facility.
• RO review in the case of a CAH:
• Of a notice by the MAC of failure of a CAH to maintain an average annual
per patient length of stay not exceeding 96 hours, or
• Whether a relocating CAH or an existing hospital seeking to convert to CAH
status satisfies the CAH location requirements.
The RO documents in the provider/supplier file the results of such administrative review
or offsite investigation. Note: depending on RO practice, such administrative review
cases may or may not be entered into ACTS.
5075.6 - Referral – Immediate (for Nursing Homes, Deemed and Non-
Deemed Non-Long Term Care Providers/Suppliers, and EMTALA)
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
Intakes are assigned a “Referral – Immediate” priority if the nature and seriousness of a
complaint/incident or State procedures requires the referral or reporting of this
information for investigation to another agency, board, or ESRD network without delay.
For example, if a complaint has criminal implications and the complainant has not
reported the incident to law enforcement, the SA must report the suspected crime to law
enforcement immediately (NOTE: In such cases, the referral is recorded in the
Contact/Refer tab under the ACTS intake). This priority may be assigned in addition to
one of the priorities in sections 5075.1 through 5075.5.
When the SA refers the complaint/incident to another agency or entity (e.g., law
enforcement, Ombudsman, licensure agency, etc.) for action, the SA must request a
written report on the results of the investigation by the outside entity. Referral to an
outside entity does not relieve the SA of the responsibility to assess compliance with
Federal conditions or requirements, when applicable. The timeframes for investigation
are not altered by the referral. (Expressed requests by law enforcement that the SA defer
an onsite investigation should be discussed with the CMS RO, as appropriate.)
5075.7 - Referral – Other (for Nursing Homes, Deemed and Non-
Deemed Non-Long Term Care Providers/Suppliers, and EMTALA)
(Rev. 120, Issued: 09-19-14, Effective: 09-19-14, Implementation: 09-19-14)
Intakes are assigned a “Referral – Other” priority when they are referred to another
agency, board, or ESRD network for investigation or for informational purposes. This
priority may be assigned in addition to one of the priorities in sections 5075.1 through
5075.5.
When the SA refers the complaint/incident to another agency or entity (e.g., law
enforcement, Ombudsman, licensure agency, etc.) for action, the SA must request a
written report on the results of the investigation by the outside entity. Referral to an
outside entity does not relieve the SA of the responsibility to assess compliance with
Federal conditions or requirements, when applicable. The time frames for investigation
are not altered by the referral. (Expressed requests by law enforcement that the SA defer
an onsite investigation should be discussed with the CMS RO, as appropriate.)
5075.8 - No Action Necessary (for Nursing Homes, Deemed and Non-
Deemed Non-Long Term Care Providers/Suppliers, and EMTALA)
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
Intakes are assigned a “No Action Necessary” priority if the SA or RO determines with
certainty that no further investigation, analysis, or action is necessary.
For example, no action is necessary if the allegation is not related to any Federal COPs,
CFCs, conditions for certification, RFPs, or EMTALA requirement(s); or situations in which
a previous survey investigated the exact same event(s) and either did not find noncompliance,
or noncompliance was previously identified and subsequently corrected by the
provider/supplier.
This category would also be used for intakes concerning an event that occurred more than 12
months in the past, unless the SA (or the RO, in the case of a deemed status
provider/supplier) determines that a complaint investigation is nevertheless warranted.
Nursing Homes
The following are examples of reports that require no further action or investigation by
the SA/RO:
1) Facility-reported incidents that are not reportable events under Federal law or
regulations;
2) Facility-reported incidents involving injuries where the resident was able to
explain or describe how he/she received the injury as long as there is no other
indication of abuse or neglect;
3) Facility-reported incidents involving lost items, which are found and no theft is
suspected; and
4) The alleged event occurred before the last standard survey and there is sufficient
evidence that the facility does not have continuing noncompliance since the last
standard survey.
NOTE: Sufficient evidence that the facility does not have continuing noncompliance may
be indicated by a recent survey that reviewed the concern, no additional complaints or
facility reported incidents have been received regarding the same issue, and interview
with the Long-term Care Ombudsman which reveal no concerns.

5075.9 - Maximum Time Frames Related to the Federal Onsite Investigation of Complaints/Incidents
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
Intake Prioritization
Provider Type
Immediate Jeopardy (IJ)
Non-IJ High
Non-IJ Medium
Non-IJ Low
Nursing home
complaints
SA must initiate an onsite survey
within 3 business days of receipt of
the initial report.
SA must initiate an onsite
survey within an annual
average of 15 business days
of receipt of the initial report,
not to exceed 18 business
days.
SA must initiate an onsite
survey within 45 calendar
days of receipt of the initial
report.
SA must track/trend for
potential focus areas during
the next onsite survey, or
initiate a new complaint
survey.
Nursing home
incidents
With inadequate resident protection,
SA must initiate an onsite survey
within 3 business days of receipt of
the initial report.
With potentially adequate resident
protection, SA must initiate an onsite
survey within 7 business days of
receipt of the initial report.
See Section 5310.2A.
SA must initiate an onsite
survey within an annual
average of 15 business days
of receipt of the initial report,
not to exceed 18 business
days.
With an inadequate facility
response, SA must initiate an
onsite survey within 45
calendar days of receipt of
the initial report.
With a potentially adequate
facility response, SA must
track/trend for potential focus
areas during the next onsite
survey, or initiate a new
complaint survey.
Non-deemed non-long
term care
providers/suppliers
SA must initiate an onsite survey
within 2 business days of receipt.
SA must initiate an onsite
survey within 45 calendar
days of prioritization
SA must investigate no later
than when the next onsite
survey occurs
SA must track/trend for
potential focus areas during
the next onsite survey.
Deemed
providers/suppliers
SA must initiate an onsite survey
within 2 business days of receipt of
RO authorization
SA must initiate an onsite
survey within 45 calendar
days of receipt of RO
authorization.
Complainant is referred to the
applicable accrediting
organization(s)
Complainant is referred to the
applicable accrediting
organization(s)
EMTALA
SA must initiate an onsite survey
within 2 business days of receipt of
RO authorization.
SA must initiate an onsite
survey within 45 calendar
days of receipt of RO
authorization
N/A
N/A
Death associated with
restraint/seclusion-
Hospitals
SA must initiate an onsite survey
within 2 business days of receipt of
RO authorization.
N/A
N/A
N/A
Fires resulting in
serious injury or death
SA must initiate an onsite survey
within 2 business days of receipt.
N/A
N/A
N/A
Investigating nursing home complaints and facility-reported incidents according to section 5075.9 will be implemented no later than
October 1, 2023, to provide time for the SAs to prepare given the resource constraints in the SAs.
5077 - State Monitoring Visits
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
“State monitoring visits” refers to visits by the SA to oversee a provider’s/supplier’s
compliance status:
• During bankruptcy, in those cases in which CMS has authorized such visits.
• After a change of ownership, as authorized by the CMS RO.
• During or shortly after removal of immediate jeopardy when the purpose of the
visit is to ensure the welfare of the residents/clients/patients by providing an
oversight presence, rather than to perform a structured follow-up visit.
• In other circumstances, as authorized by the CMS RO.
See Section 5060 for data entry requirements for this type of visit.
5078 – Pre-Survey Activities
(Rev. 120, Issued: 09-19-14, Effective: 09-19-14, Implementation: 09-19-14)
To assist in planning the complaint investigation, prior to going on-site the SA should
review the provider’s or supplier’s prior compliance record and, as applicable, quality
indicators, ESRD Outcome List and Data or supporting information received from other
programs, such as the Ombudsman program or Protection and Advocacy program. This
process may require additional contact with the complainant. More information on pre-
survey activities may be found in Section 5170 for ESRD facilities, Section 5300.1 for
long term care facilities, in the provider/supplier-specific SOM appendices and in
Appendix V concerning EMTALA of the SOM.)
5079 – Entrance Conference - Non-Long Term Care
Providers/Suppliers
(Rev. 120, Issued: 09-19-14, Effective: 09-19-14, Implementation: 09-19-14)
Onsite complaint investigations must always be unannounced. Upon entrance, advise the
provider/supplier CEO or other senior official on duty of the general purpose of the visit.
The SA explains the reason for the survey and avoids any impression that a
predetermination has been made as to the validity of the allegation. It is important to let
the facility know why you are there, but to also protect the confidentiality of those
involved in the complaint. Do not release information that will cause opportunities to be
lost for pertinent observations, interviews, and record reviews required for a thorough
investigation. For example, in the case of a hospital, critical access hospital or ambulatory
surgical center, if the complaint is that a patient developed a life-threatening infection in
a post-surgical wound, do not tell the facility the exact complaint. Rather, tell them it is a
situation related to infection control for surgical patients. Another example, in the case
of a long term care facility, would be when a complaint that food that is intended to be
served hot is always served cold. In this case, do not tell the provider the exact
complaint. Rather, tell them it is a situation related to dietary requirements.
(See Section 5300.2 for guidance on the entrance conference for long term care facilities
as well as the provider/supplier-specific appendices of the SOM, and Appendix V of the
SOM concerning EMTALA.)
5080 - Investigation Findings and Reports
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Each SA establishes reporting policies, procedures and formats including report language
targeted to specific audiences.
5080.1 - Report to the Complainant
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
The SA/CMS location provides the complainant a written report of the investigation
findings as a summary record of the investigation.
The following principles guide preparation of the report to the complainant:
• Acknowledge the complainant’s concern(s);
• Identify the SA’s regulatory authority to investigate the complaint/incident and
any statutory or regulatory limits that may bear on the authority to conduct an
investigation;
• Provide a summary of investigation methods (e.g., on-site visit, written
correspondence, telephone inquiries, etc.);
• Provide date(s) of investigation;
• Provide an explanation of your SA’s decision-making process (NOTE: CMS and
the SA should avoid using terms such as “substantiated” and “unsubstantiated”);
• Provide the complainant with information regarding whether or not
noncompliance was identified during the complaint investigation. (NOTE: To
the extent possible, the summary should not compromise the anonymity of
individuals, or include specific situations that may be used to identify individuals,
when anonymity has been requested or is appropriate in the judgment of the SA);
• Identify where the complainant may find the Statement of Deficiencies and Plan of
Correction (e.g., posted at the nursing home, Nursing Home Care Compare,
request the CMS-2567 from the SA);

• Describe how the complainant may request a copy of the investigation report,
subject to Federal and State disclosure requirements (e.g., see 42 CFR §488.325
and FOIA requirements at 45 CFR Part 5); and
• Identify appropriate referral information (i.e., other agencies that may be
involved).
5080.2 - Survey Exit Conference and Report to the Provider/Supplier
(Rev. 155, Issued: 06-10-16, Effective: 06-10-16, Implementation: 06-10-16)
Generally, the SA conducts an exit conference with the provider/supplier at the
completion of the on-site portion of the complaint investigation survey. The SA informs
the provider/supplier of the survey findings, including a general description of any
deficiencies found. The description should be detailed enough to inform the
provider/supplier of the types of activities that require the provider’s/supplier’s corrective
action. However, the SA must not comment on the scope and severity of the deficiencies
identified for long term care facilities. For non-long term care providers/suppliers, the
SA must not comment on manner and degree, that is, whether the deficiencies identified
were condition- or standard-level. Surveyors must also not make reference to any “Tags”
related to deficiencies identified in non-long term care as this identifies condition- or
standard-level. Instead identify the regulatory grouping where concerns exist. See
Section 2724 for additional information about presenting findings during the Exit
Conference.
For non-long term care providers/suppliers, the SA must not provide a list of patients
interviewed, observed, or whose medical records were reviewed, and does not identify
specific patients whose cases are associated with specific deficiencies. (The
provider/supplier has the right to request a copy of any documentation the surveyors copy
to support deficiency findings; therefore the provider/supplier should have enough
information after the exit conference to begin corrective actions.)
The SA informs the provider/supplier that survey findings will be documented on Form
CMS 2567, which will be sent to the provider/supplier and subsequently will be made
available to the public under the disclosure of survey information provisions. For
deemed providers/suppliers, the SA informs the provider/supplier that the RO will be
consulted and (depending on RO practice), either the RO or the SA will inform the
facility of the results of the survey investigation via the Form CMS 2567.
The SA/RO sends to the provider/supplier a written report of the investigation findings as
a summary record of the investigation. At a minimum, this would include the Form CMS
2567 and applicable notices. For surveys of deemed providers/suppliers (not including
EMTALA surveys), the RO sends a copy of the written report to the applicable
accrediting organization(s), following the procedures specified in Section 5110. At the
RO’s or SA’s discretion, the materials may be sent to the accrediting organization via e-
mail.

(See Section 5300.5 for guidance on the exit conference for long term care facilities,
Section 5440.5 for EMTALA investigations, as well as the provider/supplier-specific
appendices of the SOM, and Appendix V of the SOM concerning EMTALA.)
NOTE: Sections 5300 to 5390 relate to nursing homes.
5100 - Investigation of Complaints for Deemed Providers/Suppliers
(Rev. 120, Issued: 09-19-14, Effective: 09-19-14, Implementation: 09-19-14)
Sections 5100 - 5130 apply to all deemed providers and suppliers, with the exception
of clinical laboratories subject to the Clinical Laboratory Improvement
Amendments of 1988 (CLIA). See Sections 5500 – 5590 for CLIA information,
including investigation of complaints related to accredited laboratories.
5100.1 - Basis for Investigation
(Rev. 120, Issued: 09-19-14, Effective: 09-19-14, Implementation: 09-19-14)
Sections 1864(c) and 1865 of the Social Security Act (the Act) provide the basis for
conducting substantial allegation validation – i.e., complaint investigation - surveys of
deemed providers/suppliers. Before the SA may conduct a complaint investigation
survey at a deemed provider/supplier, it must receive authorization to do so from the RO.
In accordance with 42 CFR 488.7, the RO may authorize a complaint investigation only
in response to a “substantial allegation” of noncompliance. A “substantial allegation of
noncompliance” is defined at 42 CFR 488.1 as a complaint from any of a variety of
sources, including complaints submitted in person, by telephone, through written
correspondence, or in news media articles, that, if substantiated, would have an impact on
the health and safety of patients, and that raises doubts as to a provider’s or supplier’s
compliance with any Medicare condition. In other words, the complaint, if verified and
uncorrected at the time of the survey, would result in a condition-level deficiency
citation. The SA survey conducted in response to a substantial allegation is one type of
validation survey.
NOTE: Deemed status is irrelevant for EMTALA complaints. Hospitals and CAHs may
not be deemed to be in compliance with EMTALA requirements at 42 CFR 489.24 and
the related requirements at 42 CFR 489.20, since these requirements are not part of an
approved Medicare hospital or CAH Medicare accreditation program. SAs must refer all
EMTALA-related allegations concerning a hospital or CAH to the RO, regardless of
whether the hospital or CAH is deemed or not. The provisions of Section 5100 do not
apply to EMTALA investigations.
The SA must notify the RO of all complaints/incidents it receives which, if substantiated,
would by their manner and degree suggest condition-level noncompliance. The RO
authorizes the SA to conduct a complaint investigation if it concurs that the nature of the
allegation, if it were true and uncorrected, suggests condition-level noncompliance. If the
RO does not concur that the allegation rises to this level, either the RO will change the
prioritization of the intake in ACTS to the appropriate level or it will instruct the SA to

do so. Regardless of who makes the change in ACTS, the RO instructs the SA to refer
the complainant to the applicable accrediting organization, following the procedures in
section 5100.2
The RO communicates its authorization to conduct a complaint investigation of the
deemed provider/supplier by completing the applicable Form CMS 2802 (See Exhibit 33)
in ACTS, indicating which Conditions of Participation or Conditions for Coverage or
Certification are to be investigated by the SA. Absent RO authorization, the SA may not
conduct a Federal complaint investigation of the deemed provider/supplier. The SA may
have authority under State law to conduct its own non-Federal investigation.
The RO completes the Form CMS 2802 in ACTS even if the SA received an initial verbal
authorization from the RO to initiate the complaint survey of a deemed provider/supplier.
Since ACTS allows the RO to authorize a complaint survey electronically it is not
necessary for the RO to send a signed hard copy of the Form CMS 2802 to the SA via fax
or U.S. Postal Service. Once the SA receives the authorization, it may begin its
complaint investigation of a deemed provider/supplier. Whether the survey is of one or
all Medicare conditions, it will be treated as a complaint survey under ACTS rather than a
re-certification survey, since the complaint/incident is the basis for the survey.
If the RO learns directly of a complaint/incident concerning a deemed provider/supplier,
it will review the complaint/incident to assign a priority consistent with Section 5075. If
the complaint/incident is found to be a substantial allegation of noncompliance,
prioritized for investigation as either immediate jeopardy or non-IJ high, the RO
authorizes the SA to conduct a complaint investigation or, in a limited number of cases,
the RO conducts the complaint investigation.
There may be occasions during the course of a State-only activity in a deemed
provider/supplier when State surveyors observe a situation they believe may constitute IJ
or other substantial noncompliance with a Medicare condition. In such circumstances,
the State must contact the RO by telephone or e-mail, explain the situation, and request
authorization to conduct a Federal complaint survey. CMS authorizes the investigation
as a complaint validation (i.e., substantial allegation validation) survey if it concurs that
there may be condition-level noncompliance. The complaint is entered into ACTS at the
earliest possible opportunity.
5100.2 – Initial Response to Complainant
(Rev. 120, Issued: 09-19-14, Effective: 09-19-14, Implementation: 09-19-14)
• If the SA concludes that a complaint represents a substantial allegation of
noncompliance (i.e., it is appropriately triaged as an IJ or non-IJ high), it requests
authorization in ACTS from the RO to conduct a survey. If the RO authorizes a
survey, the SA acknowledges receipt of the complaint by a letter to the complainant,
and advises that a SA investigation will be initiated. The acknowledgment letter also
advises that the complainant may also wish to file a complaint with the applicable
accrediting organization (AO), naming the AO and attaching a current list of AOs and

