
U.S. Department of Health and Human Services
Public Health Service
SF424 (R&R)
Application Guide
for NIH and Other PHS
Agencies
A guide developed and maintained by NIH for preparing
and submitting applications via Grants.gov to NIH and
other PHS agencies using the SF424 (R&R)
Forms Version C application packages
Updated July 25, 2013

PHS SF424 (R&R) Forms Version C Application Guide
SF424(R&R) ii
TABLE OF CONTENTS
PART I. INSTRUCTIONS FOR PREPARING AND SUBMITTING AN APPLICATION
1. Foreword ............................................................................................................................................... 1
1.1 Application Guide Format ............................................................................................................ 3
1.2 NIH Extramural Research and Research Training Programs ....................................................... 4
1.3 Research Grant Activity Codes and Program Guidelines ............................................................. 4
1.4 Interactions with PHS Staff .......................................................................................................... 5
1.5 Grants Policy Statements .............................................................................................................. 8
1.6 References .................................................................................................................................... 8
1.7 Authorization .............................................................................................................................. 10
1.7.1 Collection of Personal Demographic Data ................................................................... 10
1.8 Paperwork Burden ...................................................................................................................... 10
2. Process for Application Submission via Grants.gov........................................................................ 11
2.1 Overview .................................................................................................................................... 11
2.2 Registration Processes ................................................................................................................ 12
2.2.1 Grants.gov Registration ............................................................................................... 12
2.2.2 eRA Commons Registration ........................................................................................ 12
2.3 Software Requirements ............................................................................................................... 15
2.3.1 Adobe Reader............................................................................................................... 15
2.3.2 Creating PDFs for Text Attachments ........................................................................... 15
2.3.3 Special Instructions for Macintosh Users .................................................................... 15
2.4. Funding Opportunities ................................................................................................................ 16
2.4.1 NIH Guide for Grants and Contracts ........................................................................... 16
2.4.2 Funding Opportunity Announcements ......................................................................... 16
2.4.3 Finding a Funding Opportunity Announcement (FOA) for Grants.gov Submission ... 17
2.5 Forms for an Application to NIH or Other PHS Agencies ......................................................... 20
2.6 Format Specifications for Text (PDF) Attachments ................................................................... 22
2.7 “Resubmission” Applications ..................................................................................................... 24
2.8 “Revision” Application ............................................................................................................... 26
2.9 Similar, Essentially Identical, or Identical Applications ............................................................ 26
2.10 Submi
tting Your Application Via Grants.gov ............................................................................ 27
2.11 After You Submit Your Application Via Grants.gov ................................................................. 28
2.12 Correcting Errors ........................................................................................................................ 30
2.13 Post-Submission Application Materials ..................................................................................... 32
2.14 Application Submission Dates .................................................................................................... 34
2.15 Submission, Review and Award Cycles ..................................................................................... 35
2.16 Resources for Finding Help ........................................................................................................ 36
2.16.1 Finding Help for Grants.gov Registration or Submissions .......................................... 36

PHS SF424 (R&R) Forms Version C Application Guide
SF424(R&R) iii
2.16.2 Finding Help for the eRA Commons Registration or eRA Commons Validation
Processes ...................................................................................................................... 37
2.16.3 Finding Help for Application Preparation ................................................................... 38
3. Using the Grant Application Package .............................................................................................. 38
3.1 Verify Grant Information ............................................................................................................ 38
3.2 Enter the Name for the Application ............................................................................................ 39
3.3 Open and Complete Mandatory Documents ............................................................................... 39
3.4 Open and Complete Optional Documents .................................................................................. 39
3.5 Submitting the Application via Grants.gov ................................................................................ 40
4. Completing the SF424 Research and Related (R&R) Forms ......................................................... 40
4.1 Overview .................................................................................................................................... 40
4.2 Cover Form ................................................................................................................................. 41
4.3 Project/Performance Site Locations Form .................................................................................. 57
4.4 Other Project Information Form ................................................................................................. 61
4.5 Senior/Key Person Profile (Expanded) Form ............................................................................. 68
4.6 Selecting the Appropriate Budget Form ..................................................................................... 77
4.7 R&R Budget Form ...................................................................................................................... 78
4.7.1 Section A and B ........................................................................................................... 78
4.7.2 Sections C through E ................................................................................................... 84
4.7.3 Sections F through K ................................................................................................... 86
4.7.4 Cumulative Budget ...................................................................................................... 91
4.8 Special Instructions for Preparing Applications with a Subaward/Consortium ......................... 93
5. Completing PHS 398 Form ................................................................................................................ 97
5.1 Overview .................................................................................................................................... 97
5.2 (Reserved) ................................................................................................................................... 98
5.3 Cover Page Supplement Form .................................................................................................... 98
5.4 Modular Budget Form .............................................................................................................. 103
5.4.1 Budget Period Form ................................................................................................... 105
5.4.2 Cumulative Budget Information ................................................................................ 107
5.5 PH
S 398 Research Plan Form................................................................................................... 109
5.6 (Reserved) ................................................................................................................................. 120
5.7 (Reserved) ................................................................................................................................. 120
5.8 Planned Enrollment Report and Cumulative Inclusion Enrollment Report .............................. 120
5.8.1 Planned Enrollment Report .............................................................................................. 121
5.8.2 Cumulative Inclusion Enrollment Report ........................................................................ 123
6. Peer Review Process ......................................................................................................................... 126
7. Supplemental Instructions to the SF 424(R&R) for Preparing an Individual Research Career
Development Award (CDA) Application (“K” Series) ................................................................. 130
7.1 Introduction .............................................................................................................................. 130

PHS SF424 (R&R) Forms Version C Application Guide
SF424(R&R) iv
7.2 Individual Career Development Award Programs .................................................................... 130
7.3 Letters of Reference (must be submitted electronically through the eRA Commons) ............. 131
7.4 K- Specific Instructions for K Applications using the SF424 (R&R) Application .................. 134
7.4.1 Special Instructions for 4.2 Cover Form .................................................................... 134
7.4.2 Special Instructions for 4.3 Research & Related Project/Performance Site Locations
Form ........................................................................................................................... 134
7.4.3 Special Instructions for 4.4 Other Project Information Form ................................... 134
7.4.4 Special Instructions for 4.5 Senior/Key Person Profile(s) Form ................................ 135
7.4.5 Special Instructions for 4.6 Selecting the Appropriate Budget Form ........................ 138
7.4.6 Special Instructions for 4.7 R&R Budget Form ......................................................... 138
7.4.7 Special Instructions for 5. Completing PHS 398 Forms ........................................... 139
7.5 PHS 398 Career Development Award Supplemental Form ..................................................... 140
7.6 Career Development Award Peer Review Process ................................................................... 160
8. Supplemental Instructions to the SF424 (R&R) for Preparing Institutional Ruth L. Kirschstein
National Research Service Award (NRSA) Application ............................................................... 161
8.1 Introduction .............................................................................................................................. 161
8.2 Institutional Research Training Programs ................................................................................ 162
8.3 Reserved ................................................................................................................................... 162
8.4 Specific Instructions for Institutional Training Grant Applications using the SF424 (R&R)
Application ............................................................................................................................... 162
8.4.1 Special Instructions for 4.2 Cover Form .................................................................... 162
8.4.2 Special Instructions for 4.3 Research & Related Project/Performance Site Locations162
8.4.3 Special Instructions for 4.4 Research & Related Other Project Information Form ... 162
8.4.4 Special Instructions for 4.5 Senior/Key Person Profile (Expanded) Form ................ 164
8.4.5 Special Instructions for 4.7 Research & Related Budget ........................................... 165
8.4.6 Special Instructions for 4.6 PHS 398 Cover Page Supplement ................................. 165
8.5 PHS 398 Training Budget ......................................................................................................... 165
8.6 PHS 398 Training Subaward Budget Attachment(s) Form ...................................................... 171
8.7 Research Training Program Plan Form .................................................................................... 174
8.8 Training Grant Peer Review Process ........................................................................................ 189
9. Supplemental Instructions to the SF 424 (R&R) for Preparing a Multi-Project Application .. 190
9.1 Introduction .............................................................................................................................. 190
9.2 Multi-Project Applications ....................................................................................................... 190
9.2.1 Coll
aborating with Other Organizations .................................................................... 191
9.3 Application Submission System & Interface for Submission Tracking (ASSIST) .................. 191
9.4 Special Instructions to the SF424 R&R for Multi-Project Applications .................................. 193
9.4.1 Special Instructions for 4.2 Cover Form .................................................................... 193
9.4.2 Special Instructions for 4.3 Project/Performance Site Locations ............................... 194
9.4.3 Special Instructions for 4.4 Other Project Information Component .......................... 194
9.4.5 Special Instructions for 4.5 Senior/Key Person Profile (Expanded) .......................... 196
9.4.6 Special Instructions for 4.7 R&R Budget .................................................................. 197
PART I
Instructions for Preparing and Submitting
an Application

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-1
1. Foreword
Forms Version C Application Guide — Released July 25, 2013
This application includes changes to SF424 Research & Related (R&R) form instructions necessitated by
the OMB renewal and Grants.gov’s subsequent release of updated forms in June 2013. Changes have also
been made to various PHS 398 forms and instructions approved by OMB in August 2012 and released by
Grants.gov in June 2013. Parts II (Supplemental Instructions for Preparing the Human Subjects Section of
the Research Plan) and III (Policies, Assurance, Definitions, and Other Information) of the previous
version of the application guide have been moved to a stand-alone document titled, “Supplemental Grant
Application Instructions.”
Modifications include the following:
SF424 (R&R) Forms
System for Award Management (SAM) Registration
• A change from the requirement to register with the Central Contractor Registry Database (CCR)
to the System for Award Management (SAM).
SF424 (R&R) Cover Form
• A new field for the “Previous Grants.gov Tracking ID” is included. Form behavior will be
adjusted so that applications with Submission Type of “Change/Corrected Application” require
an entry in the “Previous Grants.gov Tracking ID” field rather than requiring an entry in the
“Federal Identifier” field.
• The Person to be contacted section of the Applicant Information has expanded to include the
additional contact information we have been including on the PHS 398 Cover Page Supplement
form. The additional contact information has been removed from the PHS 398 Cover Page
Supplement.
• The label for the “SFLL or other Explanatory Documentation” has been changed to “SFLLL
(Disclosure of Lobbying Activities) or other Explanatory Documentation.”
• A new “Cover Letter Attachment” has been added and a separate PHS Cover Letter form will no
longer be used. However, NIH will continue to keep the Cover Letter separate from the
assembled application image and available only to authorized staff.
SF424 (R&R) Budget Form
• The cumulative Number of Participants/Trainees allowed on the SF424 (R&R) budget form and
associated Subaward Budget forms has been expanded from 999 to 9999 to address a form bug.
On previous form versions, maxing out the Number of Participants/Trainees on all budget periods
caused an application to fail because the forms didn’t allow a bigger number for the cumulative
total.
SF 424 (R&R) Sr/Key Person Expanded Form
• The number of Sr/Key Persons that can be entered has been expanded from 40 to 100.
SF 424 (R&R) Project Performance Site Locations Form
• The number of Performance Sites that can be entered has been expanded from 30 to 300.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-2
PHS 398 Forms
Planned Enrollment Report Form & PHS 398 Cumulative Inclusion Enrollment Report Form
• Added optional inclusion forms in application packages to allow for the collection as discrete data
rather than .pdf attachments.
• Removed enrollment and inclusion attachment fields from PHS 398 Research Plan, PHS 398
Career Development Award Supplemental Forms.
PHS 398 Career Development Award Supplemental Form
• Removed “Application Type” section.
• Removed “Career Development Award Attachment” header.
• Removed Inclusion and Enrollment attachments (now separate forms in the application package).
• Added a new attachment for “Letters of Support from Collaborators, Contributors, and
Consultants.”
• Renamed attachment from “Career Development/Training Activities During Award Period” to
“Candidate’s Plan for Career Development/Training Activities During Award Period.”
• Renamed attachment from “Mentoring Plan” to “Candidate's Plan to Provide Mentoring (as
applicable).”
• Changed section header from “Statements of Support” to “Statements and Letters of Support.”
• Made adjustments to format and numbering.
PHS 398 Research Plan Form
• Removed “Application Type” section.
• Removed “Research Plan Attachment” header.
• Removed Inclusion and Enrollment attachments (now separate forms in the application package).
• Made adjustments to format and numbering.
PHS 398 Training Program Plan Form
• Removed “Application Type” section.
• Removed “Research Training Program Plan Attachment” header.
• Renamed attachment field from “Introduction to Application (for REVISION or
RESUBMISSION applications only)” to “Introduction to Application (for RESUBMISSION or
REVISION only).”
• Made adjustments to format and numbering.
PHS 398 Training Subaward Budget Attachment Form
• Limit of subaward budget attachments has increased from 10 to 30.
PHS 398 Cover Page Supplement Form
• Removed Applicant Organization Contact information that will be included on SF424 R&R
Cover Form.
• Added fields from PHS 398 Checklist form. The separate PHS 398 Checklist Form will no
longer be used.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-3
• Limit of collection of human embryonic stem cell lines has increased from 20 to 200.
• Made adjustments to format and numbering.
Notes
This version of the Application Guide now also includes a separate instruction section for multi-
component applications to accommodate their transition to electronic submission effective with
submissions on/after September 25, 2013.
For additional details on all the form changes noted above, see NIH Guide Notice NOT-OD-13-074
.
This application guide contains instructions and other useful information for preparing grant applications
to the National Institutes of Health (NIH) and other Public Health Service (PHS) agencies for:
Public Health Service (PHS) Research Grants
Career Development (K) Awards
Institutional Training (T) Grants
Supplemental Instructions to the SF424 (R&R) for Preparing a Multi-Project Application
This application guide is used as a companion document to the SF424 Research and Related (R&R)
application forms. In addition to the SF424 (R&R) forms, applications to NIH and other PHS agencies
will include agency-specific forms, titled “PHS 398.” These PHS 398 forms were developed to continue
the collection of agency-specific data required for a complete application. While these agency-specific
forms are not identical to the PHS 398 application form pages, the PHS 398 reference is used to
distinguish these additional data requirements from the data collected in the SF424 (R&R) forms. A
complete application to NIH and other PHS agencies will include SF424 (R&R) forms and PHS 398
forms. Instructions for all application forms, SF424 (R&R) and PHS 398, are found in this document.
The use of these forms also involves electronic submission of completed applications through Grants.gov.
NIH and other PHS agencies continues to transition all activity codes to the new application forms and
Grants.gov submission. NIH will continue to use Requests for Applications (RFAs) and Program
Announcements (PAs) as categories of FOAs. See Section 2.4.2
for definitions.
Applicants must carefully review FOAs for guidance on when to use the SF424 (R&R) forms,
instructions, and electronic submission for a specific activity code (i.e., P01, P20, P50, etc.). This process
will apply to all types of submissions for the announced activity code—new, resubmission, renewal, and
revision grant applications. Each FOA will include a link to the most current version of these instructions.
Applicants are encouraged to check the website frequently for the most current version.
For purposes of this document, any references to “NIH” may also mean “NIH and other PHS agencies”
such as the Agency for Healthcare Research and Quality (AHRQ), the Centers for Disease Control and
Prevention (CDC), and the Food and Drug Administration (FDA).
1.1 Application Guide Format
This application guide is organized into three distinct parts:
Part I
: Instructions for Preparing and Submitting the Application. Part I includes specific
instructions for completing the application forms as well as information on electronically submitting
applications through Grants.gov.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-4
The following Parts are incorporated into this application guide by reference. They are in a separate
document titled, “Supplemental Instructions
.”
Part II: Supplemental Instructions for Preparing the Human Subjects Section of the Research
Plan. Part II is to be used if your proposed research will involve human subjects. These instructions
assist in determining whether human subjects are involved and include scenarios and detailed
instructions for completing the PHS 398 Research Plan form.
Part III: Policies, Assurance, Definitions, and Other Information. Part III includes information
on policies, assurances, definitions, and other information relating to submission of applications to
the PHS. Applicants should refer to this document as well as the instructional materials, Grants
Information (GrantsInfo), and the relevant Grants Policy Statement for additional sources of
information. The
NIH Grants Policy Statement applies to all NIH awardees; other PHS agencies use
the HHS Grants Policy Statement.
1.2 NIH Extramural Research and Research Training
Programs
The NIH Office of Extramural Research Grants homepage (http://grants.nih.gov/grants/oer.htm) provides
an array of helpful information. Applicants are encouraged to bookmark this site and visit it often.
The Division of Communications and Outreach (DCO) is the central source for general information about
NIH extramural research and research training programs, funding activity codes, the peer review system,
and application procedures. Grants Information (GrantsInfo) is a communication service within the DCO.
Information about the NIH extramural research and research training programs, funding opportunities,
and the grant application process, can be obtained by e-mailing your request to: GrantsI[email protected]
or
by calling (301) 435-0714.
1.3 Research Grant Activity Codes and Program Guidelines
A partial list of research grant activity codes and programs are provided below. As noted in the
descriptions in Supplemental Instructions
Part III: Policies, Assurances, Definitions, and Other
Information, not all awarding components use all activity codes or programs. For a complete listing of
program guidelines, visit the OER Grants website
http://grants.nih.gov/grants/funding/funding_program.htm.
Research Grants
• Basic Research Grant (R01)
• Small Research Grant (R03)
• Academic Research Enhancement Award (AREA) (R15)
• Exploratory/Developmental Grant (R21, R33, R21/R33)
• Small Business Innovation Research Grant (SBIR) (R43/R44)
• Small Business Technology Transfer Grant (STTR) (R41/R42)
• Program Project Grant (P01)
• Research Center Grant (P50)
• Scientific Meeting Support (R13, U13)