their contact information. This list may be found at:
https://www.cms.gov/Medicare/Provider-Enrollment-and-
Certification/SurveyCertificationGenInfo/Downloads/Accrediting-Organization-
Complaint-Contacts.pdf
• If the SA concludes that a complaint does not represent a substantial allegation of
noncompliance (i.e., it is appropriately triaged as non-IJ medium or low) the SA
sends the complainant a letter indicating that the complaint does not meet the criteria
for a Federal on-site investigation of an accredited health care facility. The letter also
advises the complainant which AO(s) accredit the provider/supplier for Medicare
participation purposes and provides the above AO contact information, should the
individual wish to pursue a complaint with the AO.
If the RO directly receives a complaint, it is responsible for sending the complainant a
letter which acknowledges the receipt of the complaint and advises the complainant in the
same manner as indicated above for complaints received by the SA.
5110 - Post-Survey Procedures
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
5110.1 - Substantial Compliance
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
If a condition-level deficiency is not cited at a survey, the provider/supplier is in
substantial compliance with the Federal requirements. The SA certifies its survey
findings in ACTS within 30 calendar days after the completion of the survey. A Form
CMS 2567 is prepared in all cases. Even if no deficiencies were cited, the Form CMS
2567 is issued with a statement that a survey was conducted to evaluate compliance with
the listed requirements identified on the CMS-2802 and that no deficiencies were
identified in these areas.
The RO randomly selects several Form CMS 2567s with standard-level deficiencies for
review to determine if the RO concurs with the SA’s finding of substantial compliance.
• For all cases not selected for review of the Form CMS 2567, the RO completes
the processing in ACTS of the complaint investigation Form CMS 562 and other
applicable items in ACTS, and then, depending on RO practice, either the SA or
RO uploads the complaint survey package into CASPER, the CMS National
Reporting System.
• For cases selected for review of the Form CMS 2567:
• If the RO concurs with the finding, the RO completes the processing in
ACTS of the complaint investigation Form CMS 562 and other applicable
items in ACTS, and then, depending on RO practice, either the SA or RO
uploads the complaint survey package into CASPER, the CMS National
Reporting System.
• If the RO does not concur with the SA’s findings of substantial
compliance, the RO discusses with the SA any revisions needed on the
Form CMS 2567 and, after the revisions are made, follows the procedures
for a survey finding substantial noncompliance. (See Section 5110.2 or
5110.3, as applicable.)
The RO either issues a notice, or authorizes the SA in ACTS to issue the
provider/supplier a notice of its compliance status. The RO or SA, as applicable, issues
this notice to the provider/supplier, along with a copy of the Form CMS 2567containing
the survey findings. The notice indicates that the provider/supplier was found to be in
substantial compliance even though there may, or may not, also be standard-level
deficiencies cited. In such circumstances, the provider/supplier is not required to submit
a plan of correction for any cited standard-level deficiencies, but may choose to do so
because the Form CMS 2567 is available to the public. The SA and RO do not review
any plan of correction the provider/supplier submits; no revisit survey is conducted. The
RO promptly sends a copy of the notice letter and Form CMS 2567 to the applicable
AO(s). At the RO’s or SA’s discretion, the materials may be sent to the AO via e-mail.
5110.2 - Condition-Level, IJ
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
1. IJ Removed while the SA is On-site
If deficiencies pose an IJ and the IJ is removed while the SA is on-site, deficiency
citations are made at the condition-level. Follow the procedure for condition-level
noncompliance, non-IJ, in Section 5110.3 below.
Note: The Form CMS 2567 must state at the beginning that an IJ was cited, regardless of
the fact that it was removed while the SA was on-site. An entry in ASPEN/ACTS must
also be made by the RO indicating that there was an IJ citation before the survey can be
uploaded to the national database, the CMS National Reporting System (CASPER). The
ASPEN/ACTS systems will prompt the RO whenever a survey includes condition-level
deficiencies to indicate whether there was also an IJ.
Details of the IJ situation and the actions taken by the provider/supplier to remove the IJ
must also be documented on the Form CMS 2567. Even though the IJ was removed
while the SA was on-site, the provider or supplier must still be cited for condition-level
noncompliance for the applicable Condition of Participation, Condition for Coverage, or
Condition for Certification that is cited for non-compliance related to the IJ. The
documentation must also include the date the surveyors verified that the IJ was removed
prior to completion of the survey.
2. IJ not Removed while the SA is On-site
If condition-level deficiencies pose an IJ and the IJ is not removed while the SA is on-
site, the SA certifies its findings in ACTS within 2 working days after the completion of
the survey.
If the RO concurs with the SA’s findings, the deemed provider/supplier is placed on a 23
calendar-day termination track. The RO sends the provider/supplier the Form CMS
2567, notifies the provider/supplier of the proposed termination action and effective date,
which is 23 calendar days after the date of the RO’s notice, and requests submission of an
acceptable plan of correction to the RO within 5 calendar days of the notice. The
provider/supplier is advised it will be surveyed after receipt of an acceptable plan of
correction and prior to the termination date. The notice also contains a statement that
“removes” the “deemed status” of the provider/supplier and places it under SA
jurisdiction.
The RO sends a copy of the notice to the SA and a copy of the notice and Form CMS
2567 to the applicable AO(s). At the RO’s discretion, the materials may be sent to the
AO via e-mail.
Note: Although deemed status technically has been removed and the provider/supplier is
placed under SA jurisdiction, because it is expected that deemed status will be restored
once corrections are made and verified, no change is made in ASPEN to the
provider’s/supplier’s deemed status. However, periods during which the
provider/supplier is under SA jurisdiction are entered and tracked in ASPEN. Further,
the AO may also conduct a survey of the facility, so long as it continues to accredit the
provider/supplier under its approved Medicare accreditation program.
When the RO receives a timely and acceptable plan of correction from the
provider/supplier, it directs the SA to conduct either a full survey or an IJ follow-up
survey, which is a focused, revisit-type survey, before the scheduled termination date in
order to confirm that the IJ has been removed and that the provider/supplier is in
substantial compliance. See Section 5110.3 for a discussion of factors the RO should
consider when deciding whether a full survey is needed. If the RO authorizes a full
survey, see Section 5110.4 for procedures to follow, except that the full survey must be
conducted prior to the 23-day termination date.
i. No Acceptable Plan of Correction Submitted
No revisit is necessary if the provider/supplier fails to submit a timely and
acceptable plan of correction. CMS will proceed to terminate a provider/supplier
if it does not submit a timely and acceptable POC. See SOM Section 3254F. The
public notice must be published 15 calendar days prior to the termination date.
The RO notifies the applicable Medicare Administrative Contractor (MAC) via
the Form CMS 2007 of the termination of the provider/supplier’s Medicare
agreement, including the effective date of the termination.
The RO completes the processing in ACTS of the complaint investigation Form
CMS 562 and other applicable items in ACTS and then, depending on RO
practice, either the SA or RO uploads the complaint survey package into
CASPER, the CMS National Reporting System.
The RO sends a copy of the termination letter to the applicable AO(s). At the
RO’s discretion, the copy may be sent to the AO via e-mail.
ii. Post-IJ First Revisit: IJ Not Removed
At least 5 calendar days in advance of the scheduled termination date, the SA
certifies to the RO in ACTs its findings, based on on-site verification, that the IJ
has not been removed, and recommends that the termination action proceed.
The RO reviews the SA’s findings, and if it concurs with the SA’s
recommendation, the RO completes the processing in ACTS of the complaint
investigation Form CMS 562 and other applicable items in ACTS and then,
depending on RO practice, either the SA or RO uploads the complaint survey
package into the CMS National Reporting System (CASPER). The termination of
the provider’s/supplier’s Medicare agreement is processed in ASPEN.
The RO sends the provider/supplier a final termination letter and publishes a
public notice, in accordance with the termination process in Section 3010B. The
provider/supplier is terminated from the Medicare program. The RO notifies the
applicable Medicare Administrative Contractor (MAC) via the Form CMS 2007
of the termination of the provider/supplier’s Medicare agreement, including the
effective date of the termination.
The RO sends a copy of the termination notice and the Form CMS 2567 to the
applicable AO(s). At the RO’s discretion, the materials may be sent to the AO via
e-mail.
iii. Post-IJ First Revisit: IJ Removed, Substantial Compliance
The termination action is rescinded if the IJ has been removed and substantial
compliance has been achieved prior to the effective date of the termination, i.e.,
there are no condition-level deficiencies identified during the follow-up survey by
the SA. The SA certifies its findings to the RO via ACTS at least 5 calendar days
in advance of the scheduled termination date, and recommends that the
termination action be rescinded.
The RO randomly selects several Form CMS 2567s with standard-level
deficiencies for review, to determine if the RO concurs with the SA’s finding of
substantial compliance. If the RO concurs, and in all other cases where the Form
CMS 2567 is not reviewed by the RO, the RO completes the processing in ACTS
of the complaint investigation Form CMS 562 and other applicable items in
ACTS, and then, depending on RO practice, either the SA or RO uploads the
complaint survey package into CASPER, the CMS National Reporting System. If
the RO does not concur with the SA’s finding, the RO discusses with the SA any
revisions needed on the Form CMS 2567 and, after the revisions are made,
follows the procedures for substantial noncompliance.
When substantial compliance is achieved, the RO either issues a notice, or
authorizes the SA in ACTS to issue the provider/supplier a notice of its
compliance status, restoring its deemed status. The RO or SA, as applicable,
issues this notice to the provider/supplier, along with a copy of the Form CMS
2567 containing the survey findings.
In addition, the RO sends a copy of the notice letter to the applicable AO(s). At
the RO’s discretion, the copy may be sent to the AO via e-mail.
Although the follow-up survey found the provider/supplier to be in substantial
compliance, it may have resulted in citation(s) of standard-level deficiencies of
the Form CMS 2567. Because deemed status has been restored, the
provider/supplier is not obligated to submit a plan of correction to the SA, nor are
any further revisits conducted. The provider/supplier may voluntarily choose to
submit a plan of correction because the Form CMS 2567 will be made available to
the public. The SA and RO do not review any plan of correction the
provider/supplier submits; no further revisit survey is conducted.
iv. Post-IJ First Revisit: IJ Removed, Substantial Noncompliance Remains
If the IJ has been removed but substantial noncompliance (i.e., condition-level
deficiencies), remain, the SA certifies its findings to the RO in ACTS within 10
working days after the survey completion date. The SA certifies that the IJ has
been removed and recommends rescission of the 23 calendar-day IJ termination
action, but continuation of the termination action on a 90 calendar-day
termination track.
The RO reviews the SA’s findings, and if it concurs with the SA’s
recommendation, the RO gives the provider/supplier up to 67 additional calendar
days, or a total of 90 calendar days (23 plus 67) to achieve substantial compliance.
The resulting revised termination date is 90 calendar days after the date of the
RO’s original 23-day notice. The RO sends the CMS Form 2567 from the follow-
up survey to the provider/supplier with notice of the new termination date, and
requests that an acceptable POC be provided to the SA within 10 calendar days of
the notice.
Post-IJ Second Revisit: The SA conducts the second revisit survey by the 60th
calendar day after the date of the RO’s original 23-day termination notice. Unlike the
post-IJ first revisit survey, advance authorization from the RO is not required.
(i) Post-IJ Second Revisit Survey Findings: Substantial Compliance
The termination action is rescinded if substantial compliance (i.e., no
condition-level deficiencies) is achieved and documented through the onsite
verification at the revisit survey. There may or may not be standard-level
deficiencies cited. Within 10 working days after the completion of the revisit
survey, the SA certifies to the RO its findings via ACTS and recommends that
the termination action be rescinded.
The RO randomly selects some Form CMS 2567s with only standard-level
deficiencies for review, to determine if the RO concurs with the SA’s finding
of substantial compliance. If the RO concurs, and in all other cases where the
Form CMS 2567 is not reviewed by the RO, the RO completes the processing
in ACTS of the complaint investigation Form CMS 562 and other applicable
items in ACTS and then, depending on RO practice, either the SA or the RO
uploads the complaint survey package into CASPER, the CMS National
Reporting System. If the RO does not concur, the RO discusses with the SA
revisions needed on the Form CMS 2567 and, after the revisions are made,
follows the procedures for substantial noncompliance.
The RO either issues a notice, or authorizes the SA in ACTS to issue a notice
to the provider/supplier of its compliance status and that its deemed status is
restored. The RO or SA, as applicable, issues this notice to the
provider/supplier, along with a copy of the Form CMS 2567 with the survey
findings.
The RO sends a copy of the notice and Form CMS 2567 to the applicable
AO(s). At the RO’s discretion, the materials may be sent to the AO via e-
mail.
i. Post-IJ Second Revisit Survey Findings: Substantial Noncompliance
If the second revisit shows that the provider/supplier fails to demonstrate
substantial compliance (i.e., condition-level deficiencies are identified through
on-site verification by the SA), the SA certifies to the RO its findings within
10 calendar days after the survey completion date, and recommends that the
termination action proceed.
The RO reviews the SA’s findings, and if it concurs with the SA’s
recommendation, the RO sends the provider/supplier a final termination letter
and publishes a public notice 15 calendar days prior to the termination date.
The provider/supplier is terminated from the Medicare program. The RO
notifies the applicable Medicare Administrative Contractor (MAC) via the
Form CMS 2007 of the termination of the provider/supplier’s Medicare
agreement, including the effective date of the termination.
The RO completes the processing in ACTS of the complaint investigation
Form CMS 562 and other applicable items in ACTS and then, depending on
RO practice, either the SA or RO uploads the complaint survey package into
CASPER. The provider’s or supplier’s Medicare agreement is terminated in
ASPEN.
The RO sends a copy of the final termination notice and Form CMS 2567 to
the applicable AO(s). At the RO’s discretion, the materials may be sent to the
AO via e-mail.
5110.3 - Condition-Level, Non-IJ
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
If the provider/supplier fails to demonstrate substantial compliance, i.e., condition-level
deficiencies are identified by the SA, but they do not pose an IJ, the SA certifies its
findings to the RO via ACTS within 10 working days after the survey completion date.
The RO reviews the SA’s findings, and if it concurs with the SA’s recommendation, the
RO either places the deemed provider/supplier on a 90 calendar-day termination track or
it requires a full survey after a complaint survey.
In determining whether to exercise its discretion to require a full survey for deemed
providers and suppliers, the RO may consider factors including, but not limited to, the
following:
• The manner and degree of noncompliance identified as a result of the complaint
investigation;
• The provider’s/supplier’s compliance history;
• Recent changes in the provider’s/supplier’s ownership or management;
• The length of time since the provider’s/supplier’s last accreditation survey;
• The availability of SA resources at the time required to conduct a full survey;
and/or
• The advantages associated with conducting a more extensive survey compared to
the advantages associated with the faster enforcement (and thus a faster potential
corrective action) that result when proceeding directly to enforcement action after
the complaint survey.
Paragraph a) below discusses the procedures when the RO does not require a full
survey after the complaint survey; paragraph b) discusses the procedures to follow
when the RO directs the SA to conduct a full survey.
a) No full survey – proceed directly to termination track based on the complaint
survey
If the RO places the deemed provider/supplier on a 90 calendar-day termination track
as a result of the complaint investigation, it sends the provider/supplier the Form
CMS 2567, notifies the provider/supplier of the proposed termination action and
effective date, which will be 90 calendar days after the date of the RO’s notice. The
RO requests submission of an acceptable plan of correction to the SA within 10
calendar days. The notice also contains a statement that “removes” the “deemed
status” of the provider/supplier and places it under SA jurisdiction.
The RO sends a copy of the notice to the SA and a copy of the notice and Form CMS
2567 to the applicable AO(s). At the RO’s discretion, the materials may be sent to
the AO via e-mail.
NOTE: Although deemed status has technically been “removed” and the
provider/supplier is placed under SA jurisdiction, because it is expected that deemed
status will be restored once corrections are made and verified, no change is made in
ASPEN to the provider’s/supplier’s deemed status. However, periods during which
the provider/supplier is under SA jurisdiction are entered and tracked in ASPEN.
Further, the AO may also conduct a survey of the facility so long as it continues to
accredit the provider/supplier.
The SA conducts a complaint survey revisit after the SA has received a timely and
acceptable plan of correction, but no later than the 45th calendar day after the notice
to the provider/supplier.
1) No Timely, Acceptable Plan of Correction Submitted
If the provider/supplier fails to submit a timely and acceptable plan of correction
to the SA and as a result the SA is unable to conduct a timely revisit before the
termination date, the SA notifies the RO and the RO may proceed with
termination. See SOM Section 3254F. The RO publishes a public notice 15 days
prior to the termination date. The RO notifies the applicable Medicare
Administrative Contractor (MAC) via the Form CMS 2007 of the termination of
the provider/supplier’s Medicare agreement, including the effective date of the
termination.
The RO approves the complaint investigation Form CMS 562 and other
applicable items in ACTS and then, depending on RO practice, either the SA or
RO uploads the complaint survey package into the CMS National Reporting
System (CASPER). The provider’s or supplier’s Medicare agreement is
terminated in ASPEN.
Additionally, the RO sends a copy of the notice of termination letter to the
applicable AO(s).
2) First Revisit Survey Findings: Substantial Compliance
The termination action is rescinded if substantial compliance (i.e., no condition-
level deficiencies) is achieved and documented through the onsite verification at
the first revisit survey. There may or may not be standard-level deficiencies cited.
Within 10 working days after the completion of the revisit survey, the SA certifies
to the RO in ACTS its findings and recommends that the termination action be
rescinded.
The RO randomly selects several Form CMS 2567s with standard-level
deficiencies for review to determine if the RO concurs with the SA’s finding of
substantial compliance.
• For all cases not selected for review of the Form CMS 2567, the RO
completes the processing in ACTS of the complaint investigation Form CMS
562 and other applicable items in ACTS, and then, depending on RO practice,
either the SA or RO uploads the complaint survey package into CASPER, the
CMS National Reporting System.
• For cases selected for review of the Form CMS 2567:
• If the RO concurs with the finding, the RO completes the processing in
ACTS of the complaint investigation Form CMS 562 and other
applicable items in ACTS, and then, depending on RO practice, either
the SA or RO uploads the complaint survey package into CASPER,
the CMS National Reporting System.
• If the RO does not concur with the SA’s findings of substantial
compliance, the RO discusses with the SA any revisions needed on the
Form CMS 2567 and, after the revisions are made, follows the
procedures for a survey finding substantial noncompliance. (See
Section 5110.2 or 5110.3, as applicable.)
The RO either issues a notice, or authorizes the SA in ACTS to issue a notice to
the provider/supplier of its compliance status and that its deemed status is
restored. The RO or SA, as applicable, issues this notice to the provider/supplier,
along with a copy of the Form CMS 2567 with the survey findings.
The RO sends a copy of the notice and Form CMS 2567 to the applicable AO(s).
At the RO’s discretion, the materials may be sent to the AO via e-mail.
3) First Revisit Survey Findings: Substantial Noncompliance
If the SA finds during the first revisit survey that the provider/supplier is not in
substantial compliance with one or more Medicare conditions, the SA consults
with the RO on its findings and whether to conduct a second revisit. If the RO
agrees that condition-level deficiencies remain, the RO considers whether the
survey findings warrant a second revisit or proceeding immediately to
termination. Generally the RO authorizes a second revisit, but the RO has
discretion to make an exception, based on the facts of the situation. For example,
if the SA and RO determine that an immediate jeopardy was present during the
first revisit, the RO might find it prudent to proceed to termination without a
second revisit.
If the RO agrees that condition-level deficiencies remain and does not authorize a
second revisit, the RO and SA follow the procedures outlined in paragraph 3ii.
below.
If a second revisit is authorized by the RO, the SA sends the provider/supplier the
Form CMS 2567 for the first revisit with notice that substantial noncompliance
remains, the 90-day termination date remains in effect, a new acceptable plan of
correction is required, and that an additional revisit will be conducted prior to the
termination date. The SA conducts the second revisit no later than 60 calendar
days after the date of the termination notice.
i. Second Revisit Survey Findings: Substantial Compliance
If substantial compliance is achieved by the provider/supplier (i.e., no
condition-level deficiencies are identified through the on-site verification by
the SA), the SA certifies its findings to the RO via ACTS within 10 working
days after the survey completion date, and recommends that the termination
action be rescinded.
The RO randomly selects some Form CMS 2567s with only standard-level
deficiencies for review, to determine if the RO concurs with the SA’s finding
of substantial compliance. If the RO concurs, and in all other cases where the
Form CMS 2567 is not reviewed by the RO, RO completes the processing in
ACTS of the complaint investigation Form CMS 562 and other applicable
items in ACTS and then, depending on RO practice, either the SA or RO
uploads the complaint survey package into CASPER, the CMS National
Reporting System. If the RO does not concur, the RO discusses with the SA
revisions needed on the Form CMS 2567 and, after the revisions are made,
follows the procedures for substantial noncompliance.
The RO either issues a notice, or authorizes the SA in ACTS to issue the
provider/supplier a notice letter of its compliance status and that its deemed
status is restored. The RO or SA, as applicable, forwards this notice to the
provider/supplier, along with a copy of the Form CMS 2567 with the survey
findings.
Additionally, the RO sends a copy of the notice and the Form CMS 2567 to
the applicable AO(s). At the RO’s discretion, the materials may be sent to the
AO via e-mail.
ii. Second Revisit Survey Findings – Substantial Noncompliance
If the second revisit survey shows that the provider/supplier fails to
demonstrate substantial compliance (i.e., condition-level deficiencies are
identified through on-site verification by the SA), the SA certifies its findings
to the RO via ACTS within 10 calendar days after the survey completion date,
and recommends that the termination action proceed.
The RO reviews the SA’s findings, and if it concurs with the SA’s
recommendation, the RO sends the provider/supplier a final termination letter
and publishes a public notice at least 15 calendar days prior to the termination
date, consistent with the requirements of Section 3012. The provider/supplier
is terminated from the Medicare program. The RO notifies the applicable
Medicare Administrative Contractor (MAC) via the Form CMS 2007 of the
termination of the provider/supplier’s Medicare agreement, including the
effective date of the termination.
The RO completes the processing in ACTS of the complaint investigation
Form CMS 562 and other applicable items in ACTS and then, depending on
RO practice, either the SA or RO uploads the complaint survey package into
CASPER, the CMS National Reporting System. The provider’s or supplier’s
Medicare agreement is terminated in ASPEN.
Additionally, the RO sends a copy of the termination notice and the Form
CMS 2567 to the applicable AO(s). At the RO’s discretion, the materials may
be sent to the AO via e-mail.
b) Full Survey After the Complaint Survey
If the RO directs the SA to conduct a full survey following the complaint survey, it
sends the Form CMS 2567 for the complaint survey to the provider/supplier in
addition to a notice letter indicating that it is “removing” the provider’s/supplier’s
deemed status and that a full survey will be conducted on an unannounced basis.
The provider/supplier is not required to submit a plan of correction in response to the
complaint survey findings, but may choose to do so.
The RO completes the processing in ACTS of the complaint investigation Form CMS
562 and other applicable items in ACTS and then, depending on RO practice, either
the SA or RO uploads the complaint survey package into CASPER, the CMS
National Reporting System.
Additionally, the RO sends a copy of the notice letter and Form CMS 2567 for the
complaint survey to the applicable AO(s). At the RO’s discretion, the materials may
be sent to the AO via e-mail.
NOTE: Although deemed status technically has been removed and the
provider/supplier is placed under SA jurisdiction, because it is expected that deemed
status will be restored once corrections are made and verified, no change is made in
ASPEN to the provider’s/supplier’s deemed status. However, periods during which
the provider/supplier is under SA jurisdiction are entered and tracked in ASPEN.
Further, the AO may also conduct a survey of the facility so long as, since it
continues to accredit the provider/supplier.
The full survey must be conducted within 60 calendar days after the RO’s notice
to the provider/supplier of the complaint survey results and removal of deemed
status. The RO and SA follow the procedures in Section 5110.4.
5110.4 - Full Survey after Complaint Survey with Condition-level
Deficiencies, When Authorized by the RO
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
If the RO authorizes the SA to conduct a full survey after the complaint survey, the
timeframes and procedures described in this section apply.
Timeframe
The full survey must be conducted within:
• 23 days after the RO’s notice to the provider/supplier, if the complaint survey
involved an IJ that was not removed while the survey team was on-site; or
• 60 calendar days after the RO’s notice to the provider/supplier in all other cases.
Procedures following the full survey with findings of:
a) Full Survey Findings: Substantial Compliance
If the SA full survey finds the deemed provider or supplier to be in substantial
compliance, the SA and RO follow the same procedures and timeline as at Section
5110.1. In addition, since the RO had removed deemed status, the RO either issues a
notice, or authorizes the SA to issue a notice to the provider or supplier of its compliance
status and that its deemed status is restored, along with a copy of the Form CMS 2567
with the survey findings.
The RO sends a copy of the notice and Form CMS 2567 to the applicable AO(s). At the
RO’s discretion, the materials may be sent to the AO via e-mail.
b) Full Survey Findings: Condition-Level, IJ
1. IJ Removed while the SA is On-site
If deficiencies pose an IJ and the IJ is removed while the SA is on-site, deficiency
citations are made at the condition-level. Follow the procedure for condition-
level noncompliance, non-IJ, in Section 5110.4c below.
Note: The Form CMS 2567 must state at the beginning that an IJ was cited,
regardless of the fact that it was removed while the SA was on-site. An entry in
ASPEN must also be made by the RO indicating that there was an IJ citation
before the survey can be uploaded to the national database, the CMS National
Reporting System (CASPER). The ASPEN systems will prompt the RO
whenever a survey includes condition-level deficiencies to indicate whether there
was also an IJ.
Details of the IJ situation and the actions taken by the provider/supplier to remove
the IJ must also be documented on the Form CMS 2567. Even though the IJ was
removed while the SA was on-site, the provider or supplier must still be cited for
condition-level noncompliance for the applicable Condition of Participation or
Condition for Coverage that is cited for non-compliance related to the IJ. The
documentation must also include the date the surveyors verified that the IJ was
removed prior to completion of the survey.
2. IJ not Removed while the SA is On-site
If condition-level deficiencies pose an IJ and the IJ is not removed while the SA is
on-site, the SA certifies its findings to the RO within 2 working days after the
completion of the survey.
If the RO concurs with the SA’s findings, the deemed provider/supplier is placed
on a 23 calendar-day termination track. The RO sends the provider/supplier the
Form CMS 2567, notifies the provider/supplier of the proposed termination action
and effective date, which is 23 calendar days after the date of the RO’s notice, and
requests submission of an acceptable plan of correction to the RO within 5
calendar days of the notice.
The RO sends a copy of the notice to the SA and a copy of the notice and Form
CMS 2567 to the applicable AO(s). At the RO’s discretion, the materials may be
sent to the AO via e-mail.
When the RO receives a timely and acceptable plan of correction from the
provider/supplier, it directs the SA to conduct an IJ follow-up survey before the
rescheduled termination date in order to confirm that the IJ has been removed and
that the provider/supplier complies with the conditions previously cited for
noncompliance.
2.1 First Revisit after Full Survey with IJ
i. No Acceptable Plan of Correction Submitted
No revisit is necessary if the provider/supplier fails to submit a timely and
acceptable plan of correction. CMS will proceed to terminate a provider/supplier
if it does not submit a timely and acceptable POC. See SOM Section 3254F. The
public notice must be published 15 calendar days prior to the termination date.
The RO notifies the applicable Medicare Administrative Contractor (MAC) via
the Form CMS 2007 of the termination of the provider/supplier’s Medicare
agreement, including the effective date of the termination.
The SA and RO complete the processing in ASPEN of the survey kit and then,
depending on RO practice, either the SA or RO uploads the complaint survey
package into CASPER, the CMS National Reporting System.
The RO sends a copy of the termination letter to the applicable AO(s). At the
RO’s discretion, the copy may be sent to the AO via e-mail.
ii. First Revisit Survey Findings: IJ Not Removed
At least 5 calendar days in advance of the scheduled termination date, the SA
certifies to the RO its findings, based on on-site verification, that the IJ has not
been removed, and recommends that the termination action proceed.
The RO reviews the SA’s findings, and if it concurs with the SA’s
recommendation, the SA and RO complete the processing of the survey kit in
ASPEN and then, depending on RO practice, either the SA or RO uploads the
survey package into the CMS National Reporting System (CASPER). The
termination of the provider’s/supplier’s Medicare agreement is processed in
ASPEN.
The RO sends the provider/supplier a final termination letter and publishes a
public notice, in accordance with the termination process in Section 3010B. The
provider or supplier is terminated from the Medicare program. The RO notifies
the applicable Medicare Administrative Contractor (MAC) via the Form CMS
2007 of the termination of the provider/supplier’s Medicare agreement, including
the effective date of the termination.
The RO sends a copy of the termination notice and the Form CMS 2567 to the
applicable AO(s). At the RO’s discretion, the materials may be sent to the AO via
e-mail.
iii. First Revisit Survey Findings: IJ Removed, Substantial Compliance
The termination action is rescinded if the IJ has been removed and substantial
compliance has been achieved prior to the effective date of the termination, i.e.,
there are no condition-level deficiencies identified during the first revisit survey
by the SA. The SA certifies its findings to the RO at least 5 calendar days in
advance of the scheduled termination date, and recommends that the termination
action be rescinded.
The RO randomly selects several Form CMS 2567s with standard-level
deficiencies for review, to determine if the RO concurs with the SA’s finding of
substantial compliance. If the RO concurs, and in all other cases where the Form
CMS 2567 is not reviewed by the RO, depending on RO practice, either the SA or
RO uploads the survey package into CASPER, the CMS National Reporting
System. If the RO does not concur with the SA’s finding, the RO discusses with
the SA any revisions needed on the Form CMS 2567 and, after the revisions are
made, follows the procedures for substantial noncompliance.
When substantial compliance is achieved, the RO either issues a notice, or
authorizes the SA to issue the provider/supplier a notice of its compliance status,
restoring its deemed status, along with a copy of the Form CMS 2567containing
the survey findings.
In addition, the RO sends a copy of the notice letter to the applicable AO(s). At
the RO’s discretion, the copy may be sent to the AO via e-mail.
Although the revisit survey found the provider/supplier to be in substantial
compliance, it may have resulted in citation(s) of standard-level deficiencies of
the Form CMS 2567. Because deemed status has been restored, the provider or
supplier is not obligated to submit a plan of correction to the SA, nor are any
further revisits conducted. The provider or supplier may voluntarily choose to
submit a plan of correction because the Form CMS 2567 will be made available to
the public. The SA and RO do not review any plan of correction the
provider/supplier submits; no further revisit survey is conducted.
iv. First Revisit Survey Findings: IJ Removed, Substantial Noncompliance
Remains
If the IJ has been removed but substantial noncompliance (i.e., condition-level
deficiencies), remains, the SA certifies its findings to the RO within 10 working
days after the survey completion date. If the RO concurs that the IJ has been
removed but that condition-level deficiencies remain, the RO considers whether
the survey findings warrant a second revisit or proceeding immediately to
termination. At this point the provider/supplier will have been surveyed three
times, including the preceding complaint survey, with continued substantial
noncompliance found in each survey and at least one IJ. Generally the RO
authorizes a second revisit, but the RO has discretion to make an exception, based
on the facts of the case, including the risks to patients associated with the
remaining deficiencies versus providing the provider/supplier further opportunity
to correct its problems in a timely manner.
If the RO does not authorize a second revisit, it follows the procedures in
paragraph ii above.
If the authorizes a second revisit, the RO gives the provider/supplier up to 67
additional calendar days, or a total of 90 calendar days (23 plus 67) from the date
of the notice of the IJ, to achieve substantial compliance. The resulting revised
termination date is 90 calendar days after the date of the RO’s original 23-day
termination notice. The RO provides the provider/supplier the Form CMS 2567
for the revisit with notice of the new termination date, and requests that an
acceptable POC be provided to the SA within 10 calendar days of the notice.
2.2 Second Revisit after Full Survey with IJ
The SA conducts the second revisit survey no later than 60 calendar days after the date of
the RO’s 23-day termination notice to the provider or supplier.
i. Second Revisit Survey Findings: Substantial Compliance
If substantial compliance is achieved by the provider/supplier (i.e., no condition-
level deficiencies are identified through the on-site verification by the SA) during
the second revisit survey, the SA certifies its findings to the RO within 10
working days after the survey completion date, and recommends that the
termination action be rescinded.
The RO randomly selects some Form CMS 2567s with standard-level deficiencies
for review, to determine if the RO concurs with the SA’s finding of substantial
compliance. If the RO concurs, and in all other cases where the Form CMS 2567
is not reviewed by the RO, depending on RO practice, either the SA or RO
uploads the survey kit into CASPER, the CMS National Reporting System. If the
RO does not concur, the RO discusses with the SA revisions needed on the Form
CMS 2567 and, after the revisions are made, follows the procedures for
substantial noncompliance.
The RO also either issues a notice, or authorizes the SA to issue the
provider/supplier a notice, of its compliance status and that its deemed status is
restored, along with a copy of the Form CMS 2567 with the survey findings.
Additionally, the RO sends a copy of the notice and the Form CMS 2567 to the
applicable AO(s). At the RO’s discretion, the materials may be sent to the AO via
e-mail.
ii. Second Revisit Survey Findings: Substantial Noncompliance
If the second revisit shows that substantial noncompliance (i.e., condition-level
deficiencies) remain, the SA certifies to the RO its findings within 10 calendar
days after the survey completion date, and recommends that the termination
action proceed.
The RO reviews the SA’s findings, and if it concurs with the SA’s
recommendation, the RO sends the provider or supplier a final termination letter
and publishes a public notice 15 calendar days prior to the termination date. The
provider or supplier is terminated from the Medicare program. The RO notifies
the applicable Medicare Administrative Contractor (MAC) via the Form CMS
2007 of the termination of the provider/supplier’s Medicare agreement, including
the effective date of the termination.
The SA and RO complete the processing in ASPEN of the survey kit and,
depending on RO practice, either the SA or RO uploads the survey package into
CASPER. The provider’s or supplier’s Medicare agreement is terminated in
ASPEN.
The RO sends a copy of the final termination notice and Form CMS 2567 to the
applicable AO(s). At the RO’s discretion, the materials may be sent to the AO via
e-mail.
c) Full Survey Findings: Condition-Level, Non-IJ
If the results of the full survey indicate there is substantial noncompliance (i.e., condition-
level deficiencies), but the deficiencies do not constitute an IJ, the SA certifies its
findings to the RO within 10 working days after the survey completion date.
The RO reviews the SA’s findings, and if it concurs with the SA’s recommendation, the
RO places the provider or supplier on a 90 calendar-day termination track as a result of
the full survey. The RO sends the provider or supplier the Form CMS 2567 and notifies
it of the proposed termination action and effective date, which will be 90 calendar days
after the date of the RO’s notice. The RO requests submission of an acceptable plan of
correction to the SA within 10 calendar days of the notice.
Additionally, the RO sends a copy of the notice of termination letter to the applicable
AO(s).
1. First Revisit: The SA conducts the first revisit survey no later than the 45th calendar
day after the date of the RO’s termination notice to the provider or supplier.
i. First Revisit Survey Findings: Substantial Compliance
The termination action is rescinded if substantial compliance (i.e., no condition-
level deficiencies) is achieved and documented through the onsite verification at
the revisit survey. There may or may not be standard-level deficiencies cited.
Within 10 working days after the completion of the revisit survey, the SA certifies
to the RO its findings and recommends that the termination action be rescinded.
The RO randomly selects some Form CMS 2567s with only standard-level
deficiencies for review, to determine if the RO concurs with the SA’s finding of
substantial compliance. If the RO concurs, and in all other cases where the Form
CMS 2567 is not reviewed by the RO, the RO completes the processing of the
survey kit in ASPEN and then, depending on RO practice, either the SA or the
RO uploads the survey package into CASPER, the CMS National Reporting
System. If the RO does not concur, the RO discusses with the SA revisions
needed on the Form CMS 2567 and, after the revisions are made, follows the
procedures for substantial noncompliance.
The RO either issues a notice, or authorizes the SA to issue a notice to the
provider or supplier of its compliance status and that its deemed status is restored,
along with a copy of the Form CMS 2567 with the survey findings.
The RO sends a copy of the notice and Form CMS 2567 to the applicable AO(s).
At the RO’s discretion, the materials may be sent to the AO via e-mail.
ii. First Revisit Survey Findings: Substantial Noncompliance
If the SA confirms during the first revisit survey that the provider/supplier is not
in substantial compliance with one or more Medicare conditions, the SA consults
with the RO on its findings and whether to conduct a second revisit. If the RO
concurs that condition-level deficiencies remain, the RO considers whether the
survey findings warrant a second revisit or proceeding immediately to
termination. At this point the provider/supplier will have been surveyed three
times, including the complaint survey, the full survey and the first revisit, with
substantial noncompliance found on each survey. Generally the RO authorizes a
second revisit, but the RO has discretion to make an exception, based on the facts
of the case, including the risks to patients associated with the remaining
deficiencies versus providing the provider/supplier further opportunity to correct
its problems in a timely manner.
If the RO does not authorize a second revisit, the RO and SA will follow the
procedures outlined in paragraph 2(ii). below.
If the RO authorizes a second revisit, the SA sends the provider/supplier the Form
CMS 2567 for the first revisit with notice that substantial noncompliance remains,
the 90-day termination date remains in effect, a new acceptable plan of correction
is required, and that an additional revisit will be conducted prior to the
termination date.
2. Second Revisit: The SA conducts the second revisit survey no later than 60 calendar
days after the date of the termination notice to the provider or supplier.
(i) Second Revisit Survey Findings: Substantial Compliance
If substantial compliance is achieved by the provider/supplier (i.e., no
condition-level deficiencies are identified through the on-site verification by
the SA) during the second revisit survey, the SA certifies its findings to the
RO within 10 working days after the survey completion date, and
recommends that the termination action be rescinded.
The RO randomly selects some Form CMS 2567s with standard-level
deficiencies for review, to determine if the RO concurs with the SA’s finding
of substantial compliance. If the RO concurs, and in all other cases where the
Form CMS 2567 is not reviewed by the RO, the RO completes the processing
in ASPEN of the survey kit and then, depending on RO practice, either the SA
or RO uploads the complaint and revisit surveys into CASPER, the CMS
National Reporting System. If the RO does not concur, the RO discusses with
the SA revisions needed on the Form CMS 2567 and, after the revisions are
made, follows the procedures for substantial noncompliance.
The RO also either issues a notice, or authorizes the SA in ACTS to issue the
provider or supplier a notice, of its compliance status and that its deemed
status is restored, along with a copy of the Form CMS 2567 with the survey
findings.
Additionally, the RO sends a copy of the notice and the Form CMS 2567 to
the applicable AO(s). At the RO’s discretion, the materials may be sent to the
AO via e-mail.
(ii) Second Revisit Survey Findings: Substantial Noncompliance
If the second revisit shows that the provider or supplier has substantial
noncompliance (i.e., condition-level deficiencies are identified through on-site
verification by the SA), the SA certifies to the RO its findings within 10
calendar days after the survey completion date, and recommends that the
termination action proceed.
The RO reviews the SA’s findings, and if it concurs with the SA’s
recommendation, the RO sends the provider/supplier a final termination letter
and publishes a public notice 15 calendar days prior to the termination date.
The provider or supplier is terminated from the Medicare program. The RO
notifies the applicable Medicare Administrative Contractor (MAC) via the
Form CMS 2007 of the termination of the provider/supplier’s Medicare
agreement, including the effective date of the termination.
The RO completes the processing in ASPEN of the survey kit and then,
depending on RO practice, either the SA or RO uploads the survey package
into CASPER. The provider’s or supplier’s Medicare agreement is terminated
in ASPEN.
The RO sends a copy of the final termination notice and Form CMS 2567 to the
applicable AO(s). At the RO’s discretion, the materials may be sent to the AO via e-
mail.
5120 - Life Safety Code Guidance for Deemed Providers/Suppliers
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
In most States, an engineer or other fire safety specialist surveys for compliance with the
Life Safety Code (LSC) standard and others survey the remaining standards in the
Physical Environment Condition. If the allegation pertains only to LSC requirements, it
is not necessary to survey the remainder of the Physical Environment Medicare
condition.
5130 – Deemed Provider/ Supplier Refusal of Complaint Investigation
Surveys
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
The SA informs the provider/supplier that refusal to allow a complaint investigation
survey is a basis for termination and exclusion from the Medicare program, in accordance
with Section 1128(b)(12) of the Social Security Act. The SA notifies the RO
immediately of a refusal to allow a complaint investigation survey.
5140 - Complaints Involving HIV-Infected Individuals
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
As direct recipients of Federal funds, providers and suppliers are subject to provisions of
Section 504 of the Federal Rehabilitation Act of 1973. Symptomatic and asymptomatic
individuals who are infected with the human immunodeficiency virus (HIV), or “AIDS
virus,” are protected by the Rehabilitation Act as “individuals with handicaps.”
Therefore, HIV-infected individuals who are provided services, are employed, or are to
be employed by providers and suppliers in Federally-conducted or financed programs or
activities would be treated like anyone else in the workforce, so long as these individuals
do not, on a case-by-case basis, pose a substantial health and safety risk to others, or pose
a performance problem, and are “otherwise qualified.”
A provider participating in the Medicare or Medicaid programs cannot discriminate
against individuals who are HIV-infected so long as these individuals do not, on a case-
by-case basis, pose a substantial health and safety risk to others and so long as the
provider provides comparable services and care to non HIV-infected individuals.
The SA or the RO refers discrimination complaints to the Office of Civil Rights (OCR),
which is the authority to determine whether Medicare or Medicaid providers and
suppliers comply with this non-discrimination statute.

5150 - Investigating Complaints Involving ESRD Services Provided by
Deemed Hospitals or CAHs
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
Many of the hospitals or CAHs participating in the ESRD program are deemed to meet
the Medicare Conditions of Participation on the basis of their accreditation by a CMS-
approved Medicare accreditation program. “Deemed status” applies only to the
hospital’s or CAH’s approval as a provider, not to its status as a supplier of ESRD
transplantation or dialysis services. The SA investigates all complaints and allegations
related solely to ESRD services since ESRD services fall outside the purview of
accreditation.
5160 - Investigating Complaints Against ESRD Suppliers
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
1. General
Refer to the guidance for investigation of complaints against non-deemed providers and
suppliers. See SOM §5200.
The ESRD Networks are required to have a complaint /grievance resolution system.
Networks (NW) and the SA are frequently contacted by the same complainant with the
same or similar allegations. If the allegations require an onsite investigation or allege
potential risk to patient health or safety, the SA is responsible for the investigation. If the
allegations are primarily focused on relationship or communication issues, the NW may
assume primary responsibility for the investigation. If the focus of the allegations is a
medical practice issue, the SA and NW may need to collaborate on the investigation. The
NWs and SA are encouraged to communicate and collaborate to reduce or prevent
redundant investigations.
2. Conducting the ESRD Investigation
The SA surveyors must use the ESRD survey protocol in Appendix H to investigate
complaints. The allegations of the complaint will determine the tasks needed. For
example, an allegation of inadequate patient care staffing would require use of the
following tasks, at a minimum:
• Pre survey activities;
• Entrance Conference: Provide an overview of the complaint allegations and the
planned agenda for your survey time;
• Tour and observations;
• Patient interviews;
• Staff interviews;
• Record reviews;
• Review of quality management materials; and
• Exit conference.
Conduct each of the identified survey tasks in Appendix H, “Guidance to Surveyors:
End-Stage Renal Disease Facilities.”
3. Pre-survey Task for ESRD Complaint Investigations
Review the allegations of the complaint to identify needed survey tasks. Review the
State Outcomes List and the Dialysis Facility Report to determine if there are data
outliers related to the allegations. For example, if the complaint alleges staff members do
not wash their hands, the surveyor should review the facility’s rate of hospitalization and
hospitalizations related to septicemia, and consider this information in the survey process.
To facilitate meeting the requirement of surveying each ESRD facility every 3 years, the
SA evaluates all available information (outcome list rank, Dialysis Facility Report, time
since last survey, complaint history, NW information, etc.) to determine whether a
recertification survey should be conducted at the time of the complaint investigation.
5170 – Hospital Restraints/Seclusion Death Reporting and Investigation
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
This section applies to both deemed and non-deemed hospitals, as well as to deemed and
non-deemed CAH distinct part psychiatric and rehabilitation units.
5170.1 - Background
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
The Medicare hospital restraint and seclusion requirements are found under the Patients’
Rights provisions at 42 CFR 482.13(e),(f) and (g).
Hospitals are required to report a death associated with the use of restraint/seclusion to
their CMS RO in accordance with 42 CFR 482.13(g)(1).
The interpretive guidelines found in the Hospital Appendix A at 42 CFR
482.13(e) – (g) discuss in detail what is considered a restraint or seclusion, the
requirements governing hospital use of restraint or seclusion, and these reporting
requirements.
5170.2 - Responsibilities
(Rev. 88, Issued: 08-27-13, Effective: 07-19-13, Implementation: 7-19-13)
Regional Offices (ROs)
The RO receives Hospital Restraint/Seclusion Death Reports which hospitals are required
to submit in accordance with 42 CFR 482.13(g)(1). The RO is responsible for
communicating with hospitals in its region whether the required reports are to be
submitted electronically by facsimile and/or e-mail, providing appropriate addresses or
fax numbers, or whether it will also accept mail submissions.
The RO is also responsible for data entry of reports received into the Automated Survey
Processing Environment (ASPEN) Complaint Tracking System (ACTS) Hospital
Restraint/Seclusion Death Module and for maintenance in ACTS of information related
to disclosures to Protection and Advocacy Agencies. (See Process discussion below.)
Each RO designates one contact person and a backup person who serves as the hospital
point of contact regarding reporting, and who is responsible for coordinating the review
of reports received, and authorization of complaint surveys when appropriate.
State Agencies (SAs)
Hospitals report patient deaths associated with restraint or seclusion to their CMS RO,
not to the SA. Any hospital patient restraint or seclusion death report received by a SA
directly from a hospital (or other source) must be forwarded immediately by the SA to its
RO.
The SA conducts a complaint investigation related to a patient death associated with a
hospital’s use of restraints or seclusion only when the RO authorizes the investigation.
The investigation must be completed no later than five working days after RO
authorization.
SAs assist ROs in educating the hospitals in their State about their obligation to report to
their RO any death that meets the reporting requirements found at 42 CFR 482.13(g)(1).
Upon request, SAs are to provide hospitals with the applicable RO contact information,
as well as the hospital reporting procedures contained in this policy.
The SAs respond to requests from Protection and Advocacy (P&A)
organizations, or any other parties, for information on survey findings related to
specific cases identified by the requestor. The SAs handle these requests in
accordance with standard CMS policy on disclosure of Federal survey
information.
5170.3 - Process
(Rev. 191, Issued: 07-19-19, Effective: 07-19-19, Implementation: 07-19-19)