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-5
• Research Project Cooperative Agreement (U01)
• Research Grants to Foreign Institutions and International Organizations
Training, Fellowships and Career Development Programs
• NIH Institutional Ruth L. Kirschstein National Research Service Award (T32)
• Individual Ruth L. Kirschstein National Research Service Award Fellowships (NRSA) (F30, F31,
F32, F33, F34, etc.)
• Research Career Development Award (K Award)
Applications Available from Other Offices
• Health Services Project Application (5161-1)
1.4 Interactions with PHS Staff
The PHS agencies encourage applicants to communicate with staff throughout the entire application,
review and award process. Website addresses and staff phone numbers of relevant NIH awarding
components and other PHS agencies are listed in the table below.
Table 1.4-1. PHS Agency Contact Table
PHS AGENCY
(LINK TO WEBSITE)
AWARDING COMPONENT
(LINK TO WEBSITE)
TELEPHONE
NUMBER
National Institutes of Health
(NIH)
Eunice Kennedy Shriver National Institute of Child Health and
Human Development
301-496-0104
NIH Fogarty International Center 301-496-1653
NIH National Cancer Institute 301-496-3428
NIH National Center for Complementary and Alternative Medicine 301-496-4792
NIH National Center for Advancing Translational Sciences 301-496-6023
NIH National Eye Institute 301-451-2020
NIH National Heart, Lung, and Blood Institute 301-435-0260
NIH National Human Genome Research Institute 301-496-7531
NIH National Institute on Aging 301-496-9322
NIH National Institute on Alcohol Abuse and Alcoholism 301-443-4375
NIH National Institute of Allergy and Infectious Diseases 301-496-7291
NIH National Institute of Arthritis and Musculoskeletal and Skin Diseases 301-594-2463
NIH National Institute of Biomedical Imaging and Bioengineering 301-451-4792
NIH National Institute on Deafness and Other Communication Disorders 301-496-1804
NIH National Institute of Dental and Craniofacial Research 301-594-4800
NIH National Institute of Diabetes and Digestive and Kidney Diseases 301-594-8834
NIH National Institute on Drug Abuse 301-443-2755

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-6
PHS AGENCY
(
LINK TO WEBSITE
)
AWARDING COMPONENT
(
LINK TO WEBSITE
)
TELEPHONE
NUMBER
NIH National Institute of Environmental Health Sciences 919-541-7723
NIH National Institute of General Medical Sciences 301-594-4499
NIH National Institute of Mental Health 301-443-3367
NIH National Institute on Minority Health and Health Disparities 301-402-1366
NIH National Institute of Neurological Disorders and Stroke 301-496-9248
NIH National Institute of Nursing Research 301-594-6906
NIH National Library of Medicine 301-496-4621
NIH Center For Scientific Review 301-435-0715
Agency for Healthcare
Research and Quality
Agency for Healthcare Research and Quality 301-427-1447
Centers for Disease Control
and Prevention (CDC)
Coordinating Center for Health Information and Services 404-498-1186
CDC Office of Infectious Disease 404-639-3770
CDC National Center for Environmental Health 770-488-4668
CDC National Center for Injury Prevention and Control 800-232-4636
CDC
National Center for Chronic Disease Prevention and Health
Promotion
770-488-8390
CDC National Institute for Occupational Safety and Health 404-498-2530
CDC Procurement and Grants Office 770-488-2700
Food and Drug
Administration
Food and Drug Administration 301-827-7185
Indian Health Service Indian Health Service 301-443-0578
Agency for Toxic
Substances and Disease
Registry
Agency for Toxic Substances and Disease Registry 404-842-6630
Office of Public Health and
Science
Office of Population Affairs 301-594-4004
Office of Public Health and
Science
Office of Family Planning 301-594-4008
Before Submission
You may wish to contact NIH staff with a variety of questions before submitting an application.
Contact GrantsInfo and/or the
Division of Receipt and Referral, Center for Scientific Review (CSR),
NIH:
• To identify Institutes/Centers (ICs) at NIH or other non-NIH agencies and/or a Scientific Review
Group (SRG) that might be appropriate for your application. Note requests for assignment to an
Institute/Center and/or a SRG may be made in a cover letter
at the time of application submission.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-7
• To learn about grant programs.
• To receive advice on preparing and submitting an application (e.g., format, structure).
Contact program staff in the relevant awarding component:
• To determine whether your proposed application topic would fit into the NIH IC’s or other non-
NIH agency’s programmatic area.
• To learn about programmatic areas of interest to the IC or other non-NIH agencies.
• To find out about requesting an assignment to an IC.
• To discuss whether you should respond to an RFA.
Contact Scientific Review Officers in the CSR to discuss requesting assignment to a CSR SRG.
After Submission
If the initial assignment to an IC or SRG seems inappropriate, the Program Director/Principal Investigator
(PD/PI) may request reassignment. Such requests should be made in writing to:
Division of Receipt and Referral
Center for Scientific Review
National Institutes of Health
6701 Rockledge Drive, Suite 2030, MSC 7720
Bethesda, MD 20892-7720
Fax requests (301-480-1987) are also acceptable.
Although these requests will be carefully considered, the final determination will be made by the PHS
agency.
Applicants must never contact reviewers regarding their applications because discussion of the scientific
content of an application or an attempt to influence review outcomes will create serious breaches of
confidentiality in the review process. Reviewers are required to notify the Scientific Review Officer if
they are contacted by an applicant. Communication by the applicant to a reviewer may delay the review
or result in the return of the application without review.
Communications from the Division of Receipt and Referral (DRR) are accessible to applicants and
applicant organizations in the eRA Commons in a new “Correspondence” section of the Commons
detailed status screen for the application. Applicants will be notified by email to check their Commons
account. DRR will notify an applicant when: 1) additional information is required before her/his
application can be assigned to a scientific review group (SRG) and NIH Institute or Center (IC) for
funding consideration; 2) an applicant’s request for an IC assignment cannot be honored; or 3) it has been
determined that the application does not comply with NIH policy. For additional information, see NIH
Guide Notice NOT-OD-12-108
.
After Assignment
Contact your Scientific Review Officer to discuss the review assignment, to request permission to send
additional/corrective materials, and/or to discuss any review concerns (e.g., expertise needed on your
SRG, conflicts, reviewers that may have bias).
After Peer Review
Feedback to applicants is very important. Once the PD/PI reviews the Summary Statement
in the eRA
Commons, the appropriate awarding component program official noted in the Summary Statement may
be contacted:

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-8
• To discuss the review outcome of the application and obtain guidance.
• To get feedback and answers to any questions about the Summary Statement.
• To find out the meaning of a numerical designation pertaining to human subjects or vertebrate
animals in the Summary Statement.
• To find out the funding status of an application.
A paper copy of the Peer Review Outcome Letter and Summary Statement will not be mailed to the PI
and may only be accessed through the eRA Commons.
1.5 Grants Policy Statements
• The NIH Grants Policy Statement serves as a term and condition of award and is a compilation of
the salient features of policies and various policy issues regarding the administration of NIH
awards.
• The HHS Grants Policy Statement serves as a term and condition of award and is a compilation
of the salient features of policies and various policy issues regarding the administration of grant
awards from other PHS agencies, excluding NIH awards.
1.6 References
Applicants New to NIH: Getting Started
http://grants.nih.gov/grants/useful_links.htm
Award Information and Data
http://report.nih.gov/index.aspx
NIH Research Portfolio Online Reporting Tool (RePORT)
Contact Information for an NIH Staff Person
http://ned.nih.gov
NIH locator: (301) 496-4000
Applying Electronically
For additional information on the electronic submission process, including self-help resources,
training material and answers to frequently asked questions, see:
http://grants.nih.gov/grants/ElectronicReceipt/index.htm
eRA Commons
https://commons.era.nih.gov/commons/index.jsp
Institutions and PD/PIs are required to register with the eRA Commons. Registered PD/PIs can
check assignment/contact information, review outcome, and other important information. For more
details on Commons registration, see Section 2.2.2
.
E-mail: [email protected]ih.gov.
Telephone: 1-866-504-9552 (toll-free) or 301-402-7469; 301-451-5939 (TTY). Business hours are
M-F 7am-8pm Eastern Time.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-9
Grant Writing Tips and Sample Applications
http://grants.nih.gov/grants/grant_tips.htm
Grants Information
http://grants.nih.gov/grants/giwelcome.htm
E-mail: Grant[email protected]
Telephone: (301) 435-0714; (301) 451-5936 (TTY)
Grants.gov User Guide
The Grants.gov User Guide is a comprehensive reference to information about Grants.gov.
Applicants can download the User Guide at the following address:
http://www.grants.gov/assets/ApplicantUserGuide.pdf
.
NIH Office of Extramural Research Human Subjects Website
http://grants.nih.gov/grants/policy/hs/index.htm
This site provides, in one place, DHHS and NIH requirements and resources for the extramural
community involved in human subjects research.
Office for Human Research Protections (Department of Health and Human Services)
http://www.hhs.gov/ohrp
Information about human subject protections, Institutional Review Boards, and Federal Wide
Assurances
Telephone: 1-866-447-4777 or (301) 496-7005
Office of Laboratory Animal Welfare (OLAW)
http://olaw.nih.gov
Information about animal welfare policy requirements, Institutional Animal Care and Use
Committees (IACUC), and Animal Welfare Assurances
Telephone: (301) 496-7163
Receipt/Referral of an Application
http://www.csr.nih.gov/EVENTS/AssignmentProcess.htm
Division of Receipt and Referral
Center for Scientific Review
Telephone: (301) 435-0715
Fax: (301) 480-1987
Specific Application: Before Review
Telephone or e-mail the Scientific Review Officer identified for the application in the eRA
Commons.
Specific Application: Post Review
Telephone or e-mail the NIH Program Official named in the Summary Statement for the application.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-10
1.7 Authorization
The PHS requests the information described in these instructions pursuant to its statutory authorities for
awarding grants, contained in Sections 301(a) and 487 of the PHS Act, as amended (42 U.S.C. 241a and
42 U.S.C. 288). Therefore, such information must be submitted if an application is to receive due
consideration for an award. Lack of sufficient information may hinder the ability of the PHS to review an
application and to monitor the grantee’s performance.
1.7.1 Collection of Personal Demographic Data
Federal agencies have a continuing commitment to monitor the operation of its review and award
processes to detect, and deal appropriately with, any instances of real or apparent inequities. In addition,
section 403 of the 2007 NIH Reform Act requires NIH to report to Congress specifically on postdoctoral
individuals supported on research grants, and section 489 of the PHS Act requires NIH to perform a
continuing assessment of research personnel needs. Personal demographic data on PD/PIs and those with
a postdoctoral role is vital to comply with these requirements.
NIH collects personal data through the eRA Commons Personal Profile. The data is provided one-time by
the individual through a secure, electronic system, is confidential, and is maintained under the Privacy
Act record system 09-25-0036, “Grants: IMPAC (Grant/Contract Information).” When completing the
data entry in the Commons Personal Profile, the individual is responsible for providing true, accurate, and
complete data. All analyses conducted on date of birth, citizenship, gender, race, ethnicity, disability,
and/or disadvantaged background data will report aggregate statistical findings only and will not identify
individuals. Declining to provide information does not affect consideration of an application; however,
for some programs (e.g., Ruth L. Kirschstein National Research Service Awards and Research Career
Development Awards) citizenship data is required to determine eligibility.
The PHS also requests the last four digits of the Social Security Number (SSN) for accurate identification
of individuals and for management of PHS grant programs. Please be aware that no individual will be
denied any right, benefit, or privilege provided by law because of refusal to disclose this portion of the
SSN. The PHS requests the last four digits of the SSN under Section 301(a) and 487 of the PHS act as
amended (42 U.S.C. 241a and U.S.C. 288).
1.8 Paperwork Burden
The PHS estimates that it will take approximately 22 hours to complete this application for a regular
research project grant. This estimate excludes time for development of the scientific plan. Other items
such as human subjects are cleared and accounted for separately and therefore are not part of the time
estimate. An agency may not conduct or sponsor the collection of information unless it displays a
currently valid OMB control number. Nor is a person required to respond to requests for the collection of
information without this control number. Send comments regarding this burden estimate or any other
aspect of this collection of information, including suggestions for reducing this burden, to: NIH, Project
Clearance Office, 6705 Rockledge Drive MSC 7974, Bethesda, MD 20892-7974, ATT: PRA (0925-
0001). Do not send applications or any materials related to training or career award applications to this
address.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-11
2. Process for Application Submission via
Grants.gov
Application submission through Grants.gov involves several steps. Some of the steps need only be done
one time. Others are ongoing steps that will be necessary for each application submission. Before
beginning the application process, you are encouraged to review Grants.gov
and all the resources
available there.
2.1 Overview
The following steps must be taken in order to submit a grant application through Grants.gov. Please be
sure to complete all steps to ensure that NIH receives the application in a timely manner.
1. Register your organization at Grants.gov. (This is a one-time only registration process for all
Federal agencies. However, an annual renewal of information provided to the System for Award
Management (SAM
) is necessary to retain active Grants.gov credentials. If your organization has
already completed this step for any Federal agency submission, skip to step #2. If your
organization has not completed this step, see Section 2.2 for more details.)
2. Register your organization and Program Director/Principal Investigator (PD/PI) in the eRA
Commons. (This is a one-time only registration process. If your organization has already
completed this step, skip to step #3. If your organization has not completed this step, see
Section
2.2 for more details.)
3. Find a Funding Opportunity Announcement (FOA) of interest using the NIH Guide for Grants
and Contracts or Grants.gov’s “Find Grants” feature. (See Section 2.4
for more details.)
4. Download the associated Application Package from Grants.gov. (Adobe Reader required for
download. See Section 2.3
for more details.)
5. Complete the appropriate application forms, including all text and PDF attachments. Upload all
attachments into the appropriate application form (See Section 2.6
for more details on the
requirements for text (PDF) attachments).
6. Review the completed application through your own organizational review process.
7. Coordinate with an Authorized Organization Representative (AOR) at the applicant organization
to submit the application by the date and time specified in the FOA. (Keep a copy locally at the
Applicant Organization/Institution.)
8. Receive the Grants.gov tracking number.
9. After successfully passing Grants.gov and agency validation, receive the agency tracking
number (accession number). Note: Any errors encountered at Grants.gov or eRA Commons
must be corrected in order to successfully complete your submission.
10. PD/PI and Signing Official (SO) must view the application in eRA Commons to ensure the
assembled application correctly reflects their submission. (See Section 2.11
for detailed
information.)
The following sections explain each step in more detail.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-12
2.2 Registration Processes
2.2.1 Grants.gov Registration
Grants.gov requires a one-time registration by the applicant organization in order to submit applications.
Registration is not needed to download an application package or to prepare an application for
submission. PD/PIs do not have to individually register in Grants.gov unless they also serve as the
Authorized Organization Representative (AOR) for their institution/organization. If an applicant
organization has already completed Grants.gov registration for another Federal agency, they can skip this
section and focus on the eRA Commons registration steps noted below. For those applicant organizations
still needing to register with Grants.gov, registration information can be found at the Grants.gov Get
Registered tab (http://grants.gov/applicants/get_registered.jsp
). While Grants.gov registration is a one-
time only registration process, it does involve several steps and will take some time. Applicant
organizations needing to complete this process are encouraged to start early allowing at least six (6)
weeks to complete all the steps before actually submitting an application through Grants.gov.
All registrations require that applicants be issued a Dun and Bradstreet Universal Numbering System
(DUNS) number. After obtaining a DUNS number, applicants can begin both System for Award
Management (SAM) (formerly CCR) and eRA Commons registrations. The same DUNS number must be
used for all registrations and on the grant application itself.
Note that all applicant and grantee organizations must complete and maintain an active entity registration
in SAM which requires renewal at least annually. This requires that you review and update the
information at least annually after the initial registration, and more frequently if required by changes in
your information or another award term. The renewal process may require as much time as the initial
registration. SAM registration includes the assignment of a Commercial and Government Entity (CAGE)
Code for domestic organizations which have not already been assigned a CAGE Code. Foreign
organizations must obtain a NATO Commercial and Government Entity (NCAGE) code (in lieu of a
CAGE code) in order to register in SAM. Use the SAM.gov "Manage Entity" function to manage your
entity registrations. See the Grants Registrations User Guide at http://www.sam.gov
for additional
information.
For additional information regarding maintaining an active SAM registration, please see NIH Guide
Notice NOT-OD-11-004.
The AOR is an individual authorized to act for the applicant organization and to assume the obligations
imposed by the Federal laws, requirements, and conditions for a grant or grant application, including the
applicable Federal regulations. This individual has the authority to sign grant applications and required
certifications and/or assurances that are necessary to fulfill the requirements of the application process.
Once this individual is registered, the organization can then apply for any government funding
opportunity listed in Grants.gov, including NIH and other PHS agencies grants.
Questions regarding Grants.gov registration should be directed to the Grants.gov Contact Center at
. The Contact Center is available 24 hours
a day, 7 days a week.
2.2.2 eRA Commons Registration
The applicant organization, all PD/PIs, and all individuals with a postdoctoral role (see definition of
postdoctoral scholar
in Supplemental Instructions Part III.3) and one month or more of measurable effort
must also complete a one-time registration in the eRA Commons. Individuals with a postdoctoral role do
not need an eRA Commons ID for application submission. However, individuals with a postdoctoral role
must have an eRA Commons ID for reporting on awarded grants. Access to the Commons is vital for all

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-13
steps in the process after application submission. An organization and PD/PIs must be registered in the
Commons before they can take advantage of electronic submission and retrieval of grant information,
such as reviewing grant applications, institute/center assignments, review outcomes, and Summary
Statements. Applicants must have an active DUNS number in order to complete the eRA Commons
registration. Organizations can register with the eRA Commons as they are working through their SAM
or Grants.gov registration. eRA Commons requires organizations to identify at least one Signing Official
(SO) and at least one Program Director/Principal Investigator (PD/PI) account in order to submit an
application. Institutional/organizational officials are responsible for registering PD/PIs and individuals
with a postdoctoral role in the eRA Commons. PD/PIs and individuals with a postdoctoral role should
work with their AOR (also known as the Signing Official in the eRA Commons) to determine their
institutional/organizational process for registration.
IMPORTANT: The eRA Commons registration process should be started at least six (6) weeks prior to
the submittal date of a Grants.gov submission. Failure to register in the Commons and to include a valid
PD/PI Commons ID in the credential field of the Senior/Key Person Profile Form will prevent the
successful submission of an electronic application to NIH. Commons user name IDs for those with a
postdoctoral role are not required at the time of application submission, but are required as part of the
Non-Competing Continuation Progress Report (RPRR or PHS 2590).
2.2.2.1 Commons Registration for the Organization
Organizations may verify their current registration status by accessing the “List of Grantee Organizations
Registered in eRA Commons” (http://era.nih.gov/commons/quick_queries/index.cfm#commons
).
To register an Organization in the eRA Commons:
1. Complete the online Institution Registration Form at:
https://public.era.nih.gov/commons/public/registration/registrationInstructions.jsp
2. Print and fax the registration page
a. The Signing Official (SO) must sign, date and fax the registration to the number listed
on the form
3. Signing Official must verify email address
a. Once the registration is submitted electronically, email verification is sent and the SO
must click the link to verify email
b. The above steps must be completed before NIH can process the registration
c. The SO will receive an “Approval” email from NIH
d. The “Approval” email contains a link to information that you must verify as correct
before the confirmation process is completed
4. Account Administrator (AA) receives user names and temporary passwords
a. After the completion of the confirmation, the SO and AA will receive two emails that
contain the user names and temporary password for the SO and AA accounts created
during the registration process
5. Log into Commons
a. The SO and AA log into Commons and administer additional accounts as needed
6. Affiliate your PIs
a. Your Principal Investigators must work with your organization to be registered in
eRA Commons if they do not have an existing account. If they have an account, you

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-14
must affiliate it with your organization. Verify that you’ve selected the correct PI
account!
For more information, see http://era.nih.gov/commons/faq_commons.cfm#II2
.
This registration is independent of Grants.gov and may be done at any time.
Organizational data elements, such as Institutional Profile Number (IPF), Entity Identification Number
(e.g., 5555555555A5) and DUNS Number must be accurately identified. Note the DUNS number must
be included in the Institutional Profile for applications to be accepted. In addition, the DUNS
number in the Institutional Profile must match that entered in the SF424 (R&R) Cover Form in
Section 5, Applicant Information.
Prior to submission, the AOR/SO should verify that their organization’s eRA Commons profile contains
the valid DUNS number that will be used for the submission process. The SO has the ability to edit this
field in the organization profile in Commons.
To confirm that your organization has a DUNS number or to find out if the DUNS number you have
matches the one in Commons, access the List of Grantee Organizations Registered in eRA Commons
(http://era.nih.gov/commons/quick_queries/index.cfm#commons
). This listing of grantee organizations
registered in Commons and their DUNS numbers can be accessed without logging into Commons.
2.2.2.2 Commons Registration for the Program Directors/Principal Investigators
(PD/PIs) and Individuals with a Postdoctoral Role
Once the organization is registered in the Commons, individuals can then be registered. All PD(s)/PI(s)
must have an eRA Commons account and the PI Role and should work with their organizational officials
to either create a new account or to affiliate an existing account with the applicant organization’s eRA
Commons account. If the PD/PI is also the organizational signing official, they must have two distinct
eRA Commons accounts, one for each role. This registration must be done by an organizational
official (or delegate) who is already registered in the Commons. If submitting an application reflecting
Multiple PD/PIs, the individual designated as the contact PI must be affiliated with the applicant
organization. To register PD/PIs in the Commons, refer to the eRA Commons System Users Guide
(http://era.nih.gov/commons/user_guide.cfm
). For applications reflecting Multiple PD/PIs, all such
individuals must be assigned the PI role, even those at organizations other than the applicant organization.
Once a PD/PI has received e-mail confirming his/her registration within the Commons, the PD/PI must
verify that all Personal Information located within the Personal Profile tab in the eRA Commons System
is accurate. Please have the PD/PI review and update, as needed, data elements such as first name, middle
initial, last name, prefix and/or suffix to PD/PI name (including all embedded punctuation), e-mail,
phone, fax, street address, city, state, country, zip and degrees earned. These data must contain the most
recent information in order for the application to be processed accurately.
Both PD/PI and SO need separate accounts in Commons since each role enables different features within
eRA Commons. For example, an SO can Reject a submitted application to prevent it from being sent on
to agency staff. A PI has access to review outcome information (scores and summary statement). If you
are the SO for your organization as well as a PD/PI of the grant, you will need two separate accounts with
different user names – one with SO authority and one with PI authority. When an organization is
registered, an SO account is created. Log on to the account with the SO authority role and create another
account with PI authority.
Individuals with a postdoctoral role and one month or more of effort must also be registered in the eRA
Commons and should verify that all Personal Information located within the Personal Profile tab in the
eRA Commons system is accurate. The Commons user name ID for those with a postdoctoral role is not
required at the time of application submission, but will be required as part of the Non-Competing
Continuation Progress Report (RPRR or PHS 2590).