The RO evaluates the information required to be reported by the hospital or CAH DPU
under 42 CFR 482.13(g)(1) to determine whether the situation might involve a violation
of 42 CFR 482.13(e) through 42 CFR 482.13(g) and authorizes an on-site investigation if
there appears to be a possible violation.
Using the information provided by the hospital or CAH DPU in the worksheet, the RO
evaluates whether the case warrants an on-site investigation. If the RO determines that
the restraint/seclusion death report requires on-site investigation, within two business
days of receiving the report, the RO enters the reported information into the ACTS
restraint/seclusion module and immediately notifies the SA to authorize a complaint
survey to investigate the hospital’s or CAH DPU’s compliance with the Patient’s Rights
requirement at 42 CFR 482.13(e), (f), and (g), including the reported case. The SA
accesses the ACTS restraint/seclusion module to see the information reported by the
hospital or CAH DPU prior to conducting the on-site investigation. The SA is expected
to be onsite to initiate the investigation within two business days of receipt of survey
authorization from the RO.
Notice to Protection and Advocacy Organizations
At the same time that the RO notifies the SA that it authorizes the on-site survey,
consistent with the ACTS Notice of a Modified or Altered System of Records (SOR) (71
FR 29643, May 23, 2006, SOR 09-70-0565), the RO also provides written notification,
by mail or email, to the appropriate Protection and Advocacy Organization (P&A) within
the State where the hospital is located, only if the P&A has a current Data Use
Agreement (DUA) with CMS. The RO may contact CMS Central Office for a list of
P&A’s with current DUAs. The names and addresses for each State’s P&A can be
located at the following website, at the drop down menu entitled “Get Help in Your
State:” www.ndrn.org . Notification is provided only in those cases for which an on-
site survey is authorized.
The RO provides the following information to the P&A: hospital or CAH DPU name,
hospital or CAH DPU address, name of the deceased, and a copy of the
restraint/seclusion death report submitted by the hospital or CAH DPU. An entry must
be made on the intake in ACTS indicating the name of the P&A to which the
restraint/seclusion death report data was sent and the date it was sent.
The P&A must have an approved CMS Data Use Agreement (DUA), Form CMS-R-
0235, (Exhibit 292) in place before restraint/seclusion death report data may be disclosed
to it. In order to get an approved DUA, the P&A must complete and submit a signed
CMS DUA, Form CMS-R-0235, including an initialed DUA ACTS SOR- P&A
Attachment (Exhibit 293) to the Director, Division of Information Security and Privacy
Management (DISPM), Centers for Medicare and Medicaid Services, Mailstop N2-04-27,
7500 Security Boulevard, Baltimore, MD 21244-1850. DISPM will review the DUA,
assign a unique DUA identifier and expiration date to it, and return a signed copy to the
P&A, including an expiration date. CMS Central Office Survey and Certification will
maintain and make available to ROs a list of P&As with DUAs.
When completing the Form, P&As must note the following in particular:

• Line 5 of the DUA must state “Restraint/Seclusion Hospital Death Reports.” The
“Years” and “System of Record” columns should be left blank;
• Line 12 must state “CMS DUA: ACTS SOR Attachment – P&A;”
• The DUA must be signed by the P&A official whom the P&A designates as
“Custodian,” i.e., the individual who will have actual possession of and
responsibility for the data released under the DUA; and
• A P&A may designate more than one Custodian, but if it does so, each individual
must complete and sign a Multi-Signature Addendum Form (Exhibit 294).
When approved, the DUA will have an expiration date. DISPM will alert an organization
with a DUA of its upcoming expiration date and will give the organization the option of
requesting a one-year DUA extension via e-mail, or to close the DUA with a DUA
destruction certificate. DISPM has set up a DUA resource email box which accepts all
expired DUA resolution requests at [email protected].
Custodians may be added or deleted over the life of the primary DUA. To add a new
Custodian under an existing DUA, the P&A must submit the following to CMS/DISPM:
a letter from the P&A describing the activities planned for the new Custodian and the
length of time over which the Custodian will serve, and a Multi-Signature Addendum
signed by the appropriate official from the P&A. The Multi-Signature Addendum must
show the DUA number of the existing primary P&A DUA. The P&A must assign a case
number to all Multi-Signature Addendums beginning with “1” and adding consecutively
thereafter. CMS/DISMP will use this number to track the number of Custodians in each
P&A. When a P&A seeks to delete an existing Custodian, it must send the CMS/DISPM
a letter to this effect. CMS/DISPM will strike out the name of the deleted Custodian
from the DUA or Multi-Signature Addendum that added that Custodian, dating and
initialing the deletion.
The DUA process described in this section applies to disclosure of hospital and CAH
DPU restraint/seclusion death reports by CMS to P&As in those cases where the P&A
did not first make a request specific to an identified patient; a DUA is not required for
other disclosures of information in ACTS to a P&A when permitted in accordance with
the ACTS System of Records Notice.
• A P&A may request information about an on-site survey by submitting its request
to the SA. The SA will process this request and release information to the P&A in
accordance with standard CMS policy for disclosure of Form CMS 2567,
Statement of Deficiencies and Plan of Correction.
If the P&A identifies a particular patient, hospital, and approximate date or dates when
the patient was in that hospital or CAH DPU, and if the P&A makes a request for
additional information, beyond the Form CMS 2567, related to use of restraint or
seclusion on that patient, the request is forwarded to the RO. The RO may, in accordance
with the ACTS System of Records Notice, release additional information to the P&A.
NOTE: Sections 5200 to 5240 relate to all non-deemed
provider/supplier types, excluding nursing homes (SNFs/NFs).
5200 - Investigating Complaints for Non-Deemed Providers/Suppliers,
Excluding Nursing Homes (SNFs/NFs)
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
5200.1 - General Procedures
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
For complaint surveys on non-deemed providers/suppliers, the SA uses the appropriate
survey protocol and interpretive guidelines. A focus survey is conducted on the specific
regulatory requirements related to the allegation. Based on an initial assessment or other
observations, if significant problems are identified, the SA expands the scope of review
as necessary to determine compliance or noncompliance. The SA does not refer
complaints regarding non-deemed providers/suppliers to the RO.
If deficiencies are cited, the SA documents the deficiencies on Form CMS-2567 and
obtains an acceptable POC. If non-compliance with the Medicare conditions is identified,
the SA will follow the appropriate termination procedures and document and report as
required. (See SOM Chapter 3, §§3010-3028 for termination procedures.)
5200.2 - Special Procedures for Psychiatric Hospitals
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The special conditions for psychiatric hospitals cannot be deemed to meet the Medicare
requirements. The remaining conditions for hospitals apply to psychiatric hospitals, and
a hospital may be deemed to meet those conditions.
When the SA receives a complaint allegation against a psychiatric hospital under the
general conditions of participation, it must determine whether or not the hospital is
deemed. If the hospital is deemed, the SA follows the appropriate survey protocol for
deemed facilities. If the hospital is not deemed, the SA investigates the complaint if
appropriate under these procedures listed above in §5200.1.
If the complaint allegation concerns the special conditions for psychiatric hospitals, the
SA may conduct an investigation if it has appropriate qualified personnel or refer it to the
RO. If the complaint is referred to the RO, the RO will evaluate and refer it to the CO as
required.
5210- Processing of Complaints Originating with or Investigated by the
RO

(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The RO establishes procedures and clear organizational accountability to ensure that
complaints are properly evaluated, documented, acknowledged, and handled timely and
appropriately. The RO uses ACTS to ensure timely and appropriate action on all
allegations originating with or investigated by the RO. The extent and nature of the RO
involvement with a given complaint varies depending on the nature of the allegation and
the receiving organization.
Most complaints originate through the SA and are recorded and controlled by the SA.
When a complaint is filed directly with the RO, the RO assumes those initial SA
responsibilities.
5220- Investigation Conducted Directly by the RO
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
This less frequent class of complaints includes allegations retained by the RO or
forwarded to the RO by the SA for investigation. The RO’s responsibilities vary based
on the type of complaint.
The RO uses the appropriate survey protocol and interpretive guidelines for the
provider/supplier. These procedures apply when a direct RO investigation is conducted,
such as for Federal facilities, Religious Non-Medical Health Care Institutions (RNHCIs),
or special situations. When directly investigating, the RO begins by ensuring that it or
the SA has met all initial data collection and acknowledgement requirements.
If the allegation involves an IJ, the RO investigates within two working days. Otherwise,
the RO schedules the investigation based on the severity of the allegation. (See §5075.9
for time frames related to Federal onsite investigations.)
5230 - Special RO Processing
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The following types of allegations are subject to special RO handling:
1. Over-Utilization or Inappropriate Utilization of Services- The RO refers to the
local QIO for investigation, and documents the provider’s files as for other
allegations. The RO acts, as necessary, on any findings returned by the QIO;
2. Civil Rights Violations- The RO refers to the regional OCR for investigation.
The RO documents the provider’s files as for other allegations. The RO acts as
necessary on any findings returned by OCR; and
3. Medicare/Medicaid/CLIA fraud- The RO refers to the RO of the Inspector
General/DHHS for investigation. The RO documents the provider’s files as for
other allegations.

In each of the above instances, the RO ensures that the complainant and SA are notified
of any findings.
5240 - Complaints - HHA Hotline
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Each State has a Medicare home health hotline that can be called by patients who are
dissatisfied with the home health services they are receiving or by other individuals with
a complaint about a specific HHA. Under the Medicare COPs for Patient Rights at 42
CFR 484.10, HHAs are required to provide their patients with the hotline number for
their state. Concerns about an HHA not complying with the COPs, or reports that an
HHA is misinforming beneficiaries or inappropriately terminating care for patients, can
be referred to the SA for investigation via the home health hotline. Concerned consumers
may also call the SA directly. A violation of the COPs or the provider agreement could
lead to termination of the HHA from the Medicare program.
As part of the patient rights COPs, the HHA is required to investigate complaints made
by a patient or the patient’s family or guardian regarding treatment or care that is, or fails
to be, furnished, and to document both the existence of the complaint and resolution of
the complaint.
Surveyors, as part of their investigation of the HHA’s compliance of the COPs, may ask
to review complaints received by the HHA and the resolution of these complaints. The
HHA must permit examination of these records by or on behalf of CMS, or risk
termination from the Medicare program.
NOTE: Sections 5300 to 5390 relate to nursing homes.
5300 - Investigation of Complaints for Nursing Homes
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Section 42 CFR 488.332 provides the Federal regulatory basis for the investigation of
complaints about nursing homes.
The survey agency must review all complaint allegations and conduct a standard or an
abbreviated standard survey to investigate complaints of violations of requirements if its
review of the allegation concludes that:
• A deficiency in one or more of the requirements may have occurred; and
• Only a survey can determine whether a deficiency or deficiencies exist.
The survey agency does not conduct a survey if the complaint raises issues that are
outside the purview of Federal participation requirements.

Complaint investigations follow, as appropriate, the pertinent survey tasks, and
information gathered is recorded on the appropriate survey worksheets. However, if the
documentation required is minimal, use Form CMS-807 to record information during the
complaint investigation. Record deficiencies on Form CMS-2567 and/or, the “Statement
of Isolated Deficiencies Which Cause No Harm with Only a Potential for Minimal Harm
for SNFs and NFs” . The completed Form CMS-2567 must be made a part of the
complaint record.
If necessary, a specialized team may be used to investigate complaints. Team members
may include, but are not limited to, an attorney, auditor, and appropriate health
professionals. The specialized team is not necessarily composed of qualified surveyors.
However, specialized team members provide unique talents and expertise that assist at
least one qualified surveyor in identifying, gathering, and preserving documented
evidence. Further information regarding the composition of the survey team is provided
in Chapter 7.
The timing, scope, duration and conduct of a complaint investigation are at the discretion
of the SA, except when a determination is made that immediate jeopardy may be present
and ongoing or a higher level of actual harm may be present. If the complaint concerns
conditions on a certain day (e.g., on weekends), or on a certain shift (e.g., 11 p.m. - 7
a.m.), the SA should make an attempt to investigate it at the relevant time. In most cases,
the following tasks, or portion of tasks, should be performed during a complaint
investigation.
5300.1 - Task 1: Offsite Survey Preparation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Review any information about the facility that would be helpful to know in planning the
investigation. Contact the ombudsman to discuss the nature of the complaint and whether
there have been any similar complaints reported to and substantiated by the ombudsman.
Review the related regulatory requirements or standards that pertain to the complaint.
For example, if it is a complaint about abuse, review the requirements at 42 CFR 483.13.
Plan the investigation. Before going to the nursing home, plan what information to
obtain during the complaint investigation based on the information already acquired.
Consider practical methods to obtain that information.
5300.2 - Task 2: Entrance Conference/Onsite Preparatory Activities
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Onsite complaint investigations should always be unannounced. Upon entrance, advise
the facility’s Administrator of the general purpose of the visit. The SA explains the
reason for the survey and avoids any impression that a predetermination has been made
as to the validity of the allegation. It is important to let the facility know why you are
there, but protect the confidentiality of those involved in the complaint. Do not release
information that will cause opportunities to be lost for pertinent observations, interviews,
and record reviews required for a thorough investigation. For example, if the complaint
is that food that is intended to be served hot is always served cold, do not tell the facility
the exact complaint. Rather, tell them it is a situation related to dietary requirements.
5300.3 - Task 5: Information Gathering
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The order and manner in which information is gathered depends on the type of complaint
that is being investigated. Conduct comprehensive, focused, and/or closed record
reviews as appropriate for the type of complaint. Generally, it is not necessary to review
records and information from more than one year ago. However, the SA is not precluded
from doing so if concerns identified during the investigation indicate it is necessary in
order to determine current compliance. It is very important to remember that the
determination of whether the complaint happened is not enough. The surveyor needs to
determine noncompliant facility practices related to the complaint situation and which, if
any, requirements are not met by the facility.
Perform information gathering in order of priorities, i.e., obtain the most critical
information first. Based on this critical information about the incident, determine what
other information to obtain in the investigation.
Observations, record review and interviews can be done in any order necessary. As
information is obtained, use what has been learned to determine what needs to be
clarified or verified as the investigation continues.
Observe the physical environment, situations, procedures, patterns of care, delivery of
services to residents, and interactions related to the complaint. Also, if necessary,
observe other residents with the same care need. After determining what occurred, i.e.,
what happened to the resident and the outcome, investigate what facility practice(s) or
procedures affected the occurrence of the incident.
EXAMPLE
It was verified through the investigation that a resident developed a pressure sore/ulcer
which progressed to a Stage IV, became infected and resulted in the resident requiring
hospitalization for aggressive antibiotic therapy. Observe as appropriate: dressing
changes, especially to any other residents with Stage III or IV pressure sores; infection
control techniques such as hand washing, linen handling, and care of residents with
infections; care given to prevent development of pressure sores (e.g., turning and
repositioning, use of specialized bedding when appropriate, treatments done when
ordered, keeping residents dry, and provision of adequate nutritional support for wound
healing).
Record Review: If a specific resident is involved, focus on the condition of the resident
before and after the incident. If there are care issues, determine whether the appropriate
assessments, care planning, implementation of care, and evaluations of the outcome of
care have been done as specified by the regulatory requirements.
EXAMPLE
For a complaint of verbal and physical abuse, review the record to determine the
resident’s mood and demeanor before and after the alleged abuse. Determine if there are
any other reasons for the change in the resident’s demeanor and behavior. Determine
whether an assessment has been done to determine the reason for the change in mood and
behavior. Does the record document any unexplained bruises and/or complaints of pain,
and whether they occurred in relation to the alleged incident?
Interviews: Interview the person who made the complaint. If the complainant is not at
the facility at the time of the survey, he/she should be interviewed by telephone, if
possible. Also, interview the person the complaint is about. Then, interview any other
witnesses or staff involved. In order to maintain the confidentiality of witnesses, change
the order of interviews if necessary. It may not always be desirable to interview the
person who made the complaint first, as that may identify the person as the complainant
to the facility. Interview residents with similar care needs at their convenience.
As interviews proceed, prepare outlines needed for other identified witnesses and revise
outlines as new information is obtained.
During information gathering to investigate a complaint about the care and services
provided to residents in a nursing home, findings of past noncompliance may be
identified. Before considering a citation of past noncompliance with a specific regulatory
tag, surveyors must determine if current compliance with the specific regulatory tag
exists. Similar to verifying correction of current noncompliance on a revisit, surveyors
should use a variety of methods to determine whether correction of the past
noncompliance occurred and continues. This may include, but is not limited to, the
following:
• Interviews with facility staff, such as the administrator, nursing staff, social
services staff, medical director, quality assessment and assurance committee
members, and/or other facility staff, as indicated, to determine what procedures,
systems, structures, and processes have been changed.
• Reviewing through observation, interview and record review, how the facility
identified and implemented interventions to address the noncompliance.
Examples of interventions may include, but are not limited to:
o The facility’s review, revision, or development of policies and/or
procedures to address the areas of concerns;
o The provision and use of new equipment, as necessary;
o The provision of staff training required to assure ongoing compliance for
the implementation and use of new and/or revised policies, procedures,
and/or equipment, especially with new and/or temporary staff;
o The provision of additional staffing, changes in assignments or
deployment of staff, as needed; and
o The provision of a monitoring mechanism to assure that the changes made
are being supervised, evaluated, and reinforced by responsible facility
staff.
• Evaluating whether the facility has a functioning quality assessment and
assurance committee whose responsibilities include the identification of quality
issues; providing timely response to ascertain the cause; implementing corrective
action; implementing monitoring mechanisms in place to assure continued
correction and revision of approaches, as necessary, to eliminate the potential risk
of occurrence to other residents and to assure continued compliance.
A citation of past noncompliance must meet all of the criteria described in Task 6 below.
5300.4 - Task 6: Information Analysis
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Review all information collected. If there are inconsistencies, do additional data
collection as needed, to resolve the inconsistencies. Determine if there is any other
information still needed.
Determine whether:
• The allegations are substantiated or unsubstantiated;
• The facility failed to meet any of the regulatory requirements; and
• The facility practice or procedure that contributed to the complaint has been
changed to achieve and/or maintain compliance.
To cite past noncompliance with a specific survey data tag (F-tag or K-tag), all of the
following three criteria must be met:
1) The facility was not in compliance with the specific regulatory requirement(s) (as
referenced by the specific F-tag or K-tag) at the time the situation occurred;
2) The noncompliance occurred after the exit date of the last standard recertification
survey and before the survey (standard, complaint, or revisit) currently being
conducted; and

3) There is sufficient evidence that the facility corrected the noncompliance and is
in substantial compliance at the time of the current survey for the specific
regulatory requirement(s), as referenced by the specific F-tag or K-tag.
A nursing home does not provide a plan of correction for a deficiency cited as past
noncompliance as the deficiency is already corrected; however, the survey team
documents the facility’s corrective actions on Form CMS-2567.
5300.5 - Task 7: Exit Conference
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
Conduct an Exit Conference related to a complaint survey in accordance with the process
described in the Exit Conference section located in the Long-Term Care Survey Process
(LTCSP) Procedure Guide (https://www.cms.gov/Medicare/Provider-Enrollment-and-
Certification/GuidanceforLawsAndRegulations/Nursing-Homes.html). Do not inform the
nursing facility of confidential information unless the individual who provided the
information specifically authorizes you to do so.
If a deficiency is not present now, but was present and has been corrected, notify the
facility orally and in writing that the complaint was substantiated because deficiencies
existed at the time that the complaint situation occurred. (See SOM Chapter 7, Survey
and Enforcement Process for Skilled Nursing Facilities and Nursing Facilities, for
specific information about citing past noncompliance.)
If the complaint is unsubstantiated, (i.e., the surveyor(s) cannot determine that it occurred
and there is no indication of deficient practice), notify the facility of this decision.
5310 - Action on Allegations of Resident Neglect and Abuse, and
Misappropriation of Resident Property for Nursing Homes
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
5310.1 - Written Procedures
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
The State must develop and implement written procedures for the timely review and
investigation of allegations of resident abuse and neglect, and misappropriation of
resident property, including both complaints and facility-reported incidents. The State’s
policies and procedures must be consistent with Federal requirements as well as with
procedures in the State Operations Manual.
Nursing homes send the following types of incidents to the State Survey Agency:
• All alleged violations involving abuse, neglect, exploitation or mistreatment,
including injuries of unknown source and misappropriation of resident property;

• The results of all facility investigations involving alleged violations of abuse,
neglect, exploitation or mistreatment, including injuries of unknown source and
misappropriation of resident property; and
• Reasonable suspicions of crimes against nursing home residents.
NOTE: If the SA receives information that a suspected crime may have
occurred in a facility and there is indication that it has not been reported or
the SA cannot verify that a report was made to law enforcement, then the SA
forwards the information from the initial report immediately to law
enforcement. The SA must follow applicable laws and regulations related to
information disclosures, privacy and confidentiality, as it makes referrals.
The SA may also contact the CMS Location office for more information.
A. Initial Reporting of Facility-Reported Incidents
The information collected during intake is critical in determining what may be
occurring in a facility and the effect(s) that it may have on residents. While
States have discretion in how they collect information from facilities (e.g.,
through electronic submission), at a minimum, the State Survey Agency must
provide instructions to the facility and collect sufficient information to
determine how the incident should be prioritized. See also Exhibit XX for
sample instructions with examples of information and Appendix PP, Tag
F609. If the facility has not provided sufficient information, the SA should take
this into consideration as it triages the incident.
1. Facility Reported Incidents – Initial Report
The facility must provide in its report sufficient information to describe the
alleged violation and indicate how residents are being protected [See
§483.12(c)(3)]. It is important that the facility provide as much
information as possible, to the best of its knowledge at the time of
submission of the report, so that state agencies can initiate action
necessary to oversee the protection of nursing home residents. See Exhibit
XX for a sample form for initial reporting with examples of information
and see also Appendix PP, Tag F609.
B. Reporting of Investigation Findings for Facility-Reported
Incidents
For alleged violations of abuse, neglect, exploitation or mistreatment,
including injuries of unknown source and misappropriation of resident
property, the facility is required to submit a report of the results of the
investigation within 5 working days to the State Survey Agency (See 42 C.F.R.
§483.12(c)(4), Tag F609 of Appendix PP of the State Operations Manual).

While States have discretion in how they collect information from facilities
(e.g., through electronic submission), at a minimum, the State Survey Agency
must provide instructions to the facility and collect sufficient information to
determine how the incident should be prioritized.
5-Day Final Report of Suspected Allegation
Within 5 working days of the incident, the facility must provide in its report
sufficient information to describe the results of the investigation, and indicate
any corrective actions taken, if the allegation was verified. It is important that
the facility provide as much information as possible, to the best of its
knowledge at the time of submission of the report, so that State agencies can
initiate action necessary to oversee the protection of nursing home residents
[see §483.12(c)(4)]. See Exhibit XX for a sample form for the investigation
report with examples of information, and see also Appendix PP, Tag F609.
5310.2 - Review and Triage of Allegations
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
The State reviews all allegations of resident neglect and abuse and misappropriation of
resident property regardless of the source.
5310.2A-Immediate Jeopardy Priority
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
In cases where the initial report indicates the following, the SA must initiate an onsite
survey within three business days of receipt of the initial report:
1) The alleged noncompliance may have caused, or may likely cause, serious injury,
harm, impairment, or death to a resident, and
2) The facility has not implemented adequate protection for all residents or the SA
has not received sufficient evidence to conclude that residents are adequately
protected.
For these cases, the SA will enter into ACTS: Intake Type=Incident; Priority = IJ; and
Investigate Within X Days = 3 Working Days.
In cases where the initial report indicates the following, the SA must initiate an onsite
survey within seven business days of receipt of the initial report:
1) The alleged noncompliance may have caused, or may likely cause, serious injury,
harm, impairment, or death to a resident, and
2) The facility has potentially implemented adequate protection for all residents.
For these cases, the SA will enter into ACTS: Intake Type=Incident; Priority = IJ; and
Investigate Within X Days = 7 Working Days.
NOTE: See Appendix Q of the State Operations Manual for guidance related to
immediate jeopardy situations.
Investigating nursing home facility-reported incidents according to these timeframes will
be implemented no later than October 1, 2023, to provide time for the SAs to prepare
given the resource constraints in the SAs.
Depending on the nature of the allegation, the facility would be expected to take
immediate action(s) to ensure the protection of residents. Information provided by the
facility may assist the SAs in determining whether there are potentially adequate
protections provided to the resident. Examples of such information include, but are not
limited to:
• Monitoring of the alleged victim and other identified residents who are at risk,
such as conducting unannounced management visits at different times and shifts;
• Evaluation of whether the alleged victim feels safe and if he/she does not feel
safe, taking immediate steps to alleviate the fear, such as a room relocation,
increased supervision, etc.;
• Providing social services (e.g., emotional support and counseling) to the
resident, as needed;
• Immediate assessment of the alleged victim and provision of medical treatment
as necessary;
• Provision of goods and/or services that are necessary to avoid serious injury,
harm, impairment, or death to a resident;
• Immediate notification of the alleged victim’s physician and the resident
representative, when there is injury or a change in condition or status;
• If the alleged perpetrator is staff- Removal of access by the alleged perpetrator
to the alleged victim and other residents and assurance that ongoing safety and
protection is provided for the alleged victim and other residents
• If the alleged perpetrator is a resident or visitor- Removal of access by the
alleged perpetrator to the alleged victim and, as appropriate, other residents and
assurance that ongoing safety and protection is provided for the alleged victim
and other residents
• Notification of the alleged violation to other agencies or law enforcement
authorities, within timeframes as specified under Federal or State law or
regulations; and
• Whether administrative staff, including the administrator, were informed and
involved as necessary in the investigation.
Below are examples that indicate that a resident(s) may not be protected in the facility:
• The alleged perpetrator continues to have access to the alleged victim and/or
other residents;
• Retaliation occurs against a resident who reports an alleged violation;
• A resident who repeatedly fondles other residents is moved to another unit,
where he/she continues to exhibit the same behaviors to other residents; and
• A resident with a history of striking a resident is left unsupervised with a resident
who has been targeted in the past.
The SA may contact the resident/representative to determine whether adequate
protections are provided to the resident
5310.3 - Investigating Allegations
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If there is reason to believe, either through oral or written evidence, that an individual
used by a facility to provide services to residents could have abused or neglected a
resident or misappropriated a resident's property, the State must investigate the allegation.
During the investigation, the SA should evaluate how the facility developed policies and
procedures to prevent the abuse, and after the abuse occurred, how the facility took action
to report and investigate the allegations while ensuring the safety of the residents.
5310.4 - Factors Beyond the Control of the Individual
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The State must not make a finding that an individual neglected a resident if the individual
demonstrates that such neglect was caused by factors beyond the control of the
individual.
EXAMPLE: A nurse aide could not be found negligent for not providing clean bed and
bath linens to a resident if the facility had no clean bed and bath linens available.
However, the facility is responsible for providing clean bed and bath linens to residents.
5320 – Reporting Findings of Abuse, Neglect, or Misappropriation of
Property to the Nurse Aide Registry
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
5320.1 - Notification Procedures - Preliminary Determinations
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If the State makes a preliminary determination, based on oral or written evidence and its
investigation, that resident neglect, abuse, or misappropriation of property has occurred,
the State completes the following notification procedures:
1. Individuals Notified - The State notifies the following individuals in writing
within 10 working days of the investigation:
a. Individual(s) implicated in the investigation; and
b. The current administrator of the facility in which the incident occurred.
2. Notice Information - The following information is included in the notice:
a. Nature of the allegation (specific facts);
b. Date and time of the occurrence;
c. A statement that the individual implicated in the investigation has a right to a
hearing and must request the hearing within 30 days from the date of the
notice. Provide the individual with the specific information needed to request
a hearing, such as the name and address of a contact in the State to request a
hearing;
d. Statement that if the individual fails to request a hearing, in writing, within 30
days from the date of the notice, the presumed substantiated findings is
reported to the nurse aide registry or the appropriate licensure authority;
e. The intent to report findings substantiated by a hearing in writing to the nurse
aide registry and/or to the appropriate licensure authority;
f. Consequences of waiving the right to a hearing;
g. Consequences of a finding through the hearing process that the resident abuse
or neglect, or misappropriation of property did occur; and
h. Right of the accused individual to be represented by an attorney at the
individual’s own expense.
5320.2 - Conduct of Hearing for Nurse Aides
1- Time frame to Complete the Hearing

The State must complete the hearing and the hearing record within 120 days from the day
it receives the request for a hearing.
2 - Hearing Location
The State must hold the hearing in a manner consistent with State practice at a reasonable
place and time convenient for the individual.
5320.3 - Reporting Findings
1 - Reporting to Entities
If the individual waives the right to a hearing or the time to request a hearing has expired,
or if the hearing finding is that the individual neglected or abused a resident or
misappropriated a resident’s property, the substantiated findings must be reported in
writing within 10 working days to:
a. The individual;
b. Current administrator of the facility in which the incident occurred;
c. The administrator of the facility that currently employs the individual, if it is not
the same facility in which the incident occurred;
d. Applicable licensing authorities; and
The nurse aide registry for nurse aides as specified in 42 CFR 483.156(c) and discussed
in §4141. Section 4141 discusses the function of the registry, the information contained
in the registry, and responsibility for the registry.
2 - Information Submitted to the Nurse Aide Registry
The following information must be included and remain in the registry permanently,
unless the finding was made in error, the individual was found not guilty in a court of
law, or the State is notified of the individual’s death. See §4141.B.
a. Documentation of the investigation, including the nature of the allegation and the
evidence that led to the conclusion that the allegation was valid;
b. The date of the hearing, if the individual chose to have one, and its outcome; and
c. A statement by the individual disputing the allegation if the individual chose to
make one.
3 - Information Retained in the Nurse Aide Registry Permanently

The registry must remove entries for individuals who have performed no nursing or
nursing-related services for 24 consecutive months, unless the individual’s registry entry
includes documented findings of abuse, neglect, or misappropriation of property.
5330 - Reporting Abuse to Law Enforcement and the Medicaid Fraud
Control Unit for Nursing Homes
(Rev. 212; Issued; 02-10-23; Effective: 10-21-22; Implementation: 10-24-22)
If the SA receives information that a suspected crime may have occurred in a facility and
there is indication that it has not been reported or the SA cannot verify that a report was
made to law enforcement, the SA must report the suspected crime to law enforcement
immediately.
Verifying that a complainant, facility, and/or covered individual(s) has made a report to
law enforcement would include review and confirmation of the following information:
• Who submitted the report to law enforcement, including name and contact
information;
• Who did the reporter contact, including law enforcement entity, name, and
contact information;
• Date/Time that the report was filed;
• Any copies of the report made to law enforcement, if available;
• What information was conveyed to law enforcement; and
• The police report number provided by law enforcement.
When the SA or RO substantiates a finding of abuse, the SA or RO must report the
substantiated findings to local law enforcement and, if appropriate, the Medicaid Fraud
Control Unit.
NOTE: “Covered individual” is defined in section 1150B(a)(3) of the Act as anyone who
is an owner, operator, employee, manager, agent or contractor of the facility
(§483.12(b)(5)(i)).
5340 - Post-Survey Certification Actions for Nursing Homes
(Rev. 155, Issued: 06-10-16, Effective: 06-10-16, Implementation: 06-10-16)
Following the investigation, the survey team records any findings on Form CMS-2567,
the SA conducts a supervisory review of the CMS-2567 form and sends the provider a
copy. The SA requests a POC for any uncorrected deficiencies. See §2728.
When Federal deficiencies are identified, the SA initiates certification actions as follows:
1. Noncompliance that Constitutes Immediate Jeopardy to Resident Health
and Safety - The SA initiates procedures in accordance with §§7307 to 7309.
2. Noncompliance that Does Not Constitute Immediate Jeopardy to Resident
Health and Safety - The SA initiates procedures in accordance with §§7311 to
7316.
3. In Substantial Compliance - The SA initiates procedures in accordance with
§7319.
5350 – Data Entry
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The SA enters survey information into the ASPEN system (via ASPEN Central Office or
ACTS), including Forms CMS-670 and CMS-2567.
5360 - Processing of Complaints Originating with or Investigated by the
CMS RO
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The RO establishes procedures and clear organizational accountability to ensure that any
complaint is properly evaluated, documented, acknowledged, and handled timely and
appropriately. The RO uses ACTS to ensure timely and appropriated action on all
allegations originating with or investigated by the RO.
The extent and nature of RO involvement with a given complaint varies depending on the
nature of the allegation and the receiving organization. The following procedures address
the major variants of RO involvement.
5370 - Pre-Investigation Actions on Allegations Originating Through
the RO
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Most complaints originate through the SA and are recorded and controlled by the SA.
When a complaint is filed directly with the RO, however, the RO assumes those initial
SA responsibilities.
5380 - RO Processing of RO Investigated Complaints
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
This less frequent class of complaints includes allegations retained by the RO or
forwarded to the RO by the SA for investigation or special processing. The RO
responsibilities vary based on the type of complaint.