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-15
For additional information on how to prepare for electronic submission, see:
http://grants.nih.gov/grants/ElectronicReceipt/preparing.htm
.
2.3 Software Requirements
2.3.1 Adobe Reader
In order to access, complete and submit applications using Grants.gov’s downloadable forms, applicants
need to download and install the Adobe Reader, version 8.1.1 or later. For minimum system requirements
and download instructions, please see the Grants.gov User Guide
or visit
http://grants.gov/help/download_software.jsp.
2.3.2 Creating PDFs for Text Attachments
NIH and other PHS agencies require all text attachments to the SF424 (R&R) application forms to be
submitted as PDF files.
Applicants should prepare text attachments using any word processing program (following the format
requirements in Section 2.6
) and then convert those files to PDF before attaching the files to the
appropriate form in the application package. (The PDF format is used to preserve document formatting.)
Save all files with descriptive file names of 50 characters or less and be sure to only use standard
characters in file names: A through Z, a through z, 0 through 9, underscore (_), hyphen (-), space ( ), and
period (.). Do not use any other special characters (e.g., “&”, “*”, “%”, “/”, or “#”) in the file name.
Some type of PDF-creation software is necessary to create the PDF. (The free Adobe Reader will not
create a PDF.) To assist applicants searching for free PDF-creation software, Grants.gov has published a
list of available tools and software, see Grants.gov’s Download Software page at
http://www.grants.gov/help/download_software.jsp
.
Note that all PDF attachments must be submitted as individual files. Although some software packages
allow bundling of multiple PDFs into a single file, eRA systems cannot support “Bundling” or “Portfolio”
features at this time. Use of these features may result in delays in the review of an application or an
application not being reviewed.
It is recommended that, as much as possible, applicants avoid scanning text documents to produce the
required PDFs. Instead, NIH recommends producing the documents electronically using text or word-
processing software and then converting documents to PDF. Scanning paper documents, without the
proper Optical Character Recognition (OCR) process, will hamper automated processing of your
application for NIH analysis and reporting.
DISCLAIMER: References to software packages or Internet services neither constitute nor should be
inferred to be an endorsement or recommendation of any product, service, or enterprise by the NIH or
other PHS agencies, any other agency of the United States Government, or any employee of the United
States Government. No warranties are stated or implied.
2.3.3 Special Instructions for Macintosh Users
With the conversion to Adobe Reader application submissions there are no longer special instructions for
Macintosh users.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-16
2.4. Funding Opportunities
Grants for health-related research and research training projects or activities make up the largest category
of funding provided by the NIH Institutes/Centers (ICs) and other non-NIH agencies. Most applications
for support are unsolicited and originate with individual investigators who develop proposed plans for
research or research training within an area that is relevant to the NIH. Research project grants are
awarded to organizations/institutions on behalf of PD/PIs to facilitate the pursuit of a scientific objective
when the idea for the research is initiated by the investigator. If the funding agency anticipates substantial
program involvement during the conduct of the research, a cooperative agreement will be awarded rather
than a grant. The NIH typically awards grants and cooperative agreements for terms ranging from one to
five years. Organizational/institutional sponsorship assures that the awardee organization will provide the
facilities and the financial stability necessary to conduct the research, and be accountable for the funds.
For a list and brief description of grant activity codes, see Supplemental Instructions
Part III: Policies,
Assurances, Definitions, and Other Information.
2.4.1 NIH Guide for Grants and Contracts
The NIH Guide for Grants and Contracts (http://grants.nih.gov/grants/guide), a weekly electronic
publication, contains announcements about funding opportunities, such as Requests for Applications
(RFAs) and Program Announcements (PAs), including Parent Announcements, from NIH and other PHS
agencies. The NIH Guide also contains vital information about policies and procedures. To subscribe to
the NIH Guide, visit
http://grants.nih.gov/grants/guide/listserv.htm.
2.4.2 Funding Opportunity Announcements
To hasten the development of a program or to stimulate submission of applications in an area of high
priority or special concern, an awarding component will encourage applications through the issuance of a
PA to describe new, continuing, or expanded program interests, or issuance of an RFA inviting
applications in a well-defined scientific area to accomplish a scientific purpose.
Definitions are as follows:
Parent Announcements: Electronic grant applications must be submitted in response to a Funding
Opportunity Announcement (FOA). For applicants who wish to submit what were formerly termed
“investigator-initiated” or “unsolicited” applications, NIH and other PHS agencies have developed Parent
Announcements. Responding to such an omnibus or umbrella Parent FOA ensures that the correct
application package is used and enables NIH to receive the application from Grants.gov. Additional
information about, as well as links to published Parent Announcements, can be found at:
http://grants.nih.gov/grants/guide/parent_announcements.htm
.
Program Announcement (PA): A formal statement about a new or ongoing extramural activity or
program. It may serve as a reminder of continuing interest in a research area, describe modification in an
activity or program, and/or invite applications for grant support. Most applications in response to PAs
may be submitted to a standing submission date and are reviewed with all other applications received at
that time. NIH may also make funds available through PARs (Program Announcements with special
receipt, referral, and/or review considerations) and PASs (Program Announcements with set-aside funds).
Request for Applications (RFA): A formal statement that solicits grant or cooperative agreement
applications in a well-defined scientific area to accomplish specific program objectives. An RFA indicates
the estimated amount of funds set aside for the competition, the estimated number of awards to be made,
and the application submission date(s). Applications submitted in response to an RFA are usually
reviewed by a Scientific Review Group (SRG) specially convened by the awarding component that issued
the RFA.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-17
PAs (including Parent Announcements) and RFAs are published in the NIH Guide for Grants and
Contracts (http://grants.nih.gov/grants/guide) and on Grants.gov under Find Grant Opportunities
(http://www.grants.gov/applicants/find_grant_opportunities.jsp). Read the announcement carefully for
special instructions. The instructions in the announcement may differ from these general instructions, and
the instructions in the announcement always supersede these general instructions. Each announcement
published in the
NIH Guide for Grants and Contracts, Grants.gov Find, or other public document
contains contact information under Inquiries in addition to information specific to the announcement.
While individual announcements will continue to carry an announcement number reference to “PA” or
“RFA”, all announcements are “Funding Opportunity Announcements (FOAs).” This general term will be
used to reference any type of funding announcement. NIH will continue to use the PA and RFA
references in the actual announcement number to distinguish between the various types of
announcements.
In reading any FOA in the NIH Guide for Grants and Contracts:
• A “Posted Date” refers to the date the FOA is posted on Grants.gov
. An applicant can download
the application package on that date and begin filling it out. However, the applicant has to wait
until the FOA’s “open date” to submit the application.
• An application can be submitted anytime between the “open date” and the “application due
date(s)” noted for AIDS and non-AIDS applications. (Standard dates may apply; check
http://grants.nih.gov/grants/funding/submissionschedule.htm
for details.)
• When you download an application package from Grants.gov, the “expiration date” is pre-
populated. Do not go strictly by this date since it may not apply to your particular situation; for
instance, it may reflect the submission date for AIDS applications and you may be submitting a
non-AIDS application that is due earlier. In this case, the pre-populated date has no bearing on
your application and you should not be concerned by it.
All applications submitted to the NIH must be submitted in response to a FOA published in the NIH
Guide for Grants and Contracts.
2.4.3 Finding a Funding Opportunity Announcement (FOA) for
Grants.gov Submission
Implementation of the SF424 (R&R) application and electronic submission through Grants.gov will be
announced through specific FOAs posted in the NIH Guide for Grants and Contracts and on Grants.gov
under “Find Grants” (a.k.a. “Find”) and “Apply for Grants” (a.k.a. “Apply”). From the “For Applicants”
section of the Grants.gov home page, select “Apply for Grants” and follow the steps provided. FOAs
posted in Grants.gov Apply reflect those the agency is prepared to receive through electronic Grants.gov
submission. Applicants are encouraged to read each FOA carefully for specific guidance on the use of
Grants.gov submission.
There are several ways a prospective applicant can find a FOA on Grants.gov.
Using the NIH Guide for Grants and Contracts
FOAs in the NIH Guide for Grants and Contracts that reference electronic submission via Grants.gov
include a link from the FOA directly to the Grants.gov site where you can download the specific
application package. The Apply for Grants Electronically button is found in the NIH Guide FOA
directly above the Table of Contents. This link is only provided in those announcements involving
electronic submission through Grants.gov.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-18
Using “Find Grants” (Find) Feature
Grants.gov Find provides general search capabilities. From the “Search Grants” page, you will find
various options for: providing search criteria (e.g., keyword, Funding Opportunity Number, status, etc.)
and identifying FOAs of interest.
Once you find an opportunity for which you wish to apply, you may initiate the application download
process by selecting the “Application Package” tab.
Using “Apply for Grants” (Apply) Feature
If you know the specific funding opportunity number, a more direct route is to use the “Apply for Grants”
feature. From the Grants.gov home page, select “Apply for Grants” and follow the steps provided. “Step
1” allows you to download an application package by inserting a specific Funding Opportunity Number
(FOA). If you do not know the specific Funding Opportunity Number there is a link that will take you
back to the Search Grants page.
A Funding Opportunity Announcement (FOA) Number is referenced in every announcement. Enter this
number in the Funding Opportunity Number field and click Download Package. This takes you to a
“Selected Grant Applications for Download” screen.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-19
Click the corresponding download link to access the actual application form pages and instruction
material. If more than one application package is listed, use the Competition ID and Competition Title to
determine the appropriate package to download. The following screens appear:

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-20
To access the instructions, click Download Application Instructions. For NIH opportunities and other
PHS agencies using this Application Guide, this action will download a document containing a link to the
NIH website where the most current set of application instructions is available
(http://grants.nih.gov/grants/funding/424/index.htm
). Applicants are encouraged to check this site
regularly for the most current version.
To access the form pages, click Download Application Package. Section 2.5 provides specific
information regarding the forms for an Application Package. Section 3
provides additional instructions for
properly using a package.
On the Download Opportunity Instructions and Applications screen you will be given an opportunity to
provide an e-mail address if you would like to be notified of any changes to this particular opportunity.
Applicants to NIH and other PHS agencies are strongly encouraged to complete this information. The
agency can then use it to provide additional information to prospective applicants.
Note: The forms may or may not show a CFDA number and the CFDA field is not editable. Do not be
concerned. The CFDA number is not used for assignment of the application. Be assured the correct
CFDA number will be assigned to the record once the appropriate IC assignment has been made.
2.5 Forms for an Application to NIH or Other PHS Agencies
The SF424 (R&R) form set comprises a number of forms, each listed in the table below as a separate
“document.” In addition to these forms, NIH and other PHS agencies applicants will also complete
supplemental forms listed as “PHS 398” forms in the table below.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-21
Table 2.5-1. Table 2.5-1. Forms for an NIH or Other PHS Agencies Application
DOCUMENT
REQUIRED
OPTIONAL
INSTRUCTIONS
SF424 (R&R) Cover
x
Section 4.2
SF424 (R&R) Project/Performance Site Locations
x
Section 4.3
SF424 (R&R) Other Project Information
x
Section 4.4
SF424 (R&R) Senior / Key Person Profile(s)
x
Section 4.5
SF424 (R&R) Budget
(Use when required or allowed by the FOA)
*
Section 4.7
SF424 (R&R) Subaward Budget Attachment Form
(Use when required or allowed by the FOA)
x
Section 4.8
PHS 398 Cover Page Supplement
x
Section 5.3
PHS 398 Modular Budget
(Use when required or allowed by the FOA)
*
Section 5.4
PHS 398 Research Plan
(Not required for Career Development Award (K)
or
Institutional Training Award (T) applications)
x
Section 5.5
Planned Enrollment Report
x
Section 5.8
PHS 398 Cumulative Inclusion Enrollment Report
x
Section 5.8
PHS 398 Career Development Award Supplemental
Form
(Required only for Career Development Award (K)
applications)
x
Section 7.5
PHS 398 Research Training Program Plan
(Required only for Institutional Training Award (T)
applications)
x
Section 8.7
PHS 398 Training Budget
(Use when required or allowed by the FOA)
*
Section 8.5
PHS 398 Training Subaward Budget Attachment
(Use when required or allowed by the FOA)
*
Section 8.6
* The application forms package associated with most NIH research grant funding opportunities includes two
optional budget forms: (1) SF424 (R&R) Budget and (2) PHS 398 Modular Budget. NIH application submissions
must include either the SF424 (R&R) Budget Form or the PHS 398 Modular Budget Form, but never both. (Note
AHRQ does not accept modular budgets.) Unless otherwise stated in a funding announcement, an application
must always be submitted with a budget form. For those programs where either form is a possibility, the budget
forms will be considered “optional” by the Grants.gov package. Nonetheless, it is still required that you select and
submit one of these budget forms for an application to be accepted by the NIH.
To determine which budget form to use for NIH applications, consult the modular budget guidelines found in
Section 5.4
. Additional guidance may also be provided in the specific funding opportunity announcement.
Some funding opportunities will explicitly state the use of only one of the budget forms. In this case, the application
package will only include the accepted budget form which will appear in the list of “mandatory” forms (not in the
optional list).