1 - Direct RO Investigation
These procedures apply when a direct RO investigation is conducted. When directly
investigating, the RO begins by ensuring that it or the SA has met all intake,
acknowledgment, and priority assignment requirements in §5010 to §5020.
2 - Conducting the Investigation
The RO follows the procedures for investigation in §5300.
3 – RO Certification Actions
When Federal deficiencies are identified, the RO initiates certification actions as follows:
a. Noncompliance that Constitutes Immediate Jeopardy to Resident Health and
Safety - The RO initiates procedures in accordance with §§7307 to 7309. The RO
performs the SA responsibilities described in these sections.
b. Noncompliance that Does Not Constitute Immediate Jeopardy to Resident Health
and Safety - The RO initiates procedures in accordance with §§7311 to 7316.
c. In Substantial Compliance - The RO initiates procedures in accordance with
§7319.
4 - Reporting
The RO should report survey information into the ASPEN system (via ASPEN Central
Office or ACTS), including Forms CMS-670 and CMS-2567.
5390 – RO Oversight of Complaint-Related Processes
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
1. The RO considers any complaint data in targeting look-behind surveys or reviews.
2. The RO monitors data in summary form - either through a log or data system. See
§5060.
These records should include:
• Identification of region or State-wide patterns;
• Pinpointing of problem providers or States;
• Evaluation of SA processing times, workloads, performance, etc.; and

• Identification of overall SA workloads, including unsubstantiated and Medicaid-
only complaint volumes.
3. Based on needs identified from oversight activities, the RO provides SA training and
technical assistance.
NOTE: Sections 5400 to 5480.2 relate to alleged EMTALA violations.
5400 - Investigations Involving Alleged Emergency Medical Treatment
and Labor Act (EMTALA) Violations
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Section 1866 of the Act, Agreements with Providers of Services, specifies that for a
hospital, or any provider of services, to qualify for participation in the Medicare program,
it must enter into an agreement with the Secretary of HHS. Effective August 1, 1986,
participating hospitals with emergency departments must comply with the requirements
of §1867 of the Act as a condition of their provider agreement.
The following Medicare provider agreement requirements, which closely parallel
provisions contained in §1866of the Act, must be met by Medicare participating hospitals
with emergency departments:
• 42 CFR 489.20(l) requires a hospital to comply with the requirements of 42 CFR
489.24. Section 1866(a)(1)(I) of the Act requires a hospital to have and enforce
policies to ensure compliance with the requirements of §1867;
• 42 CFR 489.20(m) requires a hospital to report to CMS or the SA any time it has
reason to believe it may have received an individual who has been transferred in
an unstable emergency medical condition (EMC) from another hospital in
violation of the requirements of 42 CFR 489.24(e);
• 42 CFR 489.20(q) requires a hospital to post conspicuously a sign(s) specifying
the rights of individuals, under §1867 of the Act, with respect to examination and
treatment for emergency medical conditions and women in labor and to indicate
whether or not the hospital participates in the Medicaid program. The letters
within the signs must be clearly readable at a distance of at least 20 feet or the
expected vantage point of the emergency department clients. The wording of the
sign(s) must be clear and in simple terms and language(s) that are understandable
by the population served by the hospital;
• 42 CFR 489.20(r)(1) requires a hospital to maintain medical and other records
related to individuals transferred, including discharges, to or from the hospital for
a period of five years from the date of transfer;

• 42 CFR 489.20(r)(2) requires a hospital to maintain a list of physicians who are
on call to provide treatment necessary to stabilize an individual with an
emergency medical condition;
• 42 CFR 489.20(r)(3) requires a hospital to maintain a central log on each
individual who comes seeking assistance and whether he or she refused treatment,
was refused treatment, or whether the individual was transferred, admitted and
treated, stabilized and transferred, or discharged.
When hospitals do not conform to the requirements of §1867 of the Act, the practice is
commonly called “dumping.” A hospital with a dedicated emergency department is
defined in 42 CFR 489.24(b) as a hospital that offers services for emergency medical
conditions within its capacity to do so. The regulations at 42 CFR 489.24 parallel the
provisions of §1867 of the Act and contain the following requirements that a hospital
with a dedicated emergency department must meet:
• 42 CFR §489.24(a) General. Applicability of provisions of this section.
• 42 CFR §489.24(b) Definitions. As used in this section.
• 42 CFR §489.24(c) Use of dedicated emergency department for non-emergency
services.
• 42 CFR §489.24(d) Necessary stabilizing treatment for emergency medical
conditions.
• 42 CFR §489.24(d)(1) General. Subject to the provisions of paragraph (d)(2).
• 42 CFR §489.24(d)(2) Exception: Application to inpatients.
• 42 CFR §489.24(d)(3) Refusal to consent to treatment.
• 42 CFR §489.24(d)(4) Delay in examination or treatment.
• 42 CFR §489.24(d)(5) Refusal to consent to transfer.
• 42 CFR §489.24(e) Restricting transfer until the individual is stabilized.
• 42 CFR §489.24(e)(1) General.
• 42 CFR §489.24(e)(2) Appropriate transfer to another medical facility.
• 42 CFR §489.24(e)(3) Provides whistleblower protection to physicians and
qualified medical personnel.

• 42 CFR §489.24(f) Recipient hospital responsibilities.
• 42 CFR §489.24(g) Termination of provider agreement.
• 42 CFR §489.24(h) Consultation with Quality Improvement Organization (QIO).
• 42 CFR §489.24(i) Release of QIO Assessment.
• 42 CFR §489.24(j) Availability of on-call physicians.
• 42 CFR §489.24 (j)(1) On-call list.
• 42 CFR §489.24 (j)(2) Hospital on-call policy and procedures.
If a hospital fails to meet these requirements, CMS may terminate the provider agreement
in accordance with 42 CFR 489.53. The Office of the Inspector General (OIG) has the
responsibility and authority to assess civil monetary penalties (CMPs) or to exclude
physicians from the Medicare program when a hospital or physician violates these
requirements. Additionally, individuals suffering personal harm and medical facilities
suffering financial loss as a result of a violation of these provisions can bring civil action
against the offending hospital and physicians. Filing for such civil action is limited to a
period of 2 years after the date of the alleged violation. This legislation does not preempt
any State or local laws, except to the extent that State or local requirements directly
conflict with a requirement of this legislation.
5410 – EMTALA and Born-Alive Infants Protection Act of 2002
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
5410.1 - Interaction of the Born-Alive Infant Protection Act and
EMTALA
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
With the definition of the terms “person” and “individual” codified at 1 U.S.C. §8, it is
clear that there are circumstances where EMTALA protections are applicable to an infant
who is born alive, as that term is defined in 1 U.S.C. §8(b).
For example, assume that a hospital’s labor and delivery department meets the definition
of a “dedicated emergency department.” If an infant was born alive in that dedicated
emergency department, and a request was made on that infant’s behalf for screening for a
medical condition, (or if a prudent layperson would conclude, based on the infant’s
appearance or behavior, that the infant needed examination or treatment for an
emergency medical condition and that a request would have been made for screening) the
hospital and physician could be liable for violating EMTALA for failure to provide such
a screening examination. The born-alive infant is a “person” and an “individual” under 1
U.S.C. § 8(a) and the screening requirement of EMTALA applies to “any individual”
who comes to the emergency department.
Another example is a case of an infant born alive elsewhere on the hospital’s campus
(i.e., not in the hospital’s dedicated emergency department) and a prudent layperson
observer concluded, based on the born-alive infant’s appearance or behavior, that the
infant was suffering from an emergency medical condition. In such a circumstance, the
hospital and its medical staff are required to perform a medical screening examination on
that infant to determine whether or not an emergency medical condition existed. If the
hospital or its medical staff determined that the infant was suffering from an emergency
medical condition, the hospital has an obligation to admit the infant, or to comply with
either the stabilization requirement or the transfer requirement of EMTALA. The born-
alive infant is a “person” and an “individual,” as described above, and the stabilization
and transfer requirements of EMTALA apply to “any individual” who comes to the
hospital.
Finally, a third example is when the hospital admits a born-alive infant. EMTALA does
not apply to inpatients. If a born–alive infant is admitted to the hospital, EMTALA would
not apply to protect the infant in most circumstances. However, the Medicare COPs
would apply to the infant once he or she was admitted to the hospital as an inpatient.
5410.2 - Conduct of Investigations
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If a complaint indicates that a born-alive infant has been denied a screening examination,
stabilizing treatment, or an appropriate transfer, the complaint is prioritized as an alleged
EMTALA violation. It is not necessary to determine that the hospital acted with an
improper motive in any failure to provide a screening examination, stabilizing treatment,
or an appropriate transfer in order to conclude that an EMTALA violation has occurred.
The Supreme Court of the United States has held that a finding of improper motive is not
required to conclude that an EMTALA violation has occurred.
5420 - Basis for Investigation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The SA enters alleged EMTALA violations into ACTS. The RO approves or disapproves
requests for EMTALA investigations in ACTS.
5430 - RO Direction of Investigation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
5430.1 - Evaluation of Allegation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The RO evaluates all complaints and refers to the SA those that warrant SA investigation.
The SA or the RO sends a letter to the complainant acknowledging the complaint and
informing the complainant of whether an investigation is warranted. The SA’s

responsibility is to verify whether a violation of 42 CFR 489.24 and/or the related
requirements at 42 CFR 489.20 occurred, and if there were other violations.
5430.2 - Request for Investigation of Allegations
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The RO gives an initial verbal authorization to the SA to investigate the EMTALA
allegation, and then completes Form CMS-1541A in ACTS. If the RO identifies
Medicare conditions or standards it wants the SA to survey, related to the EMTALA
allegation at a deemed hospital, the RO completes Form CMS-2802 in ACTS. If the RO
identifies conditions or standards it wants the SA to survey related to the EMTALA
allegation at a non-deemed hospital, it directs the SA to conduct a survey by completing
Form CMS-1541A in ACTS.
5440 - Conducting an Investigation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
5440.1 - Selecting the Team
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The SA selects surveyors with a background in the profession or area to be investigated.
Preferably, the surveyors should have acute care training and experience. All surveyors
must be adequately trained in the evaluation of 42 CFR 489.24 cases. Physicians should
have experience in peer review.
5440.2 - Scheduling the Investigation
(Rev. 191, Issued: 07-19-19, Effective: 07-19-19, Implementation: 07-19-19)
Allegations of EMTALA violation against a non-deemed or deemed hospital or CAH
may represent a probable immediate jeopardy to the next individual who comes to the
hospital requesting examination and treatment for an emergency medical condition.
Therefore, when triaged as IJ by the RO, initiate the investigation within two business
days after receipt of the authorization from the RO. The onsite investigation must be
conducted on consecutive business days. The survey must be completed promptly and is
not to be interrupted by other activities. DO NOT ANNOUNCE ANY
INVESTIGATIONS.
Based on review of the complaint allegations by the RO, the EMTALA complaint may
also be prioritized as Non-IJ High. In these situations, the investigation must be initiated
within 45 business days of RO authorization. The onsite investigation must be conducted
on consecutive business days. The survey must be completed promptly, should not be
interrupted by other activities, and must be unannounced.
5440.3 - Guidelines for Surveyors Conducting Investigations
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)

Attention to Procedures
The purpose of conducting the investigation is to ascertain whether or not the hospital
violated the EMTALA requirements. The survey must be in accordance with applicable
survey procedures and policies. Review instructions in Appendix V, before beginning
the investigation. The guidelines provide a detailed interpretation of the regulations.
Involvement of Complainants
Complainants, if known, receive a letter of acknowledgment from the SA or RO. Do not
disclose the identity of complainants. When information obtained during the
investigation appears to be in conflict with the information supplied by the complainant,
consult with the complainant, if this can be done without disclosing the person’s identity.
5440.4 - Conducting the Investigation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
A complete investigation consists of assessment of the following components:
• Completeness, adequacy and enforcement of policies and procedures which
address the provisions of 42 CFR 489.24;
• Prompt reports to the SA or CMS of receipt of an improperly transferred
individual by the receiving hospital;
• Presence and completeness of signs posted in emergency departments specifying
the rights of individuals under 42 CFR 489.24, and information indicating
whether the hospital participates in the Medicaid program;
• Maintenance of medical and other records related to individuals transferred to or
from the hospital for a period of five years from the date of transfer, including
discharged patients;
• Maintenance of a list of physicians who are on call to provide necessary
stabilizing treatment;
• Maintenance of a central log on each individual who comes to the hospital
seeking emergency services;
• Provision of an appropriate medical screening examination sufficient to determine
the presence of an emergency medical condition;
• Provision of necessary stabilizing treatment;
• Provision of no delay in examination or treatment in order to inquire about
insurance status or capability for payment;

• Provision of an appropriate transfer to another medical facility/provider;
• Provision of whistleblower protections; and
• Adequacy of responsibilities of the recipient hospital with specialized capabilities
(nondiscrimination).
The survey tasks are listed below for easy reference. See Appendix V for detailed
guidance.
• Task 1: Entrance Conference;
• Task 2: Case Selection Methodology;
• Task 3: Record review;
• Task 4: Interviews;
• Task 5: Exit Conference;
• Task 6: Professional Medical Review; and
• Task 7: Assessment of Compliance and Completion of the Deficiency Report.
After the investigation is concluded, complete a Form CMS-1541B (Exhibit 137). If one
or more of the provisions of EMTALA are not met, complete Form CMS-2567, using
“Principles of Documentation.” Describe in detail the facts of each individual case. In
addition, specify whether the hospital was aware of the problem and took steps to remedy
it prior to the survey. If a SA physician was a member of the investigation team, include
the medical review of the case. Use the “Physician Review Outline for Emergency Care
Obligations of Medicare Hospitals,” (Exhibit 138) for this purpose. In addition, complete
Form CMS-562. All the forms must be signed, showing the professional titles of all
participating surveyors, and dated.
A hospital may have multiple sites listed under its Medicare provider number. These sites
may not be in close proximity of each other and each site may have its own dedicated
emergency department (DED). In cases where the alleged EMTALA violation is against
a specific site of the hospital, the surveyors should focus their survey investigation at the
hospital site mentioned in the complaint intake. However, the surveyors should review
all EMTALA related Policies and Procedures of all sites of the hospital. The surveyors
need to survey the other sites of the hospital if the survey findings indicate that the
potential EMTALA violation maybe widespread.
5440.5 - Exit Conference
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)

It is usually desirable and appropriate to conduct an exit conference. The surveyor(s) may
outline the basic facts uncovered during the onsite investigation. However, the
surveyor(s) must inform the hospital that the RO makes the final compliance
determination, and the determination is often made with information obtained after the
onsite investigation. Do not reveal the complainant and do not venture an opinion on
what determination the RO might make. The exit conference should include a
description of the process that is followed if the RO determines that a violation has
occurred.
5450 - Forwarding Report of Investigation to the RO
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Transmit the results of the investigation and your recommendations to the RO through
ACTS within 10 working days following completion of the onsite survey, if it appears
there may be an EMTALA violation. If there appears to be no violation, this time frame
may be extended to 15 working days, in order to allow the SA additional processing time.
Transmit the following materials to the RO through ACTS:
• Form CMS-562,”Medicare/Medicaid/CLIA Complaint Form;”
• Form CMS-1541B, “Responsibilities of Medicare Participating Hospitals in
Emergency Cases Investigation Report.” Recommend one or more of the actions
below on the form:
o None - This means the complaint was not substantiated;
o In Compliance, but Previously Out of Compliance - This means that the
hospital identified the problem on its own and took effective corrective
action prior to the investigation. In addition to this recommendation,
document on the Form CMS-2567 when the hospital identified the
violation or a similar problem, the corrective action taken, and the date of
such action. Also, document that the hospital has had no violations or
similar problems for at least the past 6 months;
o Recommend Termination (23 calendar day track) - This means that the
hospital is out of compliance with 42 CFR 489.24 or the related
requirements at 42 CFR 489.20(l), (m), (q) or (r) and the violation presents
an immediate jeopardy to patient health and safety;
o Recommend Termination (90 calendar day track) - This means that the
hospital is out of compliance with 42 CFR 489.24 or the related
requirements at 42 CFR 489.20(l), (m), (q) or (r), but the violation does
not present an immediate jeopardy to patient health and safety;
o Request Physician Review. This means that it is recommended that the
RO obtain a medical review of the case;
o Possible Discrimination. This means that it is believed that
discrimination occurred based on financial status, race, color, nationality,
handicap, or diagnosis.
• Form CMS-670, “Survey Team Composition and Workload Report;”
• Form CMS-2567, “Statement of Deficiencies and POC;”
NOTE: If the hospital had identified the deficiency and took corrective action
prior to the investigation, indicate on the Form CMS-2567 that the
requirement was not met. However, indicate on the Form CMS-2567
and the narrative report that the hospital took corrective action prior to
the investigation, what action was taken, and for how long the hospital
has been in compliance.
• Physician Review Outline for Emergency Care Obligations of Medicare Hospital
(if physician review was done by SA);
• Complaint investigation narrative;
• Copies of pertinent hospital policies and procedures that relate to the identified
deficiencies;
• Summary listing of all patients comprising the sample, including an explanation
of how and why the cases were selected for review;
• Summary of interviews.
Transmit the following to the RO by overnight mail:
• Copies of medical records for substantiated cases, medical records of individuals
named in the complaints, and other medical records for which a QIO review is
requested;
• Certification of benefits versus risks of the transfer, if this is a transfer case.
5460 - RO Review of Investigation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Upon receiving the case from the SA, the RO has 10 working days to review the
investigation findings. The RO requests a 5-day advisory medical review of the case by
the QIO to determine if there is an EMTALA violation. The RO has 5 working days to
review the case upon return from the QIO. With this information, and any other

additional information, the RO determines whether the hospital complied with the
EMTALA requirements and determines whether the violation constitutes an immediate
jeopardy to patient health and safety.
Prior to determining compliance or noncompliance, the RO is encouraged to confer with
the State Agency, and may confer with the hospital’s representatives. The RO shares as
much data as possible in accordance with current Privacy Act requirements.
5460.1 - Hospital Is In Compliance - No Past Violation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If the RO determines that the allegation is not substantiated and that the hospital is in
compliance with 42 CFR 489.24 and/or the related requirements at 42 CFR 489.20, the
RO notifies the hospital and forwards a copy of the letter to the SA. If the SA received
the complaint, it notifies the complainant that the complaint was not substantiated. If the
RO received the complaint, the RO notifies the complainant.
5460.2 - Hospital Is In Compliance - Past Violation, No Termination
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If the RO determines that the allegation was substantiated, but the hospital had identified
the violation on its own, took effective corrective action prior to the investigation, and
has had no EMTALA violations for at least the past 6 months, termination action is not
initiated. The RO notifies the hospital via a “Past Violation - No Termination Letter.”
The SA receives a copy of the letter through ACTS. The RO or SA sends a letter to the
complainant regarding the outcome of the investigation. Although no termination action
is taken, the RO refers past violations of 42 CFR 489.24 to the OIG for assessment of
civil monetary penalties (CMPs) if warranted.
5460.3 - Hospital Is Not in Compliance - Immediate Jeopardy to Patient
Health and Safety
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If the RO determines that the hospital is not in compliance and the violation represents an
immediate jeopardy to patient health and safety, the RO follows a 23 calendar-day
termination process. The termination procedures in §3010 are followed. Uncorrected
deficiencies that resulted in a violation of 42 CFR 489.24 may pose an immediate
jeopardy to people seeking emergency care. The RO notifies the complainant that the
complaint was substantiated. It also informs the hospital in writing of the specific
violations via a preliminary determination letter, and sends the hospital a copy of Form
CMS-2567. The SA receives a copy of the letter through ACTS.
5460.4 - Hospital Is Not in Compliance - Situation Does Not Pose an
Immediate Jeopardy to Patient Health and Safety
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)

If the RO determines that the hospital is not in compliance with the EMTALA
requirements, but the violation does not pose an immediate jeopardy to patient’s health
and safety, or the hospital took corrective action after the investigation to remove the
immediate jeopardy, the RO follows a 90 calendar-day termination process. The
termination procedures in §3012 are followed. The RO notifies the complainant that the
complaint was substantiated. The RO informs the hospital, in writing, of the specific
violations via a preliminary determination letter and sends the hospital a copy of Form
CMS-2567. The SA receives a copy of the letter through ACTS.
Examples of noncompliance that usually do not pose an immediate jeopardy:
1. A transfer which was appropriate, but not signed or dated by the physicians;
2. An appropriate, functioning, central log that on one particular day is not fully
completed; and
3. A written hospital policy that is missing, but is nonetheless being implemented.
The fact that the hospital has completed a POC should not be interpreted to mean that the
hospital admits violating the EMTALA requirements. However, the hospital is included
on the log of facilities with EMTALA violations, with the notation that an acceptable
POC was received by CMS, and termination action was stopped.
5465 - Procedures for the 5-day QIO Review of Alleged Violations of 42
CFR 489.24
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Prior to terminating a hospital from the Medicare program because of possible
violation(s) of EMTALA, the RO requests the QIO to assess whether the individual
involved was provided an appropriate medical screening examination, stabilizing
treatment, or an appropriate transfer as required by EMTALA.
The QIO 5-day review is mandatory if the RO determines that a case involves a possible
violation of 42 CFR 489.24 to support possible termination action against a hospital if in
fact it violated EMTALA. The RO is responsible for providing the QIO with all
information relevant to the case that is within its possession and control. The RO sends
the “Physician Review Outline for Emergency Care Obligations of Medicare Hospitals,”
(Exhibit 138) to capture this information. The QIO completes the review within 5
working days upon the receipt of RO’s request. The QIO sends the case file back to the
RO including a copy of the review report. It is not required that the physician reviewer
give the hospital and/or the physician an opportunity to respond to the allegations at this
time. If the affected physician and/or hospitals have questions concerning the case, they
are to consult with the RO.