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-22
For a small number of programs (e.g., S10, DP1, DP2, DP3, X01, X02), neither budget form is included and the
only budget information required is the Estimated Project Funding section of the SF424 (R&R) cover.
All required and optional forms for electronic submission listed above are available through Grants.gov and should
be downloaded from the FOA being applied to. Do not use any forms or format pages from other sources; these
may include extraneous headers/footers or other information that could interfere with the electronic application
process.
2.6 Format Specifications for Text (PDF) Attachments
Designed to maximize system-enforced validations, multiple separate attachments are required for a
complete application. When the application is received by the agency, all submitted forms and all separate
attachments are combined into a single document that is used by peer reviewers and agency staff.
NIH and other PHS agencies require all text attachments to the Adobe application forms be submitted as
PDFs and that all text attachments conform to the agency-specific formatting requirements noted below.
Failure to follow these requirements may lead to rejection of the application during agency validation or
delay in the review process. (See Section 2.3.2
for more information on creating PDFs.)
Text attachments should be generated using word processing software and then converted to PDF using
PDF generating software. Avoid scanning text attachments to convert to PDF since that causes problems
for the agency handling the application. Additional tips for creating PDF files can be found at
http://grants.nih.gov/grants/ElectronicReceipt/pdf_guidelines.htm
.
When attaching a PDF document to the actual forms, please note you are attaching an actual document,
not just pointing to the location of an externally stored document. Therefore, if you revise the document
after it has been attached, you must delete the previous attachment and then reattach the revised
document to the application form. Use the View Attachment button to determine if the correct version
has been attached.
File Name
Save all files with descriptive file names of 50 characters or less and be sure to only use standard
characters in file names: A through Z, a through z, 0 through 9, underscore (_), hyphen (-), space ( ), and
period (.). Do not use any other special characters (example: “&”, “*”, “%”, “/”, and “#”) or spacing in
the file name.
Font
Use an Arial, Helvetica, Palatino Linotype, or Georgia typeface, a black font color, and a font size of 11
points or larger. (A Symbol font may be used to insert Greek letters or special characters; the font size
requirement still applies.)
Type density, including characters and spaces, must be no more than 15 characters per inch.
Type may be no more than six lines per inch.
Paper Size and Page Margins
Use standard paper size (8 ½" x 11).
Use at least one-half inch margins (top, bottom, left, and right) for all pages. No information should
appear in the margins, including the PI’s name and page numbers.
Page Formatting
Since a number of reviewers will be reviewing applications as an electronic document and not a paper
version, applicants are strongly encouraged to use only a standard, single-column format for the text.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-23
Avoid using a two-column format since it can cause difficulties when reviewing the document
electronically.
Do not include any information in a header or footer of the attachments. A header will be system-
generated that references the name of the PD/PI. Page numbers for the footer will be system-generated in
the complete application, with all pages sequentially numbered.
Figures, Graphs, Diagrams, Charts, Tables, Figure Legends, and Footnotes
You may use a smaller type size but it must be in a black font color, readily legible, and follow the font
typeface requirement. Color can be used in figures; however, all text must be in a black font color, clear
and legible.
Grantsmanship
Use English and avoid jargon.
If terms are not universally known, spell out the term the first time it is used and note the appropriate
abbreviation in parentheses. The abbreviation may be used thereafter.
Page Limits
Although many of the sections of this application are separate text (PDF) attachments, page limits
referenced in these instructions and/or funding opportunity announcement must still be followed. Agency
validations will include many checks for page limits. Some accommodation will be made for sections that
when combined must fit within a specified limitation. Note that while these computer validations will
help minimize incomplete and/or non-compliant applications, they may not address all page limit
requirements for a specific FOA and do not replace the validations conducted by NIH staff. Applications
found not to comply with the requirements may lead to rejection of the application during agency
validation or delay in the review process.
All applications for NIH and other PHS agency funding must be self-contained within specified page
limits. Unless otherwise specified in an NIH solicitation, internet website addresses (URLs) may not be
used to provide information necessary to the review because reviewers are under no obligation to view the
internet sites. Moreover, reviewers are cautioned that they should not directly access an Internet site as it
could compromise their anonymity.
Observe the page number limits given in Table 2.6-1
. Only when specifically allowed in a FOA will the
PHS accept applications that exceed the page number limitations. However, specific page number limits
may apply to each subproject. For information pertaining to page number limits for such projects, contact
the awarding component to which the application may be assigned. (See
Table 1.4-1. Agency Contact
Table.) The page number limitations may also be different for other specialized grant applications.
Applicants are prohibited from using the Appendix to circumvent page limitations in any section of the
application for which a page limit applies. For additional information regarding Appendix material and
page limits, please refer to the NIH Guide Notice
NOT-OD-11-080.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-24
Table 2.6-1. Page Limits
Please visit http://grants.nih.gov/grants/forms_page_limits.htm for a more detailed Table of Page
Limits.
SECTION OF APPLICATION
Also refer to the relevant section of the application instructions and the FOA.
PAGE LIMITS *
Introduction to Resubmission Application
(3 pages for R25 on PHS 398 Research Plan and 3 pages for K12, T and D Training
Grants on PHS 398 Training Program Plan)
1 page
Introduction to Revision Application
1 page
Specific Aims
1 page
Research Strategy (Item of Research Plan)
6 pages
or 12 pages
or Follow FOA instructions
Research Education Program Plan
For R25 Research Education Grant Applications
25 pages
Plan for Instruction in the Responsible Conduct of Research
Required for all Training Grant Activity Codes
Required for all Individual Career Development Grant Activity Codes
3 pages
1 page
Biosketch (per person)
(2 pages for DP1 and DP2 Activity Codes)
4 pages
Career Development Award (K) Application
Upload to PHS 398 Career Development Award Supplemental Form: Combined
Candidate Information (Candidate’s Background, Career Goals and Candidate’s Plan
for Career Development/Training Activities During Award Period attachments) and
Research Strategy attachment
12 pages
Institutional Research Training and Career Development Applicants,
Including Ruth L. Kirschstein NRSA Application
Research Training Program Plan: Combined Sections 8.7.2.2 – 8.7.2.4 (Background,
Program Plan, and Recruitment and Retention Plan to Enhance Diversity)
25 pages
* FOA instructions always supersede these instructions.
2.7 “Resubmission” Applications
For all original new (i.e., never submitted) competing renewal applications submitted for the January 25,
2009 due date and beyond, NIH will accept only a single amendment (A1) to the original application
(called a resubmission application). A lengthy hiatus after the initial submission may be marked by
significant advances in the scientific field and the comments of the reviewers may no longer be relevant.
Therefore, a resubmission application must be submitted within 37 months after the date of receipt
("receipt date") of the initial New, Renewal, or Revision application (see NOT-OD-10-140 and
NOT-OD-

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-25
12-128). After 37 months, you may submit a New application. Any second resubmission will be
administratively withdrawn and not accepted for review.
NIH has established policies for application resubmissions of certain categories. See Resubmission of
Unpaid RFA Applications and Resubmission of Applications with a Changed Grant Activity Code in
Supplemental Instructions Part III, 1.2.
There are five requirements for a Resubmission application:
• The Summary Statement must be available in the eRA Commons
(http://commons.era.nih.gov/commons
).
• The PD/PI(s) must make significant changes to the application.
• An Introduction must be included that summarizes the substantial additions, deletions, and
changes to the application. The Introduction must also include a response to the issues and
criticism raised in the Summary Statement. The Introduction is separate from the Cover Letter.
Use “ Introduction” of the PHS 398 Research Plan Form to provide this information. The page
limit for the Introduction may not exceed one page unless indicated otherwise. Please refer to the
relevant section of the application instructions and the FOA.
• The substantial scientific changes must be marked in the text of the application by bracketing,
indenting, or change of typography. Do not underline or shade the changes. Deleted sections
should be described but not marked as deletions. If the changes are so extensive that essentially
all of the text would be marked, explain this in the Introduction. The Preliminary Studies/Progress
Report section should incorporate work completed since the prior version of the application was
submitted.
• For Mentored Career Development Award applications, new Letters of Reference must be
submitted providing an up-to-date evaluation of the applicant’s potential to become an
independent researcher, and the continued need for additional supervised research experience.
See NOT-OD-11-057
for special conditions and due dates for new investigator resubmission applications
submitted for consecutive review cycles. Note this applies only to new investigator R01s submitted for
standard receipt dates and reviewed in recurring study sections in CSR.
Acceptance of a resubmission application will not automatically withdraw the prior version. eRA keeps
all versions (e.g., 01, A1) of a grant application active and provides an internal Multiple Active
Applications (MAA) flag for each application in an active cluster. The cluster allows applicants to
identify quickly all versions of one application. If any version in a cluster is awarded, all other
applications within the cluster will be automatically withdrawn without any additional action by
applicants or staff.
Investigators who have submitted two versions of an application and have not been successful often ask
NIH what constitutes a “new application.” It is recognized that investigators are trained in a particular
field of science and are not likely to make drastic changes in their research interests. However, a new
application following two reviews is expected to be substantially different in content and scope with more
significant differences than are normally encountered in a Resubmission application. Simply rewording
the title and Specific Aims or incorporating minor changes in response to comments in the previous
Summary Statement does not constitute a substantial change in scope or content. Changes to the Research
Strategy should produce a significant change in direction and approach for the research project. Thus, a
new application would include substantial changes in all portions of the Specific Aims and Research
Strategy. Requests for review by a different review committee or funding consideration by a different
NIH IC are not sufficient reasons to consider an application as new.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-26
In the referral process, NIH staff look at all aspects of the application, not just the title and Description
(abstract). Requesting review by a different review committee does not affect the implementation of this
policy. When necessary, previous applications are analyzed for similarities to the present one. Thus,
identical applications or those with only minor changes will not be accepted for review. If identified after
assignment or review, identical applications will be withdrawn.
2.8 “Revision” Application
A Revision application (formerly called a competing supplement) may be submitted to request support for
a significant expansion of a project’s scope or research protocol. Applications for revisions are not
appropriate when the sole purpose is to restore awards to the full SRG-recommended level if they were
administratively reduced by the funding agency. A revision application should not be submitted until after
the original application has been awarded and must not extend beyond the term of the current award
period.
Provide a one-page “Introduction” that describes the nature of the supplement and how it will influence
the specific aims, research design, and methods of the current grant. Use Introduction to application, of
the PHS 398 Research Plan form to provide this information. The body of the application should contain
sufficient information from the original grant application to allow evaluation of the proposed supplement
in relation to the goals of the original application. Note that all revision applications must be submitted by
the same PD/PI (or Contact PD/PI for multi-PI grants) as listed on the current award and applicants must
use the same budget format (i.e. R&R Budget Form or PHS 398 Modular Budget Form) as the current
award. Also, any budgetary changes for the remainder of the project period of the current grant should be
discussed in the Budget Justification.
If the revision application relates to a specific line of investigation presented in the original application
that was not recommended for approval by the SRG, then the applicant must respond to the criticisms in
the prior Summary Statement, and substantial revisions must be clearly evident and summarized in the
“Introduction.”
Administrative Supplements
An administrative supplement provides additional funding to meet increased costs that are within the
scope of an approved application, but that were unforeseen when the new or competing renewal
application was submitted. If considering administrative supplement funding, you must consult in
advance with your designated Grants Management Officer and Program Official. It is important to submit
a request before the grant expires. NIH now publishes all administrative supplement programs as FOAs,
not Guide Notices as done previously. Administrative Supplement requests may now be submitted in
response to an FOA through either the eRA Commons or Grants.gov, as described in NOT-OD-12-024
,
or through the existing, paper-based process. If you submit a request in writing, you must submit to the
IC (not to the Division of Receipt and Referral, Center for Scientific Review). For additional
information, see NIH Guide Notice
NOT-OD-12-043. The request must be signed by the authorized
Business Official and describe the need for additional funding and the categorical costs. In the letter, point
out what will NOT be able to be accomplished if such a request is denied.
2.9 Similar, Essentially Identical, or Identical Applications
Submissions of identical applications to one or more components of the PHS are not allowed.
Submissions of identical applications to one or more components of the PHS are not allowed, and the
NIH will not accept similar grant applications with essentially the same research focus from the same
applicant organization. This includes derivative or multiple applications that propose to develop a single