The QIO Review is not required in cases where a delay in effecting a sanction would
jeopardize the health and safety of individuals or in situations where medical review is
inappropriate (e.g., cases where the individual was denied a medical screening
examination). The QIO 5-day review is required to seek medical expertise on whether
the individual was adequately screened, examined and treated.
The RO shall release upon request the 5 day QIO review to the affected physician and/or
hospital, after the RO has made a determination as to whether the hospital violated or is
in compliance with EMTALA. In addition, the RO may release the QIO review to the
complainant or his/her representative upon request. The physician reviewer’s identity is
confidential, therefore, when releasing the QIO report the physician’s identity is not to be
disclosed unless he or she consents to the release of their identity in accordance with the
disclosure regulations at 42 CFR 480.132 and 480.133.
The cases in which the RO determined that the hospital was in compliance with 42 CFR
489.24 but in violation of 42 CFR 489.20 of the EMTALA regulation do not need to be
forwarded to the QIO for review. The RO takes action as warranted.
5470 - Termination Procedures for EMTALA Violations
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
5470.1 - Procedures for Termination when the EMTALA Violation is an
Immediate Jeopardy to Patient Health and Safety
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
In cases where the RO determined that an immediate jeopardy existed, after a 5-day QIO
advisory review has been completed, the RO follows the termination procedures in
§3010. The processing timeframes are the maximum allowed. The termination
procedures are not postponed or stopped unless evidence of correction of the deficiencies
or proof that the violation did not exist is provided by the hospital to the RO. The RO
forwards the supporting documents to the QIO (for a 60 day QIO review) in order to
provide a medical opinion on the case. The RO refers the case to the OIG that has the
responsibility for assessment of CMPs against the hospital and/or physician and
physician exclusion provisions for violations of 42 CFR 489.24. The case is also referred
to the Office for Civil Rights (OCR) because OCR may take action under the Hill-Burton
Subpart G Community Services regulations at 42 CFR 124.603(b) (1).
The termination track starts on the date that the RO makes the determination of
noncompliance with 42 CFR 489.24. It is the date of the preliminary determination
letter. The letter is forwarded to the hospital by the fastest method available (fax, e-mail
or telephone). In addition, a written letter follows up by mail. The preliminary
determination letter informs the hospital of:
• The RO’s findings based on the investigation and the results of medical review;

• The projected termination date (the 23rd calendar day from the date of the
preliminary determination letter);
• The date on which the RO issues a Notice of Termination Letter and notifies the
public (at least two calendar days, but no more than four calendar days prior to
the termination date); and
• That the hospital may avoid the termination action and notice to the public by
either providing acceptable POCs for the deficiencies or by successfully showing
that the deficiencies did not exist. In either case, the necessary information must
be furnished to the CMS RO in time for the SA to verify the corrections before
the projected termination date.
If, during the resurvey, the SA finds that the provider had implemented systems and
processes to ensure that the likelihood of further violation is remote and there is adequate
evidence that the provider is in compliance with the requirements, the termination action
is rescinded and the provider is put back in compliance.
If, during the resurvey, the SA finds that the provider has not adequately implemented
systems and processes to ensure compliance, the RO gives the hospital an additional 67
days or a total of 90 days (23 plus 67) to achieve compliance.
This allows the hospital time to prove that the corrective action is good for the long-term
(i.e., the corrective action is adequate to ensure that no further violations will occur). The
RO directs the SA to conduct a second survey by the 60th calendar day. On the resurvey,
the surveyor(s) reviews patients’ emergency department (ED) records and other relevant
documents for the period since the last survey to assess continued compliance. If the
hospital fails to achieve compliance, it is terminated from the Medicare program. The
RO sends the complainant a letter reporting the final results of the investigation.
If the termination takes place and the hospital desires to become re-certified as a
Medicare provider, the hospital must provide reasonable assurance that compliance will
be maintained. The procedures at §2016 are followed.
5470.2 - Procedures for Termination When the EMTALA Violation is
Not Immediate Jeopardy to Patient Health and Safety
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
In cases where the RO determined that a violation existed but not an immediate jeopardy,
after a 5-day QIO advisory review has been completed when it was warranted, the RO
follows the termination procedures in §3012 The processing timeframes are the
maximum allowed. The termination procedures are not postponed or stopped unless
evidence of correction of the deficiencies or proof that the violation did not exist is
provided to the RO by the hospital. If warranted, the RO forwards supporting documents
to the QIO (for a 60 day QIO review) in order to provide a medical opinion on the case.
The RO refers the case to the OIG, if warranted, that has the responsibility for assessment

of CMPs against the hospital and/or physician and physician exclusion provisions for
violations of 42 CFR 489.24 The case is also referred to the Office for Civil Rights
(OCR) because OCR may take action under the Hill-Burton Subpart G Community
Services regulations at 42 CFR 124.603(b)(1)
The termination track starts on the date that the RO makes the determination of
noncompliance with 42 CFR 489.24 and/or the related requirements at 42 CFR 489.20. It
is the date of the preliminary determination letter.
5480 - Procedures for QIO Review of Confirmed EMTALA Violation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Before imposing sanctions under §1867 of the Act for violations of 42 CFR 489.24, 42
CFR 489.24(h) requires that CMS obtain consultation from the QIO. The OIG holds the
authority to assess CMPs against the hospital or physicians or to exclude physicians from
the Medicare program for violations of 42 CFR 489.24.
5480.1 - Procedures for Coordinating 60 day QIO Review
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The RO requests the QIO to provide a medical opinion on EMTALA violation cases
within 60 calendar days. The cases referred for 60-day QIO review are outlined in
§5480B. The RO uses the “Model Letter Requesting QIO Review of a Confirmed
Violation of 42 CFR 489.24 for Purposes of Assessing Civil Monetary Penalties or
Excluding Physicians,” (Exhibit 212). The QIO provides the physician and the hospital
reasonable notice of its review a reasonable opportunity for discussion, and an
opportunity for the physician and hospital to submit additional information before issuing
its report. (Instructions on notice of review and opportunity for discussion, and
additional information that follow the regulatory requirements in 42 CFR 489.24(h) are
found in §§9100-9150 of the QIO Manual.)
The RO is responsible for providing the QIO with all information relevant to the case that
is within its possession and control. The RO sends the “Physician Review Outline for
Emergency Care Obligations of Medicare Hospitals,” (Exhibit 138) to capture this
information. This outline is helpful for organizing the review of the medical record. The
specialty of the reviewing physician should be matched to the specialty of the physician
who attended the patient and/or the individual’s medical condition. If the patient was not
seen by a physician, the QIO uses the diagnosis of the patient or the usual physician
assignment practice of the hospital to determine the specialty of the physician reviewer.
Within 60 calendar days of receiving the case, the QIO must submit to the RO a report on
its findings. The report provides an expert medical opinion regarding whether the
individual involved had an emergency medical condition, whether the individual’s
emergency medical condition was stabilized, whether the individual was transferred
appropriately, and whether there are any medical utilization or quality of care issues

involved in the case. Upon request, the RO provides copies of the QIO report to the
affected physician and/or hospital after all investigative activity has been completed.
When there was no screening examination or when a delay would jeopardize the health or
safety of individuals, QIO Review is not required before the OIG may impose CMPs or
exclude a physician from the Medicare program. In addition, if the QIO determines, after
a preliminary review, that there was an appropriate medical screening examination and
the individual did not have an emergency medical condition, the QIO returns the case to
the RO with its documented opinion. The RO will close the case and no referral to OIG is
necessary.
When the RO determines that a hospital was non-compliant with the requirements of 42
CFR 489.24, one of its notice requirements is to notify the OIG that the violation was
confirmed and that termination action was initiated. (See Exhibit 208.) The RO
completes the notification after receipt of the QIO 60-day review report. If the QIO
report does not support an EMTALA violation, the RO closes the case without referring
it to the OIG.
The RO forwards the following documents to the OIG:
• Form CMS-1541B;
• Form CMS-2567;
• Medical record;
• Summary of interviews;
• Explanation of sample selection;
• Copies of pertinent hospital policies and procedures related to the identified
deficiencies;
• Complaint investigation narrative;
• Certification of benefits versus risks of the transfer (if this is a transfer case);
• Copy of the 5 working-day advisory QIO Review, and
• Copy of the 60 calendar-day advisory QIO Review.
The RO sends the above information and any other pertinent documentation in its
possession to the OIG at the following address:
Office of Inspector General
Office of Counsel to the Inspector General
Department of Health and Human Services
Room 5527, Cohen Building
330 Independence Avenue SW
Washington, D.C. 20201
5480.2 - EMTALA Case Referral to OIG
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
CMS refers appropriate cases to the OIG for investigation. Periodically, OIG will advise
us of the criteria they would prefer CMS to use in referring cases. Examples of the types
of cases that may be referred include:
1. Financial Screening - The hospital and/or responsible physician refused to
examine or treat a person based on the person’s insurance status or inability to pay
a fee. The financial basis for the decision must be clearly supported by evidence
in the file, e.g., documented policy, interview reports.
2. Patient with Trauma or Acute Emergency Condition - The hospital and/or
responsible physician (including an on-call physician who failed to come to the
hospital) failed to screen, stabilize, or appropriately transfer (or, in the case of a
hospital with specialized capabilities or facilities, refused to accept an appropriate
transfer of) a person with trauma, e.g., a severe head injury, or other acute
emergency condition, e.g., heart attack or stroke, requiring immediate and
substantial medical intervention.
3. High Risk Event (such as Birth) Occurs Prior to Arrival at Another Hospital
- The hospital and/or responsible physician discharged or refused to screen/treat a
person who gave birth (or is subject to another high risk medical event) prior to
arriving at another hospital (especially if transport is by private vehicle).
4. Death or Serious Harm Results from Dump - The evidence in the file
(including the QIO Review) demonstrates that the dumping violation caused
serious medical harm or death to the victim of the violation.
5. Egregious Violation Prioritized by CMS - CMS concludes that a CMP is
appropriate because of the seriousness of the violation (the person must have had
an emergency medical condition) and other relevant factors, e.g., long history of
noncompliance, hospital policy resulting in violations, pattern of serious
violations, knowing and willful violation. This category is for those cases that
CMS determines are very serious and merit a CMP but do not fit within other
categories identified by OIG.
5480.3 - Releasing QIO Assessment
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)

Upon request, the RO may release QIO assessment(s) to the physician and/or hospital or
the affected individual, or his/her representative. The QIO physician’s identity is
confidential unless he/she consents to its release. The QIO Review may be released
pursuant to the requirements of 42 CFR 480.132 and 480.133.
Sections 5500 to 5590 relate CLIA.
5500 - Complaints Involving Unaccredited Laboratories
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
NOTE: This section applies to complaints against laboratories that hold a CLIA
certificate of compliance, certificate of waiver (COW), and certificate of PPM
(See §§5540-5590 for complaints regarding accredited laboratories).
A complaint is an allegation that could result in citing noncompliance with CLIA
requirements. A complaint may be substantiated or unsubstantiated as a result of an
investigation or survey. A substantiated complaint is one resulting in a finding of
noncompliance at the time of the investigation, or a finding that noncompliance was
proven to exist, but was corrected prior to the investigation. An unsubstantiated
complaint is an allegation where sufficient evidence could not be found to conclude that
noncompliance with CLIA requirements existed during the investigation or at the time of
the alleged violation. A complaint may be received in either the SA or the RO. The
receiving organization should follow the procedures outlined below.
The SA obtains the following information for every complaint:
• Complainant’s name, address, and telephone number, unless the complainant
requests anonymity;
• Laboratory’s name and address; and
• Description of problem, (e.g., personnel, places, and dates of occurrence).
5500.1 - Control
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The SA establishes a file for the complaint and logs the action in a control system. The
system may be manual or automated, but must facilitate tracking and control of the
complaint.
5500.2 - Acknowledgment
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If the complainant is known, the SA promptly issues written acknowledgment that the
complaint is being investigated. The SA should not delay acknowledgment pending an
investigation unless the investigation takes place within three working days. The SA
must take appropriate precautions to protect the complainant’s anonymity and privacy.
The SA maintains a copy or record of the notification with the complaint documentation.
5500.3 - Evaluation
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The SA evaluates any complaint to determine whether it should be investigated by the
SA, or whether it should be forwarded to the RO for investigation or referral to the
appropriate authority (e.g., OCR, OSHA, RO). The SA assesses the complaint to
determine if an immediate survey is necessary. While the SA will perform most
complaint surveys, complaints involving State-operated facilities are the responsibility of
the RO. When the SA does not have jurisdiction, it should forward the complaint to the
RO within three working days. If referral is not necessary, the SA considers whether or
not any special notification is appropriate.
If a complaint is especially significant, sensitive, or attracting broad public or media
attention, the SA informs the RO immediately.
5500.4 - Scheduling Investigations
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The SA investigates within two working days of receiving the complaint and focuses on
the specific problem area if the complaint involves possible immediate jeopardy to
patient health and safety. Otherwise, the SA follows procedures for prioritizing and
investigating certification-related complaints. Laboratories with complaints pending are
identified and given priority in scheduling of regular certification surveys.
5500.5 - Conducting Investigations
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The SA investigates complaints by means of an onsite survey, by telephone, by electronic
communication, by letter, or by a documentary review. Complaint investigations are
unannounced.
For onsite complaint investigations, the SA performs a full or partial survey based on the
allegations. If a complaint alleges generalized inappropriate laboratory practices, the SA
evaluates compliance with applicable requirements or conducts a full survey, as needed.
If the complaint is of a specific nature, the SA performs a survey focused on areas
relevant to the complaint.
5500.6 - Conducting Investigations in a Laboratory with a Certificate of
Waiver
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The RO authorizes an unannounced complaint survey of a laboratory holding a certificate
of waiver only if it is based on a substantial allegation of noncompliance. The fact that a

deficiency is not at the Condition-level does not preclude taking adverse action based on
provisions contained in 42 CFR 493.1840. As with other laboratories, the SA
investigates complaints made against laboratories with a certificate of waiver by means of
an onsite survey, by telephone, letter, or by a review of documents.
The SA performs the onsite investigation based on the allegation and determines whether
a laboratory is performing only waived tests and if the laboratory is following the
manufacturer’s instructions for performing the tests (See Appendix C).
5500.7 - Conducting Investigations in a Laboratory with a Certificate
for PPM Procedures
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The RO authorizes an unannounced complaint survey of a laboratory holding a certificate
for PPM procedures only if based on a substantial allegation of noncompliance. This
survey should not differ from a complaint survey done in any other laboratory performing
non-waived testing, as all requirements for moderate complexity apply except routine
survey.
Substantial indication that a laboratory is performing tests that do not appear on the PPM
procedures test list; e.g., through billing procedures, should prompt a complaint survey of
a certificate for PPM procedures laboratory followed by either proper registration or
appropriate sanctions.
5500.8 - Post Investigation Actions
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Following the investigation, the SA records any deficiencies on a Form CMS-2567 and
provides it to the facility using regular procedures. Subsequent actions depend on the
severity and nature of the deficiencies cited and the facility’s willingness or ability to
correct them.
When deficiencies are identified, the SA initiates actions as follows:
1. Condition-Level Deficiencies - Immediate Jeopardy - Certifies noncompliance
and initiates procedures to recommend imposing alternative and principal
sanctions.
2. Condition-Level Deficiencies - No Immediate Jeopardy; Facility Provides an
Acceptable POC - Certifies noncompliance and initiates procedures to
recommend imposing alternative sanctions based on the severity and nature of the
deficiencies found.
3. Lower Level Deficiencies - Facility Provides an Acceptable POC - Certifies
compliance based upon an acceptable POC and assembles documentation for RO
review.
4. Lower Level Deficiencies - Facility Unable or Unwilling to Provide
Acceptable POC - A facility with deficiencies may not participate without an
acceptable POC. The SA recommends sanction action to the RO.
When no deficiencies are identified, no certification action is required.
5500.9 - Resolution/Closeout
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
1 - Unsubstantiated
The SA enters the unsubstantiated complaint into ACTS and documents the facility’s
certification file.
2 - Substantiated
The SA reports substantiated complaints using the Form CMS-2567 and any appropriate
supporting documentation. The SA logs summary information in the control system and
files a copy of the complaint documents in the facility’s certification file. The SA enters
complaints into ACTS. The laboratory will be charged a fee to cover the cost of the
survey if noncompliance is documented.
The SA closes out all complaints with a follow-up notice to the complainant with the
findings and disposition of the complaint. The SA should send this notice soon after the
investigation and retains a copy with the complaint record.
The SA provides follow-up reports, as necessary, to any other appropriate parties such as
the State Medicaid agency and/or initial referring agencies. The SA must be sure to
protect the anonymity and privacy of the complainant.
The SA inputs the investigation information into ACTS within 45 days of the completion
of the complaint survey.
5510 - CLIA-Exempt Laboratory Complaint Investigations - General
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
Complaints may be from any source, including verbal, written, electronic or in the media.
Complaints are investigated if they meet the following criteria:
• If substantiated, would have an impact on the health and safety of the general
public or individuals served by the laboratory, and
• Would raise doubt as to the laboratory's compliance with one or more CLIA
Conditions and/or requirements.
An attempt to maintain the anonymity of the complainant should always be made.
There are a number of entities that must address laboratory complaints including: CMS
Central and Regional Offices, the state agencies (including those with state licensure
programs), approved states, and accreditation organizations. Each of these entities shares
a strong interest in ensuring the quality of patient care and the services provided by
laboratories. When the complaint involves more than one of these entities, there should
be coordination and communication to ensure an effective and timely resolution of the
issue.
5520 - Review of CLIA-Exempt Laboratory Complaints
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If the RO receives a complaint against a CLIA-exempt laboratory, the RO determines
what action is appropriate. The RO may do any of the following:
• Determine the severity of the complaint;
• Send the information to the approved State for their action;
• Conduct a survey (full or partial);
• Investigate the complaint during the course of a validation survey (full survey), if
it is conducted within 45 days of the laboratory’s licensure survey and the
complaint does not present immediate jeopardy concerns.
• If the seriousness of the complaint or the circumstances warrant, the RO should
invoke the Rapid Response Alert Protocol.
NOTE: Transfusion-related fatality investigations must be conducted by the RO. They
may not be delegated to the approved State; however, the approved State may
accompany the RO on the investigation. In either case, there must be
coordination and communication between the RO and the State. Where State
laws apply to transfusion-related incidents, the approved State program should
follow its established procedures and coordinate with the RO.
The RO Reviews the approved State program’s complaint activities as part of the overall
annual review. The RO has the discretion to maintain its own complaint tracking system
for those that have been forwarded to the approved State program. However, this
information should be an integral part of the State’s annual review.
If the approved State program receives a complaint against a CLIA-exempt laboratory,
the approved State program determines what action is appropriate. If the approved State
sanctions a CLIA-exempt laboratory in any way (e.g., licensure is withdrawn), it must
notify the RO within 30 days.
If the laboratory against which the complaint is alleged is accredited, the State must also
notify the accreditation organization.
5530 - Conducting Complaint Investigations and Surveys for CLIA-
Exempt Laboratories
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The RO will complete the “Medicare/Medicaid/CLIA Complaint Form,” Form CMS-562,
for every complaint investigation it performs in a CLIA-exempt laboratory. When an
investigation can be conducted via telephone (e.g., personnel credentials), the RO should
do so. The RO obtains the following information for every allegation:
• Complainant’s name and address, unless complainant requests anonymity. Do not
disclose the identity of the complainant to the laboratory;
• Laboratory’s name and address; and
• Description of problem, involving names, places, and dates.
The RO follows the same procedures for control and acknowledgement indicated in
§5500. Complaints involving potential immediate jeopardy will be investigated by the
RO within 2 working days of receipt. Complaints not involving potential immediate
jeopardy are investigated within 45 days. All complaint surveys are unannounced.
If a laboratory representative refuses to permit a complaint survey, the RO contacts the
State and requests that it contact the laboratory to explain the protocol and, if necessary,
suggest that the State take enforcement action against the CLIA-exempt laboratory. The
RO conducts the complaint survey in accordance with the survey protocol and uses the
appropriate survey forms specified in Exhibit 63 and the outcome-oriented protocol
found in Appendix C.
Initially, the RO focuses the survey only on the Condition(s) or requirement(s) related to
the complaint area(s). If the complaint is substantiated or if additional deficiencies are
found during the course of the investigation, the RO expands the scope of the survey to
include additional standards, conditions, and other CLIA requirements. If the complaint
is not substantiated, the RO notifies the laboratory that it is in compliance with the CLIA
Condition(s) (Exhibit 243). The RO also notifies the approved State program of the
Condition-level compliance (Exhibit 244).
At the exit conference, the RO informs the laboratory of the deficiencies found. If the
deficiencies pose immediate jeopardy to the health and safety of individuals served by a
laboratory or that of the general public, the RO notifies the approved State program and
the laboratory within two working days by overnight mail and includes a copy of the
Form CMS-2567. The RO directs the State program to take the appropriate enforcement
action. (See Exhibits 231 and 228). The RO follows-up with the State program within
15 working days of its notification to the laboratory to verify that the enforcement action
has either been taken against the laboratory or that the laboratory has achieved
compliance with CLIA requirements.
If the State program fails to take appropriate enforcement action for an immediate
jeopardy case within 23 days of the RO’s notification, and the laboratory has not
achieved Condition-level compliance, the RO may request CO to either contact the State
or attempt other resolution to eliminate the jeopardy.
If the deficiencies do not pose immediate jeopardy to the health and safety of individuals
served by a laboratory or that of the general public, the RO prepares a Form CMS-2567
and forwards a letter along with the Form CMS-2567 to the laboratory and to the State
program within 10 working days of completing the survey. The State program is
responsible for taking any enforcement action, if necessary, monitoring the correction of
the deficiencies, and providing a report to the RO. (See Exhibit 231.)
The RO completes a Survey Team Composition and Workload Report, Form CMS-670,
for all complaint surveys and related activity.
If the approved State program fails to take appropriate enforcement action in non-
immediate jeopardy situations, the RO documents its files accordingly and notifies CO.
Failure to take and document the necessary enforcement action may subsequently
jeopardize current or future approval of the State's laboratory licensure program.
5540 - Complaint Investigations and Surveys of Accredited
Laboratories Under CLIA
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
There are a number of entities that must address laboratory complaints including: CMS
Central and Regional Offices, the state agencies (including those with state licensure
programs), approved states, and accreditation organizations. Each of these entities shares
a strong interest in ensuring the quality of patient care and the services provided by
laboratories. When the complaint involves more than one of these entities, there should
be coordination and communication to ensure an effective and timely resolution of the
issue.
The statutory basis for conducting surveys of accredited laboratories based on allegations
of noncompliance is found in §353(e)(2)(D) of the Public Health Service Act (PHSA).
Since accreditation organization (AO) requirements are equivalent to CLIA requirements,
a complaint may affect the laboratory’s accreditation status as well.
Complaints are investigated if they meet the following criteria:
• If substantiated, would have an impact on the health and safety of the general
public or individuals served by the laboratory, and