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-27
product, process or service that, with non-substantive modifications, can be applied to a variety of
purposes. Likewise, identical or essentially identical grant applications submitted by different applicant
organizations will not be accepted. Applicant organizations should ascertain and assure that the materials
they are submitting on behalf of the principal investigator are the original work of the principal
investigator and have not been used elsewhere in the preparation and submission of a similar grant
application. Applications to the NIH are grouped by scientific discipline for review by individual
Scientific Review Groups and not by disease or disease state. The reviewers can thus easily identify
multiple grant applications for essentially the same project. In these cases, application processing may be
delayed or the application(s) may not be reviewed.
Essentially identical applications will not be reviewed except for: 1) individuals submitting an application
for an Independent Scientist Award (K02) proposing essentially identical research in an application for an
individual research project; 2) individuals submitting an individual research project identical to a
subproject that is part of a program project or center grant application; 3) submissions of applications
previously submitted to an RFA that were not paid or resubmissions of investigator-initiated applications
originally submitted to an RFA (see Grants Policy Statement section 2.3.7.3); and 4) resubmissions of
applications with a changed grant activity code.
2.10 Submitting Your Application Via Grants.gov
The Authorized Organization Representatives (AORs) registered in Grants.gov are the only official with
the authority to actually submit applications through Grants.gov. Therefore, PD/PIs will need to work
closely with their AOR(s) to determine that all the necessary steps have been accomplished prior to
submitting an application. This includes any internal review process required by the applicant
organization.
Before starting the final submission step, applicants are encouraged to save a copy of the final
application locally. Once you have properly completed all required documents and attached any required
or optional documentation, click on the Check Package for Errors button to ensure that you have
successfully completed all Grants.gov required data fields. If any of the required fields are not completed
you will receive an error notice which will indicate where revision is needed within your package. Correct
any errors or if none are found, save the application package. Note that the Check Package for Errors
button only checks a subset of Grants.gov application checks and does not guarantee that your application
will pass subsequent Grants.gov and agency business rule checks done after submission. Only after the
package has been saved with no errors will the Save & Submit button become active. Clicking the Save
& Submit button will begin the application submission process. The application package must then be
saved once more before the submission process begins. Only an AOR will be able to perform the submit
action, and they will be taken to the applicant login page to enter the Grants.gov username and password
that was established in the Register with Grants.gov process (if not connected to the internet you will be
instructed to do so).
Once logged in, the application package will be automatically uploaded to Grants.gov. A confirmation
screen will appear once the upload is complete and a Grants.gov Tracking Number will be provided on
this screen. Applicants should record this number so that they may refer to it should they need to contact
Grants.gov Customer Support or the eRA Commons Help Desk.
For additional information, see http://www.grants.gov/applicants/apply_for_grants.jsp
.
Applicants should be aware that on-time submission means that an application is submitted error free (of
both Grants.gov and eRA Commons errors) before 5 p.m. on the receipt date, local time of the applicant
organization. Applicants are encouraged to submit their applications to Grants.gov several days early to
ensure enough time to correct any errors before the deadline.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-28
2.11 After You Submit Your Application Via Grants.gov
Grants.gov’s Track My Application feature for Applicants provides basic status information for a
specified Grants.gov Tracking Number. The Authorized Organization Representative (AOR) can also
login to Grants.gov with their username and password to obtain a more detailed status of an application
by using the Check My Application Status feature.
Once an application has been submitted via Grants.gov, several e-mails are sent to the email address on
file with Grants.gov for the AOR (known at NIH/in eRA Commons as the Signing Official [SO]) who
submitted the application. Each email notification will reference a Grants.gov tracking number that is
assigned to the submission:
1) Submission Receipt: An e-mail is sent indicating your application has been received by Grants.gov
and is currently being validated.
2) Submission Validation Receipt: An e-mail is sent indicating your application has been received and
validated by Grants.gov and is being prepared for Grantor agency retrieval.
3) Grantor Agency Retrieval Receipt: An e-mail is sent indicating your application has been retrieved
by the Grantor agency.
4) Agency Tracking Number Assignment for Application: An e-mail is sent indicating your application
has been assigned an Agency Tracking Number.
If the AOR/SO has not received a confirmation message from Grants.gov within 48 hours of submission,
please contact:
Grants.gov Contact Center
Telephone: 1-800-518-4726
At that point, the application will be scheduled for download into the eRA system for agency validation.
It is imperative that the e-mail address provided for the PD/PI and for the AOR/SO on the SF424 (R&R)
Cover form be current and accurate. Once agency validation is completed, an agency notification (not
Grants.gov) will be e-mailed to the PD/PI, AOR/SO, and the Applicant Contact e-mail (if provided)
named in the application.
This e-mail notification will inform the PD/PI, AOR/SO, and the Applicant Contact (if named) that the
application has been received and processed by the agency and will indicate whether any errors or
warnings resulted during the validation process. The PD/PI, AOR/SO, and the Applicant Contact will be
invited to log on the eRA Commons
to view the assembled application or review the list of
warnings/errors that were encountered during the validation process. SOs for the applicant organization,
the PD/PI for the application and any Assistants that have been delegated in eRA Commons to view the
PD/PIs status information will have access to the submission results in eRA Commons.
If there were no validation errors, this e-mail notification will also inform the PD/PI, AOR/SO, and the
Applicant Contact of an agency accession number, which represents the “agency tracking number.” This
number replaces the Grants.gov tracking number that was assigned when the application was first
submitted. The Grants.gov system will indicate that the agency tracking number has been assigned, and
will reflect both numbers.
The eRA system will make every effort to send an e-mail to the PD/PI, AOR/SO, and the Applicant
Contact summarizing download and validation results. However, since e-mail can be unreliable,
applicants are responsible for checking on their application status in the Commons.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-29
Once an application package has been successfully submitted through Grants.gov, any encountered errors
have been corrected by the applicant, and an application image has been assembled by the eRA Commons,
PD/PIs and AORs/SOs will have two business days (Monday – Friday, excluding Federal holidays) to check
the assembled application before it automatically moves forward to NIH staff for further processing and
consideration. Once the application has moved forward, no additional changes to the application will be
accepted through Grants.gov or eRA Commons. This window is known as the application viewing window.
Within the viewing window, SOs have the authority to reject an application to stop it from completing the
submission process. After an application is rejected, a changed/corrected application can be submitted to
address warnings or other issues if it is still before the submission deadline. However, changed/corrected
applications submitted after the submission deadline will be subject to the NIH Late Policy and may not be
accepted.
Remember, warnings do not stop further application processing. If an application submission results in
warnings (but not errors), the application will automatically move forward after the application viewing
window if no action is taken. Some warnings may need to be addressed later in the process.
It is your responsibility to view the entire assembled application in eRA Commons and notify the eRA
Commons Help Desk within this window if the assembled application does not correctly reflect the
information submitted to Grants.gov (e.g., submitted information is missing in image, graph/chart appears
upside-down). The eRA Commons Help Desk will provide guidance on appropriate corrective actions.
If you encounter a system issue beyond your control that threatens your ability to complete the submission
process on-time, you must follow the Guidelines for Applicants Experiencing System Issues.
Only the eRA Help Desk can confirm whether a system issue has taken place and provide instructions on
how to resolve the issue. Applications affected by confirmed system issues will not be considered late as
long as the applicant works diligently with the eRA Help Desk on a resolution. If the application needs to be
Rejected and resubmitted, applicants should follow the instructions for correcting errors in Section 2.12,
including the requirement for cover letters on late applications.
PIs should work with their AOR/SO to determine when the “Reject” feature is appropriate.
To view the assembled application the AOR/SO should:
1. Login to the eRA Commons (https://commons.era.nih.gov/commons/
) with your Signing
Official (SO) account.
2. Click the Status tab on the Commons menu bar.
3. Click Recent/Pending eSubmissions on the left-hand side of the screen.
4. Search for your application by date received, Grants.gov tracking number, or accession number,
to view a hit list of available applications.
5. When you find the appropriate application, click the accession number in the Application ID
column to view the Status Information screen.
6. Click e-Application from the Other Relevant Documents section to view the assembled
application.
Note: The SO can Reject the application by clicking on the Reject eApplication hypertext link
from the Action column of the search hit list.
To view the assembled application the PD/PI should:

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-30
1. Login to the eRA Commons (https://commons.era.nih.gov/commons/) with your Principal
Investigator (PI) account.
2. Click the Status tab on the Commons menu bar.
3. Click Recent/Pending eSubmissions near the top of the screen to view a hit list of available
applications.
4. When you find the appropriate application, click the accession number in the Application ID
column to view the status information screen.
5. Click e-Application from the Other Relevant Documents section to view the assembled
application.
2.12 Correcting Errors
Prior to a specified application due date, applicants may make corrections and resubmit an application
through Grants.gov. If applicants make corrections and resubmit the application after the due date, the
application will be considered late. In this case, applicants must include a cover letter explaining the
reasons for the delay and acceptance will be considered on a case-by-case basis. Also see Section 2.14
for
additional information on submission dates.
If errors or warnings result from the validation process, the PD/PI, AOR/SO and Applicant Contact will
be issued an e-mail instructing them to log on to the eRA Commons to review the list of warnings/errors
that were encountered during the validation process. The eRA system will make every effort to send an e-
mail to the PD/PI, AOR/SO and Applicant Contact indicating whether errors or warnings were detected.
However, since e-mail can be unreliable, applicants are responsible for periodically checking on their
application status in the eRA Commons, so that any errors or warnings can be resolved in the timeliest
manner possible, before the deadline. SOs for the applicant organization, the PD/PI for the application
and any Assistants that have been delegated in eRA Commons to view the PD/PIs status information will
have access to the submission results in eRA Commons.
Please be aware of the distinction between errors and warnings. The word error is used to characterize
any condition which causes the application to be deemed unacceptable for further consideration.
Generally, errors will indicate significant inaccuracies, inconsistencies, omissions, or incorrect formatting
that have been identified in the body of the application. Conversely, the word warning characterizes any
condition that is acceptable, at least for the time being, but worthy of bringing to the applicant’s attention.
It is at the applicant’s discretion whether a warning condition requires any action, but some warnings may
need to be addressed later in the process.
Failure to comply with stated NIH policies can also result in a submitted application being returned to the
applicant without being reviewed. For this reason, applicants are strongly encouraged to review all
warnings, to ensure that they require no further attention and that they are satisfied with the validation
results. If desired, warnings can be corrected in the same manner as errors.
A Changed/Corrected application may also be submitted if the PDF image, as viewed in the eRA
Commons, is incomplete or inaccurate from that submitted.
Submitting a Changed/Corrected application will overwrite the previous submission and the previous
application cannot be reinstated.
Errors and warnings may be reviewed in the Commons by performing the following steps:
1. After the application has been downloaded from Grants.gov and validated by the system, login
to the eRA Commons (https://commons.era.nih.gov/commons/
) using your username and
password.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-31
2. Click the Status tab on the Commons menu bar.
3. Click Recent/Pending eSubmissions.
4. Search for your application by date received, Grants.gov tracking number, or accession number,
if you are the SO. If you are the PI, clicking Recent/Pending eSubmissions will automatically
display a hit list of your applications.
5. A hit list of applications is displayed. If the application was validated with warnings only, or
without encountering any problems whatsoever, then it is identified in the hit list by its NIH
accession number (e.g., “AN:2911064”). This is the same number that Grants.gov displays, and
refers to as the “agency tracking number.”
If any errors were identified during validation, then the application still appears in the hit list,
but in this case it is identified by its Grants.gov tracking number (e.g., “GRANT87654321”).
This is the number that Grants.gov assigned to your application at the time of submission.
6. When you find the appropriate application in the hit list (Application Status will read
“eSubmission Error” if errors were received), click its Application ID link to view the list of
errors/warnings.
7. The error/warning page appears, and you are then able to review all conditions that were
identified during validation. If only warnings were identified, you may elect to take action and
resubmit; however you may also disregard the warnings and proceed to view the application, as
described earlier.
To correct errors and resubmit the application:
1. Make whatever corrections are necessary, wherever appropriate, to your local copy of the
application. Most often this means that you have to edit the data within the application forms to
correct whatever problem or inconsistency that was noted. Be as careful as possible when
correcting your application; NIH’s post-submission materials policy does not allow for
applicants to correct oversights in their application after the due date.
2. Check the “Changed/Corrected Application” box in the Type of Submission field of the SF424
(R&R) Cover form.
o If you are submitting this Changed/Corrected application after the due date, be sure to
document the reason for the late submission in the form of a cover letter. NIH makes no
guarantees that applications submitted after the due date will be accepted. See the NIH
late policy for more information.
o When you check the Changed/Corrected Application box, Previous Grants.gov Tracking
ID field becomes required.
o When you have made all of your corrections, save the Changed/Corrected application to
your computer.
3. The AOR will have to submit the Changed/Corrected application package to Grants.gov. A new
Grants.gov tracking number will be assigned and the applicant will have to follow the
Changed/Corrected application through Grants.gov to the eRA Commons to view the application
image or the list of errors/warnings received during the validation process. It is the applicant’s
responsibility to track the application through to the eRA Commons. If you cannot view your
application image in the Commons, NIH can’t review your application! Successful submission
may take several rounds of Changed/Corrected applications, since correcting one error may
reveal or create an additional error.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-32
The same e-mail notifications will be issued once the agency has downloaded and validated the re-
submitted application and the PD/PI, AOR/SO, and Applicant Contact will once again be required to log
on to the Commons either to view the application or to review the errors that were encountered during
validation.
The application will only be assigned for scientific review once errors are resolved.
In addition to the validations performed by the eRA system, further administrative review will be
conducted by agency staff. The PD/PI and/or the applicant organization may be contacted for further
corrections/clarifications.
2.13 Post-Submission Application Materials
Grant application materials will only be accepted after submission of the application but before the initial
peer review if they result from unforeseen administrative issues. Exceptions to this policy are indicated
below. See NOT-OD-10-091 and NOT-OD-13-030
for additional information.
The materials should be sent as a PDF attachment to an e-mail. E-mail communication is preferred. If e-
mail is not feasible, please send in a hard copy.
The original application is kept intact; any application material sent post-submission is sent separately to
reviewers. Updated or supplemental grant application materials used in the peer review process will be
retained as part of the official grant file and remain part of the permanent record for that application.
Acceptable post-submission materials include:
• Revised budget page(s) (e.g., change in budget request due to new funding or institutional
acquisition)
• Biographical sketches (e.g., change in senior/key personnel due to the loss of an investigator)
• Letters of support or collaboration resulting from a change in senior/key personnel due to the loss
of an investigator
• Adjustments resulting from natural disasters (e.g., loss of an animal colony)
• Adjustments resulting from change of institution (e.g., PI moved to another university)
• News of an article accepted for publication
• News of a professional promotion or positive tenure decision for any Program Directors/Principal
Investigators and Senior/Key Personnel also will be accepted as post-submission application
material. The news must be received by the Scientific Review Officer one month (30 calendar
days) prior to the peer review meeting, and demonstrate concurrence from the Authorized
Organization Representative (AOR) of the applicant organization.
• If an application proposed to use a human embryonic cell line(s) that is approved by the NIH
Stem Cell Registry at least 30 days prior to the peer review meeting, the SRO can include this
update to the reviewers as part of the post-submission materials (see NOT-OD-12-111
).
• Videos, within defined limits, (see, NOT-OD-12-141). Devices and other non-traditional
materials will not be accepted after submission, unless the application is submitted for a Funding
Opportunity Announcement that specifies other types of allowable post-submission materials.
• For institutional training and training-related grants [not fellowships (F series) or career (K
series)] applications, the following types of information will be accepted:

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-33
o News of a trainee's or former trainee's graduation, employment, promotion, funding,
publication or other professional achievement since the training grant was submitted;
o News of a faculty member's promotion, funding, publication, or other professional
achievement since the training grant was submitted; and
o News of an additional faculty member who will be involved in the training activity.
Unacceptable post-submission materials [for all applications but those under Exceptions below]
include:
• Updated Specific Aims or Research Strategy pages
• Late-breaking research findings
• Supplemental pages - information not contained in the existing application
• New letters of support or collaboration that do not result from a change in senior/key personnel
due to the loss of an investigator
Exceptions to this policy include:
• Applications submitted in response to Requests for Applications (RFAs) that have only one due
date. Post-submission materials for these applications will be accepted as outlined in
NOT-OD-
10-070
• Applications submitted in response to Requests for Applications (RFAs) that have multiple due
dates, the exception to this policy applies to applications submitted for the last due date of an
RFA (see NOT-OD-13-030
)
• Applications for training grants (see NOT-OD-10-104)
• Certain NIH Funding Opportunity Announcements (FOAs) may allow certain other types of post-
submission materials to facilitate the goals of the program. Such stipulations must be explained in
the FOA in the NIH Guide for Grants and Contracts
Page limits for post-submission materials under the new policy:
• All post-submission materials must conform to NIH policy on font size, margins, and paper size
as referenced in Part I.2.6 of the applicable application instructions
• NIH additional form pages such as budget, biographical sketches, and other required forms must
follow NIH standards for required NIH form pages.
• If post-submission material is not required on a form page, each explanation or letter is limited to
one page (see Acceptable post-submission materials above)
• If the application has subprojects or cores, each subproject or core is allowed explanations or
letters (see Acceptable post-submission materials above), but each explanation or letter is limited
to one page
The additional materials must be submitted to the NIH SRO with the concurrence of the applicant
organization’s designated AOR/SO. Although the content of post-submission materials may originate
from the PD/PI, Contact PD/PI for multiple PD/PI applications, or organizational officials, the AOR must
send the materials directly to the SRO, or must send his/her concurrence to the PD/PI who will forward
the materials and concurrence to the SRO. A communication from the PD/PI only or with a “cc” to the
AOR will not be accepted.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-34
The deadline for receipt of additional materials is one month (30 calendar days) prior to the peer review
meeting.
After the initial peer review phase is completed, the NIH Chief Grants Management Officer
is the NIH
official responsible for accepting additional materials. Most of the materials submitted after the initial
peer review can be submitted as part of the
Just-In-Time process (see Supplemental Instructions Part
III.1.7).
2.14 Application Submission Dates
For submission of applications to NIH, each FOA includes an Opportunity Open Date and an Opportunity
Close Date. Many announcements, including those using the “Standard Submission Dates, include
multiple submission/receipt dates and are active for several years. These announcements are posted in
Grants.gov and the NIH Guide to Grants and Contracts showing an Open/Close period that spans the
entire active period of the announcement. Applicants should read the Funding Opportunity
Announcement carefully for specific submission/receipt dates. If specific dates are not referenced in the
announcement, applicants should refer to the Standard Submission Dates for Competing Applications at
this website: http://grants.nih.gov/grants/funding/submissionschedule.htm
Applications submitted for the Standard Submission Dates listed are considered on time if they are
submitted to Grants.gov on or before 5 p.m. local time on the appropriate date listed. Applications
submitted to FOAs with a single submission date are considered on time if they are submitted to
Grants.gov on or before 5 p.m. local time on the appropriate date listed. Applications submitted for
Special Receipt Dates are considered on time if they are submitted to Grants.gov on or before 5 p.m. local
time on the specified Application Due Date(s). Requests for Applications (RFAs) and Program
Announcements with Special Referral Considerations (PARs) with special receipt dates always must be
received by Grants.gov on the dates designated in the announcement.
Weekend/Federal Holiday Submission Dates. When a postmark/submission date falls on a weekend or
Federal holiday, the application deadline is automatically extended to the next business day. If the
submission deadline shifts beyond the expiration date of the FOA, applicants submitting electronically
may receive a warning message from Grants.gov. This warning will not keep your application from
processing through Grants.gov. As always, applicants must check the eRA Commons to view their
application and to ensure they have addressed any errors (instances of non-compliance with NIH business
rules) identified by NIH systems.
Modified Application Submission and Review Policy. A continuous submission process is available to
appointed members of chartered standing NIH Study Sections, Boards of Scientific Counselors, Advisory
Boards or Councils, Program Advisory Committees, and peer reviewers who have served as regular or
temporary members of peer review committees six times in 18 months. This alternative submission and
review process is limited to R01, R21, and R34 application that would normally be received on standard
submission dates. See the Peer Review Policies & Practices, Continuous Submission
web page for
additional information on the continuous submission process and eligibility requirements.
Late Applications. Permission for a late submission is not granted in advance. In rare cases, late
applications will be accepted but only when accompanied by a cover letter that details the compelling
reasons for the delay. While the reasons are sometimes personal in nature, an objective evaluation of their
merit requires that some details be provided. Late applications have been accepted for reasons such as:
death of an immediate family member of the PD/PI, sudden acute severe illness of the PD/PI or
immediate family member, or large scale natural disaster.
NIH will consider accepting late applications based on the acceptability of the explanation and the
processing time required for two different kinds of submission/receipt dates.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-35
• Regular Standard Submission/Receipt Dates: To be considered applications must be received
at the NIH within two weeks of the standard submission date.
• Expedited Standard Submission/Receipt Dates: To be considered applications must be
received at the NIH within one week of the standard submission date.
• NIH will not consider late applications for the Special Receipt Dates for RFAs and PARs. This
includes the special receipt dates (March 20, July 20, and November 20) for resubmission
applications that are part of the New Investigator Initiative
(http:grants.nih.gov/grants/guide/notice-files/NOT-OD-07-083.html
).
• NIH does not expect to accept any applications received beyond the window of consideration.
The windows of time for consideration of late applications have been carefully chosen so that the late
applications can be processed with the cohort of on-time applications. In all cases, when the regular
standard submission date or expedited submission date falls on a weekend or federal holiday and is
extended to the next business day, the window of consideration for late applications will be calculated
from that business day. Note that the late window always ends in a receipt (not submission) date.
If an application is submitted late, attach a Cover Letter to the SF424 (R&R) cover form using the Cover
Letter Attachment field to provide specific information on the timing and nature of the cause of the delay.
No other documentation is expected. Late applications are evaluated on an individual basis considering
the reasons provided. Contacting the
Division of Receipt and Referral, Center for Scientific Review
(CSR), NIH in advance will not influence the acceptance of a late application.
Related Guide Notices include:
• NOT-OD-12-001 Update of NIH Late Application Policy to Reflect Change in Due Dates for
New Investigator R01 Resubmission Applications at http://grants.nih.gov/grants/guide/notice-
files/NOT-OD-12-001.html;
• NOT-OD-11-035 NIH Policy on Late Submission of Grant Applications at
http://grants.nih.gov/grants/guide/notice-files/NOT-OD-11-035.html
; and
• NOT-OD-11-093 Change in the NIH Continuous Submission Policy for Reviewers with Recent
Substantial Service at http://grants.nih.gov/grants/guide/notice-files/NOT-OD-11-093.html
.
2.15 Submission, Review and Award Cycles
The PHS submission, review, and award schedule is provided at this website:
http://grants.nih.gov/grants/dates.htm
. For specialized grant applications, consult with the appropriate
PHS agency prior to the preparation of an application.
The SF424 (R&R) should only be used for those activity codes where the Application Form is identified
as the SF424 (R&R). Applicants should refer to the OER Applying Electronically website:
http://grants.nih.gov/grants/ElectronicReceipt/
for details on the transition to electronic submission.
Application Assignment Information
Competing grant applications that have been successfully submitted through Grants.gov (including
correcting all errors and the grant application assembled by the eRA Commons system) will be processed
through the Division of Receipt and Referral, CSR, NIH unless otherwise stated. The application will be
assigned to an appropriate Scientific Review Group and awarding component(s). Assignment is based on
the scientific content of the application using established referral guidelines. Business rule validations are
conducted by the system as well as NIH staff.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-36
Assignment to Review Group. The Center for Scientific Review (CSR) will assign appropriately
completed applications to the Scientific Review Groups (commonly referred to as “SRGs” or “study
sections”) that will perform the scientific/technical merit review. The CSR lists the recurring review
panels (
http://cms.csr.nih.gov/PeerReviewMeetings/CSRIRGDescription/), and you may suggest a
specific group in the PHS Cover Letter form. See Part I, Section 5.2 of this Guide for a suggested format
for requesting a specific SRG in the Cover Letter.
Assignment to Relevant Potential Awarding Component(s) (ICs). In addition, CSR will assign each
application to the agency awarding component that is the potential funding component. When the
scientific areas and the research proposed in a grant application are sufficiently relevant to the program
responsibilities of two or more awarding components, CSR may assign your application to all such
components. The component that has the most relevant program responsibility is designated as the
primary assignee. The other components that have an interest in your application are designated as
secondary assignees. If your application is eligible for funding and the primary assignee does not intend
to make an award, the secondary assignees will be given the opportunity to do so. Although these
suggestions will be taken into consideration, the final determination will be made by the agencies
participating in this solicitation.
After the submission date, usually within two (2) weeks, the PD/PI and the applicant organization will be
able to access in the eRA Commons and view the following information regarding the grant application:
• Application assignment number;
• Name, address, and telephone number of the Scientific Review Officer (if the review takes place
in CSR) of the Scientific Review Group to which the application has been assigned for peer
review; and
• Assigned Institute/Center information.
Review outcome and other important information are also available in the Commons.
If assignment information is not available in the eRA Commons within two weeks of the submission date,
contact the Division of Receipt and Referral, Center for Scientific Review (CSR), National Institutes of Health,
Bethesda, MD 20892-7720, (301) 435-0715; TTY (301) 451-5936. If there is a change in assignment, you will
receive a notification and the change will be reflected in the eRA Commons.
Applicant investigators must not communicate directly with any review group member about an
application either before or after the review. Failure to strictly observe this policy will create serious
breaches of confidentiality and conflicts of interest in the peer review process. From the time of
assignment to the time the review of your application is complete, applicant investigators must direct all
questions to the Scientific Review Officer. This individual is in charge of the review group and is
identified in the eRA Commons.
2.16 Resources for Finding Help
2.16.1 Finding Help for Grants.gov Registration or Submissions
If help is needed with the Grants.gov registration process or with the technical aspects of submitting an
application through the Grants.gov system, check first the resources available at Grants.gov
(http://grants.gov/
).

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-37
Grants.gov customer support is also provided by the following office:
Grants.gov Program Management Office
200 Independence Avenue, SW
HHH Building, Room 739F
Washington, DC 20201
Grants.gov Self-Service Web Portal: https://grants-portal.psc.gov/Welcome.aspx?pt=Grants
Grants.gov Help Desk: support@grants.gov
Grants.gov Contact Center Phone Number: 800-518-4726 (Toll Free)
606-545-5035 (Local or International)
The Contact Center is available 24 hours a day, 7 days a week (except Federal holidays).
2.16.2 Finding Help for the eRA Commons Registration or
eRA Commons Validation Processes
If help is needed with the eRA Commons registration process for the applicant organization and PD/PIs;
with using ASSIST for the preparation and submission of multi-project applications; or with the
application validation process in the Commons after submission through Grants.gov, check first the
resources available at the Applying Electronically website:
(http://grants.nih.gov/grants/ElectronicReceipt/
).
eRA Commons customer support is also provided by the eRA Commons Help Desk:
eRA Website: http://era.nih.gov
eRA Commons Website: https://commons.era.nih.gov/commons/index.jsp
eRA Commons On-line Resources and Web Ticketing: http://era.nih.gov/help/
eRA Commons Phone: 301-402-7469
866-504-9552 (Toll Free)
301-451-5939 (TTY)
The eRA Commons Help Desk hours of operation are Monday-Friday from 7:00 a.m. to 8:00 p.m.
Eastern Time (except Federal holidays).
NOTE: To help expedite your Help Desk request, we recommend that you have the following information
readily available (NOTE: Additional details may be required depending upon the type of issue/request):
• Full Name of Affected User
• Full Name of Institution/Organization
• Grants.gov Tracking Number
• Submission Date
• Funding Opportunity Announcement (FOA)
• Principal Investigator’s (PI) Username
• Signing Official’s (SO) Username

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-38
2.16.3 Finding Help for Application Preparation
If after reviewing this application instruction guide, help is still needed in preparing the application,
contact GrantsInfo:
GrantsInfo Phone: 301-435-0714
301-451-5936 (TTY)
GrantsInfo E-mail: [email protected]
3. Using the Grant Application Package
This section describes the steps an applicant takes once the appropriate FOA (see Section 2.4) has been
located and the corresponding grant application package has been successfully downloaded.
3.1 Verify Grant Information
When you select a funding opportunity in Grants.gov Apply, verify that the information shown in the
Grant Application Package screen corresponds to the funding opportunity for which you wish to apply.
Grants.gov auto-populates the following information:
• Opportunity Title
• Offering Agency
• CFDA Number
• CFDA Description
• Opportunity Number
• Competition ID
• Opportunity Open Date
• Opportunity Close Date
• Agency Contact