• Would raise doubt as to the laboratory's compliance with one or more CLIA
Conditions and/or requirements.
The RO should evaluate the complaint and take appropriate investigatory action. If the
seriousness of the complaint or the circumstances warrant, the RO should invoke the
Rapid Response Alert Protocol. Every effort should be made to secure a written form of
the complaint, while maintaining anonymity, if requested.
All complaint surveys are unannounced and conducted according to outcome-oriented
survey principles (See Appendix C). If an investigation can be conducted by letter or
telephone, in lieu of an onsite survey, those means should be utilized.
Upon receipt, all complaints are logged and tracked and the same information as for CMS
certified laboratories is collected, monitored and maintained (see Section 5500).
5550 - RO Direction of Complaint Investigation of an Accredited
Laboratory
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
The RO has primary responsibility for the coordination of all activities involving
complaints relating to an accredited laboratory.
This includes:
• Ensuring that all pertinent information concerning the complaint is obtained;
• Assessing the level of severity of the complaint;
• Determining actions required for investigation;
• Determining whether multiple AOs may be involved; and
• When warranted (e.g., in cases potentially involving media coverage,
Federal/State Congressional or political interest, legal intervention, etc.),
informing and coordination with all affected parties, including AO's, State
Agencies and Central Office.
Although the RO has the lead role in directing the investigation of complaints involving
accredited laboratories, all affected entities (i.e., State Agencies, AO's, Central Office)
share responsibility in ensuring timely and effective action is taken.
Complaints received by the SA:
If the SA receives a substantial allegation of noncompliance directly from a complainant
about an accredited laboratory, it promptly acknowledges receipt of the complaint and
advises the complainant that it is being forwarded to the RO for action. The SA forwards
a copy of the acknowledgment letter and the complaint to the RO. This includes SAs
with a State laboratory licensure program.
Complaints received by the RO:
If the complaint is received directly by the RO, the RO will promptly send a letter to the
complainant acknowledging the complaint and advising the complainant of the intended
course of action, and subsequently the results of any investigation, if appropriate, and of
the corrective action taken.
In either case (complaint received by SA or RO), the RO evaluates the complaint and has
the lead in determining the course of action. The RO determines whether the RO, the
SA, or the AO, including multiple AOs if circumstances so warrant, will investigate the
complaint. The RO will also determine whether one or multiple AOs may be impacted
by the complaint and, if so, alert them of the pending action. In certain instances the RO
may enlist CO support to help determine the most effective course of action.
If the RO determines that the SA should investigate the complaint, the RO prepares a
“Request for Complaint Investigation or Validation Survey of Accredited Laboratory,
Form CMS-2802A,” (See Exhibit 107) and a “Medicare/Medicaid/CLIA Complaint,”
Form CMS-562, and forwards them to the SA along with a copy of the complaint and
notifies the AO. If the RO authorizes the SA to perform a full survey (all specialties and
subspecialties covered by the certificate), and the survey can be performed within 90 days
of the AO’s inspection, the survey can be counted in the SA’s validation workload.
If the RO determines that the complaint involves a potential immediate jeopardy to the
individuals served by the laboratory, or to the general public, the SA investigates the
complaint within two working days of receiving it from the RO. Otherwise, the RO will
direct the SA to investigate non-Immediate Jeopardy complaints within 45 days and
report their findings to the RO and AO at the conclusion of the survey.
Complaints investigated by AOs:
If the RO determines that the accreditation organization should carry out its own
investigation, it promptly forwards the complaint to the accreditation organization for
immediate attention. The RO will request to be notified of the results of any investigative
action taken. The RO will then notify the SA and, if warranted, CO.
NOTE: Transfusion-related fatality investigations must be conducted by the RO or
SA. Transfusion-related fatality investigations must not be referred to an
accreditation organization for action. However, the AO or multiple AOs,
as appropriate, should be notified when such an investigation is taking
place.
Complaints received by AOs:

Complaints received directly by AOs will be investigated under each AOs own standards
and procedures. If multiple AOs are potentially impacted, the AO receiving the complaint
will promptly inform the other AOs and a determination should be reached regarding the
need for coordinated action. In cases potentially involving media coverage, Federal/State
Congressional or political interest, legal intervention, etc., the SA, RO and CO should be
promptly alerted by the AO receiving the complaint and consulted concerning
appropriate action.
5560 - Conducting Complaint Survey of an Accredited Laboratory
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If an onsite survey is warranted, the SA will conduct an unannounced survey of an
accredited laboratory based on the substantial allegation of noncompliance. The SA
conducts the complaint survey in accordance with outcome-oriented principles (see
Appendix C). The SA conducts a focused complaint survey, as instructed by the RO on
Form CMS-2802A. If the SA finds additional deficiencies during the course of the
complaint investigation, it may expand the scope of the survey with RO approval.
At the exit conference, the SA informs the laboratory director of the deficiencies found
and the procedures to respond to them. If the deficiencies do not pose an immediate
jeopardy to the health and safety of individuals served by a laboratory, or to the general
public, the SA prepares a Form CMS-2567 and requests that the laboratory submit a POC
for all Condition-level deficiencies. Condition level deficiencies must be corrected;
those at the standard level are optional. The SA informs the laboratory that the Form
CMS-2567 will be made available to the public under the disclosure of survey
information provisions. The SA indicates to the laboratory that the “Statement of
Deficiencies” (Form CMS-2567) will be forwarded to the laboratory within 10 working
days and that the POC must be returned to the SA within 10 calendar days. Upon receipt
of the survey information and POC, the RO makes a determination of whether or not
sanctions will be imposed against the laboratory and notifies the AO.
5570 – Forwarding Investigation Report to RO
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If non-immediate jeopardy is found, the SA will submit the appropriate information as
specified in the List of Documents in the Certification Packet (see Exhibit 63) to the RO,
or through an update to ACTS within 45 days of completing the survey and notifies the
RO of the entry. If the laboratory chooses not to submit a POC when deficiencies are
found, the SA reports any known information about the laboratory’s efforts to correct
deficiencies to the RO and AO.
5580 - Accredited Laboratory Found in Compliance Following a
Complaint Survey
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)

If after review of the documentation the RO determines that the accredited laboratory is
in compliance with all CLIA Condition-level requirements, it officially notifies the
laboratory and forwards a copy of this letter to the SA and the AO. This letter advises
that the accreditation organization may contact the laboratory about correcting any
deficiencies below Condition-level.
5590 - Accredited Laboratory Found Not in Condition-level Compliance
Following a Complaint Survey
(Rev. 18, Issued: 03-17-06; Effective/Implementation Dates: 03-17-06)
If the deficiencies found pose an immediate jeopardy to the health and safety of
individuals, the SA prepares the Form CMS-2567, (which is included as part of the List
of Documents in the Certification Package, See Exhibit 63) and notifies the RO and sends
Form CMS-2576 to the laboratory within 2 working days. RO will notify the AO. Based
on the information forwarded, and the laboratory’s POC, the RO determines if sanctions
are to be imposed against the laboratory. The RO will then notify the AO.
Should the immediate jeopardy situation be corrected before the adverse action is taken
or completed, the SA will advise the laboratory that it will revisit it to inspect all
remaining Conditions not in compliance. The RO will notify the AO.
If non-immediate jeopardy is found, the SA will submit the appropriate information as
specified in the List of Documents in the Certification Packet (See Exhibit 63) to the RO,
or through an update to ACTS within 45 days of completing the survey and notifies the
RO of the entry. The POC should also be forwarded to the RO. If the laboratory chooses
not to submit a POC when deficiencies are found, the SA reports any known information
about the laboratory’s efforts to correct deficiencies to the RO and the RO will notify the
AO and the laboratory that the laboratory is out of compliance and has been placed under
SA monitoring jurisdiction (see Exhibit 241). The laboratory is monitored by the SA, RO,
and/or AO until it reaches Condition-level compliance or its certificate of accreditation is
revoked. A copy of all correspondence is provided to the accreditation organization by
the RO.
For standard only deficiencies, responsibility rests with the AO to follow-up and pursue
corrective action. The laboratory continues to be accredited by its accreditation
organization and retains its CLIA certificate of accreditation during this monitoring
period; however, it becomes subject to the same CLIA requirements, survey and
enforcement procedures as applied to non-accredited laboratories found out of
compliance.
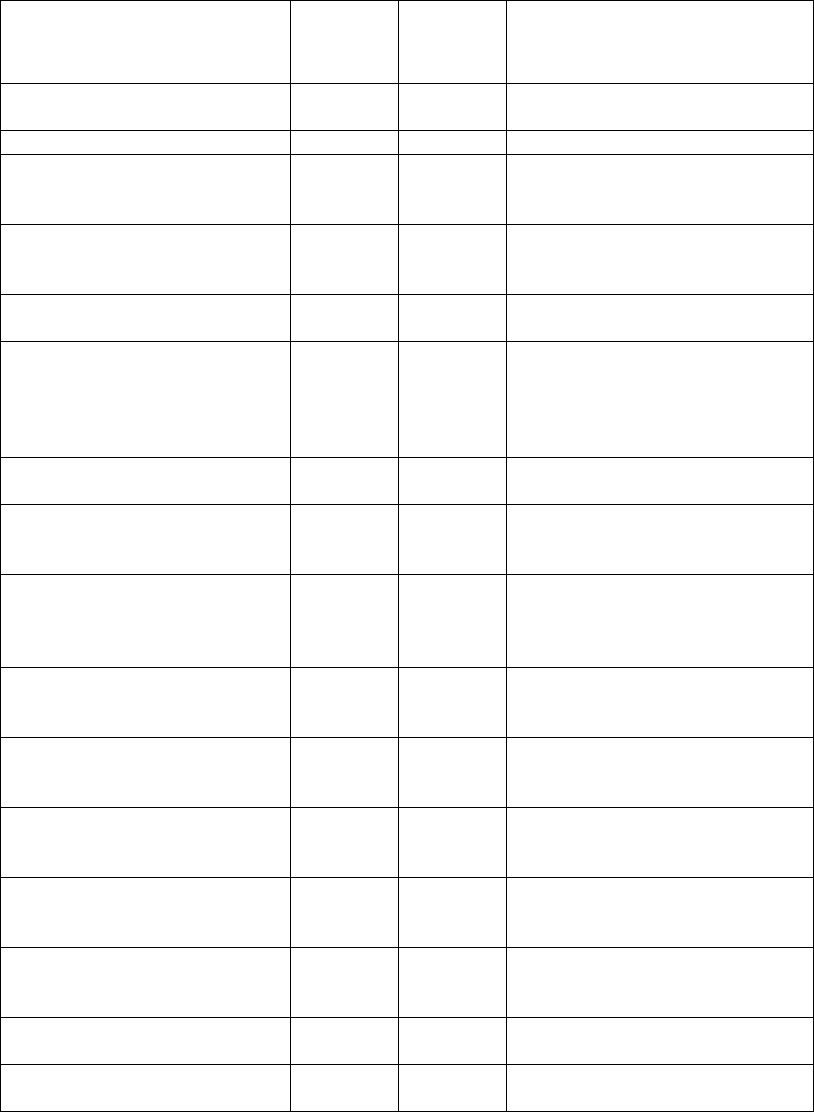
CROSSWALK TO THE OLD CHAPTER 5
A crosswalk from sections of the State Operations Manual Chapter Five published 5-
21-2004 to the revised chapter five is as follows:
Name of Old Section
Old
Section
Number
New
Section
Number
Name of New Section
Management of Complaints and
Incidents
5000
5000
Management of Complaints and
Incidents
Intake Process
5010
5010
Intake Process
Triage and Priority Assignment
5020
5070
Priority Assessment for Nursing
Homes, Deemed and Non-Deemed
Providers/Suppliers, and EMTALA
Priority Definitions
5030
5075
Priority Definitions for Nursing
Homes, Deemed and Non-Deemed
Providers/Suppliers, and EMTALA
Investigation Findings and
Reports
5040
5080
Investigation Findings and Reports
CMS Regional Office
Responsibility for Monitoring
SA Management of Complaints
and Incidents
5050
5050
CMS Regional Office
Responsibility for Monitoring SA
Management of Complaints and
Incidents
Aspen Complaints/Incidents
Tracking System (ACTS)
5060
5060
ASPEN Complaints/Incidents
Tracking System (ACTS)
Investigation of Complaints
Against Accredited/Deemed
Providers and Suppliers
5100
5100
Investigation of Complaints for
Deemed Providers/Suppliers
Basis for Investigation of
Complaints Against
Accredited/Deemed Providers
and Suppliers
5110
5100.1
Basis for Investigation
RO Direction of Accredited
Hospital Complaint Investigation
5120
5210
Processing of Complaints
Originating with or Investigated by
RO
Conducting an Accredited
Hospital Complaint Validation
Survey
5130
5050
CMS RO Responsibility for
Monitoring SA Management of
Complaints and Incidents
Forwarding Investigation Report
to RO
5140
5050
CMS RO Responsibility for
Monitoring SA Management of
Complaints and Incidents
Accredited Hospital Found in
Compliance Following
Complaint Validation Survey
5150
5100
Investigation of Complaints for
Deemed Providers/Suppliers
Accredited Hospital Found Not
in Compliance Following
Complaint Validation Survey
5160
5100
Investigation of Complaints for
Deemed Providers/Suppliers
Reinstatement to Accreditation
Organization Jurisdiction
5170
5100.2
Post-Survey Procedures
Termination of Accredited
Hospital
5180
5100.2
Post-Survey Procedures

Investigating Complaints
Involving ESRD Services
Provided by Accredited
Hospitals
5190
5160
Investigating Complaints Involving
ESRD Services Provided by
Deemed Hospitals
Investigation of Complaints
Against Other Than
Accredited/Deemed Providers
and Suppliers
5200
5200
Investigating Complaints for Non-
Deemed Providers/Suppliers,
Excluding Nursing Homes
SA Processing of General,
Certification-Related Complaints
5210
5010
General Intake Process
Hospital Restraints/Seclusion
Death Reporting and
Investigation
5240
5140
Hospital Restraints/Seclusion
Death Reporting and Investigation
Complaints Involving HIV-
Infected Individuals
5250
5150
Complaints Involving HIV-
Infected Individuals
Investigations Involving Alleged
EMTALA Violations
5300-
5400
5400-
5500
Investigations Involving Alleged
EMTALA Violations
Complaints Involving
Unaccredited Laboratories
5500-
5590
5500-
5590
Complaints Involving
Unaccredited Laboratories
Chapter Five State Operations Manual
Acronyms
(Rev. 18, 03-17-06)
ACTS ASPEN Complaint Tracking System
AIDS Auto-immune deficiency syndrome
AO Accreditation Organization
the ACT Social Security Act
CFC Conditions for Coverage
CFR Code of Federal Regulations
CLIA Clinical Laboratory Improvement Amendments
CMP Civil Monetary Penalties
CMS Center for Medicare and Medicaid Services
CO Central Office
CoP Conditions of Participation
COW Certificate of Waiver
DED Dedicated Emergency Department
DHHS Department of Health and Human Services
EMS Emergency Medical System
EMTALA Emergency Medical Treatment and Labor Act
ESRD End-Stage Renal Disease
FDA Food and Drug Administration
HHA Home Health Agency
HIV Human Immunodeficiency Virus
IJ Immediate Jeopardy
JCAHO Joint Commission on Accreditation of Healthcare Organizations
LSC Life Safety Code
NF Nursing Facility
NW Networks
OCR Office of Civil Rights
OIG Office of Inspector General
OSHA Occupational Safety and Health Administration
P & A Protection and Advocacy Group
PHSA Public Health Service Act
POC Plan of Correction
PPM Provider Perform Microscopy (PPM) Procedures
PRTF Psychiatric Residential Treatment Facility
QIO Quality Improvement Organization
RFP Requirements for Participation
RNHCI Religious Non-Medical Health Care Institutions
RO Regional Office
SA State Agency
SMA State Medicaid Agency
SNF Skilled Nursing Facility
SOM State Operations Manual
USC United States Code

SOM Chapter 5 - Page 103
Transmittals Issued for this Chapter
Rev #
Issue Date
Subject
Impl Date
CR#
R212SOM
02/10/2023
Revisions to State Operations Manual (SOM)
Chapter 5
10/24/2022
N/A
R208SOM
10/21/2022
Revisions to State Operation Manual (SOM),
Appendix PP
Guidance to Surveyors for Long
Term Care Facilities
10/24/2022
N/A
R191SOM
07/19/2019
Revisions to the State Operations Manual
(SOM) Chapter 5 and Appendix V
07/19/2019
N/A
R155SOM
06/10/2016
Revisions to State Operations Manual (SOM)
Chapter 5
06/10/2016
N/A
R120SOM
09/19/2014
Revisions to State Operations Manual (SOM)
Chapter 5
09/19/2014
N/A
R88SOM
08/27/2013
Revisions to State Operations Manual (SOM)
Chapter 5
07/19/2013
N/A
R86SOM
07/19/2013
Revisions to State Operations Manual (SOM)
Chapter 5 – Rescinded and replaced by
Transmittal 88
07/19/2013
N/A
R50SOMA
07/10/2009
Revisions to Chapter 5, "Complaint
Procedures"
07/10/2009
N/A
R18SOMA
03/17/2006
Revisions to Chapter 5, "Complaint
Procedures"
03/17/2006
N/A
R01SOM
05/21/2004
Initial Issuance of Pub 100-07
N/A
N/A