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-39
CFDA Number Field: Many FOAs include multiple CFDA (Catalog for Domestic Assistance) numbers.
When this is the case, the CFDA Number and CFDA Description fields will appear blank in the
Grants.gov Grant Application Package screen shown above. The appropriate CFDA number will be
automatically assigned once the application is assigned to the appropriate agency awarding component.
Opportunity Open Date & Close Date Fields: Many FOAs posted by NIH and other PHS agencies
include multiple submission/receipt dates and are active for several years. These announcements are
posted in Grants.gov showing an Open/Close period that spans the entire active period of the
announcement. Applicants should read the funding opportunity announcement carefully for specific
submission/receipt dates. If specific dates are not referenced in the announcement, applicants should refer
to the PHS submission, review, and award schedule at
http://grants.nih.gov/grants/dates.htm. Applications
submitted after a posted submission date will normally not be held over into the next review cycle.
Instead, the PD/PI will be notified and will have to submit the application again. See
Part I, Section 2.14
of this Guide for more information on the late application policy.
3.2 Enter the Name for the Application
Enter a name for the application in the Application Filing Name field (this is a required field). This name
is for use solely by the applicant for tracking the application through the Grants.gov submission process.
It is not used by the receiving agency.
3.3 Open and Complete Mandatory Documents
Click the form name to navigate to the form in the application package and complete all of the Mandatory
forms. Complete the form titled SF424 (R&R) first. Data entered in this form populates other
mandatory and optional forms where applicable.
3.4 Open and Complete Optional Documents
These documents can be used to provide additional information for the application or may be required for
specific types of grant activities. Information on each of these documents is found later in these

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-40
instructions. In some application packages applicants will see two budget options; e.g., Research &
Related Budget and PHS 398 Modular Budget in the Optional Documents section. While both appear as
Optional Documents, one or the other will always be required. See
Section 4.6 Selecting the Appropriate
Budget Form for additional details.
Click the check box next to the form name to add the form to the application package. To navigate to the
form, click on the form name. To remove an optional form from the application package, uncheck the box
next to the form name.
3.5 Submitting the Application via Grants.gov
Once you have properly completed all required documents and attached any required or optional
documentation, click on the Check Package for Errors button to ensure that you have successfully
completed all required data fields. If any of the fields required by Grants.gov are not completed you will
receive an error notice which will indicate where revision is needed within your package. Correct any
errors or if none are found, save the application package. The Save & Submit button will now become
active and clicking this button will begin the application submission process. Only after the package has
been saved with no errors will the Save & Submit button become active. The application package must
then be saved once more before the submission process begins. Only an AOR will be able to perform the
submit action, and they will be taken to the applicant login page to enter the Grants.gov username and
password that was established in the Grants.gov registration process (if not connected to the internet you
will be instructed to do so).
4. Completing the SF424 Research and Related
(R&R) Forms
4.1 Overview
This section contains all of the instructions you will need to complete the SF424 (R&R) forms.
Any agency-specific instructions on SF424 (R&R) forms used federal-wide are denoted by the
DHHS logo displayed to the left of the paragraph, as illustrated here.
Conformance to all instructions is required and strictly enforced. Agencies may withdraw any
applications from the review process that are not consistent with these instructions.
As you navigate through the Grants.gov forms, required fields are highlighted and outlined in red.
Optional fields and completed fields are displayed in white. Data entered into a specific field is not
accepted until you have navigated to the next field. If you enter invalid or incomplete information in a
field, you will receive an error message.
Note the highlighted fields required for submissions, and the Check Package for Errors button,
only refer to Grants.gov requirements and errors. They do not reflect all agency requirements or

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-41
PHS business processes. Agency validations will be performed by the eRA Commons system
after the application has been submitted.
For those forms that are more than one page, click the Next button at the top of the form or scroll down
(using the scroll bar on the right hand side of the screen) to navigate to a subsequent page. Once all data
have been entered scroll up using the scroll bar to return to the Grant Application Package Screen.
4.2 Cover Form

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-42
1. Type of Submission
Check one of the Type of Submission boxes. If this submission is to change or correct a previously
submitted "New", "Resubmission", "Renewal", or "Revision" application, click the Changed/Corrected
Application box and enter the Grants.gov tracking number in the Previous Grants.gov Tracking ID field.
Unless requested by the agency, applicants may not use this to submit changes after the closing date. This
field is required.
Pre-Application: Unless specifically noted in a program announcement, the Pre-application
option is not used by NIH and other PHS agencies.
Changed/Corrected Application: This box must be used if you need to submit the same
application again to correct system validation errors, application assembly problems, or to
incorporate other changes. When submitting a Changed/Corrected Application:
• If submitting after the submission date, include an explanation in the Cover Letter Form.
• Submitting a Changed/Corrected application replaces the previous submission and removes
the previous submission from consideration. Once an application has moved forward to
agency staff following the two-day application viewing window
, subsequent
Changed/Corrected applications will not be accepted unless the application is withdrawn.
Note that if you are submitting additional grant application materials after the submission
date some special guidelines may apply. See NIH Guide Notice NOT-OD-10-115
(
http://grants.nih.gov/grants/guide/notice-files/NOT-OD-10-115.html) for the NIH Policy on
Post-Submission Application Materials.
• When you check the Changed/Correct Application box the Previous Grants.gov Tracking ID
becomes a required field.
• Do not use the Changed/Corrected Application box to denote a submission of a resubmission
or amended application. That will be indicated in the Type of Application.
2. Date Submitted and Applicant Identifier
Enter the date the application is submitted to Federal agency (or State if applicable). In the applicant
identifier field enter the applicant’s control number (if applicable).
Note the Applicant Identifier field is a control number created by the applicant organization, not
the Federal agency.
3. Date Received by State and State Application Identifier
Enter the date received by state (if applicable). In the State Application Identifier field, enter the state
application identifier, if applicable.
For submissions to NIH and other PHS agencies, leave these fields blank.
4.a. Federal Identifier
When a New Application is being submitted following a Pre-Application, enter the agency-assigned pre-
application number, if applicable. If this is a continuation, revision, or renewal application, enter the
assigned Federal Identifier number (for example, award number)--even if submitting a
Changed/Corrected application.
For submissions to NIH and other PHS agencies, include only the IC and serial number of the
previously assigned application/award number (e.g., use CA987654 from 1R01CA987654-01A1).
The Federal Identifier is required for Resubmission, Renewal, and Revision applications.
Existing definitions for NIH and other PHS agencies applications are somewhat different:

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-43
• New is the same; i.e., an application that is submitted for the first time. See also the policy
Resubmission of Unpaid RFA Applications and Resubmission of Applications with a
Changed Grant Activity Code.
• Resubmission is equivalent to NIH and other PHS agencies Revision. Check this option when
submitting a revised (altered or corrected) or amended application. See also the
NIH Policy
on Resubmission Applications. Institutions submitting revision or renewal applications that
are also resubmissions (A1 or A2 if allowed, see NIH Grant Notice NOT-OD-09-003,
http://grants.nih.gov/grants/guide/notice-files/not-od-09-003.html
) are instructed to select
“Resubmission.” For additional information, see NIH Guide Notice NOT-OD-10-052,
http://grants.nih.gov/grants/guide/notice-files/not-od-10-052.html).
• Renewal is equivalent to NIH and other PHS agencies Competing Continuation.
• Continuation is equivalent to NIH and other PHS agencies Progress Report. For the purposes
of NIH and other PHS agencies, the box for Continuation will not be used and should not be
checked.
• Revision is somewhat equivalent to NIH and other PHS agencies Competing Supplement, but
proposes a change in 1) the Federal Government’s financial obligations or contingent liability
from an existing obligation, or 2) any other change in the terms and conditions of the existing
award. In general, changes to the “terms and conditions of the existing award” would not
require the submission of another application through Grants.gov. Applicants should contact
the awarding agency for advice on submitting any revision/supplement application.
Applicants to NIH and other PHS agencies should complete this field when submitting a
resubmission, renewal or revision application. When submitting a “New” application, this field
should remain blank
4.b. Agency Routing Identifier
Enter the agency-assigned routing identifier per the agency-specific instructions. This is an optional field.
Unless specifically noted in a program announcement, the Agency Routing Identifier is not used by NIH
or other PHS agencies.
4.c. Previous Grants.gov Tracking ID
Enter the previous Grants.gov tracking number, if applicable.
5. Applicant Information
This information is for the Applicant Organization, not a specific individual.
Field Name Instructions
Organizational DUNS Enter the DUNS or DUNS+4 number of the applicant organization. This
field is required.
For submission to NIH and other PHS agencies, this DUNS
must match the number entered in the eRA Commons
Institutional Profile for the applicant organization. The applicant
AOR is encouraged to confirm that a DUNS has been entered in
the eRA Commons Institutional Profile (IPF) prior to submitting
an application. If your organization does not already have a
DUNS number, you will need to go to the Dun & Bradstreet
website at http://fedgov.dnb.com/webform to obtain the number.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-44
Field Name Instructions
The same DUNS should be used in the eRA Commons IPF,
Grants.gov registration and in the DUNS field in the application.
Legal Name Enter the legal name of the applicant which will undertake the assistance
activity, enter the complete address of the applicant (including
county/parish and country), and name, telephone number, e-mail, and
fax of the person to contact on matters related to this application.
Department Enter the name of the primary organizational department, service,
laboratory, or equivalent level within the organization that will undertake
the assistance activity.
Division Enter the name of the primary organizational division, office, or major
subdivision which will undertake the assistance activity.
Street1 Enter the first line of the street address for the applicant in “Street1”
field. This field is required.
Street2 Enter the second line of the street address for the applicant in “Street2”
field. This field is optional.
City Enter the city for address of applicant. This field is required.
County/Parish Enter the county/parish for address of applicant.
State Enter the State where the applicant is located. This field is required if the
applicant is located in the United States.
Province Enter the Province.
If “Country” is not Canada, please leave blank.
Country Select the country for the applicant address. This field is required.
ZIP Code Enter the nine-digit Postal Code (e.g., ZIP code) of applicant. This field
is required if the applicant is located in the United States. This field is
required if a State is selected; optional for Province.
Person to be contacted on matters involving this application:
This information is for the Administrative or Business Official, not the PD/PI. This person is the
individual to be notified if additional information is needed and/or if an award is made. To avoid
potential errors and delays in processing, please ensure that the information provided in this
section is identical to the AO profile information contained in the eRA Commons.
Field Name Instructions

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-45
Field Name Instructions
Prefix The Project Director/Principal Investigator (PD/PI) is the individual
responsible for the overall scientific and technical direction of the
project. Enter the prefix (e.g., Mr., Mrs., Rev.) for the name of the
PD/PI.
First Name Enter the first (given) name of the person to contact on matters related to
this application. This field is required.
Middle Name Enter the middle name of the person to contact on matters related to this
application.
Last Name Enter the last (family) name of the person to contact on matters related
to this application. This field is required.
Suffix Enter the suffix (e.g., Jr., Sr., Ph.D.) for the person to contact on matters
related to this application.
Position/Title Enter the Position/Title for the person to contact on matters related to
this application.
Street 1 Enter the first line of the street address for the applicant in “Street1”
field. This field is required.
Street 2 Enter second line of the street address of the person to contact on matters
related to this application. This field is optional.
City Enter the city of the person to contact on matters related to this
application. This field is required.
County/Parish Enter the county/parish of the person to contact on matters related to this
application.
State Enter the State where the applicant is located. This field is required if the
applicant is located in the United States.
Province Enter the Province.
If “Country” is not Canada, please leave blank.
County Select the country for the applicant address. This field is required.
ZIP Code Enter the nine-digit Postal Code (e.g., ZIP code) of applicant. This field
is required if the applicant is located in the United States. This field is
required if a State is selected; optional for Province.
Phone Number Enter the daytime phone number for the person to contact on matters
related to this application. This field is required

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-46
Field Name Instructions
Fax Enter the fax number for the person to contact on matters related to this
application.
Email Enter the e-mail address for the person to contact on matters related to
this application
6. Employer Identification
Enter either TIN or EIN as assigned by the Internal Revenue Service. If your organization is not in the
U.S., enter 44-4444444. This field is required.
If you have a 12-digit EIN established for grant awards from NIH or other PHS agencies, enter
all 12 digits (e.g., 1123456789A1); this includes non-U.S. organizations.
7. Type of Applicant
This information is for the Applicant Organization, not a specific individual AOR or PD/PI.
Field Name Instructions
Type of Applicant Select the appropriate applicant type code. If Small Business is selected
as Type of Applicant, then note if the organization is Woman-owned
and/or Socially and Economically Disadvantaged. This field is required.
For eligible Agencies of the Federal Government, select X:
Other (specify), and then indicate the name of the appropriate
Federal agency in the space below.
Other (Specify) Complete only if “Other” is selected as the Type of Applicant.
Woman Owned Check if you are a woman-owned small business – a small business that
is at least 51% owned by a woman or women, who also control and
operate it.
Socially and Economically
Disadvantaged
Check if you are a socially and economically disadvantaged small
business, as determined by the U.S. Small Business Administration
pursuant to Section 8(a) of the Small Business Act U.S.C. 637(a).
8. Type of Application
Field Name Instructions
Type of Application Select the type from the following list. Check only one. This field is
required.
• New: An application that is being submitted to an agency for
the first time.
• Resubmission: An application that has been previously
submitted, but was not funded, and is being resubmitted for
new consideration.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-47
Field Name Instructions
• Renewal: An application requesting additional funding for a
period subsequent to that provided by a current award. A
renewal application competes with all other applications and
must be developed as fully as though the applicant is
applying for the first time.
• Continuation: A non-competing application for an additional
funding/budget period within a previously approved project
period.
• Revision: An application that proposes a change in
1) the Federal Government’s financial obligations or contingent
liability from an existing obligation, or
2) any other change in the terms and conditions of the existing award.
Existing definitions for NIH and other PHS agencies Type of
Application are somewhat different:
• New is the same. Check this option when submitting an
application for the first time. See also the policy
Resubmission of Unpaid RFA Applications and
Resubmission of Applications with a Changed Grant
Activity Code.
• Resubmission is equivalent to NIH and other PHS agencies
Revision. Check this option when submitting a revised
(altered or corrected) or amended application. See also the
NIH Policy on Resubmission Applications
. Institutions
submitting revision or renewal applications that are also
resubmissions (A1 or A2 if allowed, see NIH Grant Notice
NOT-OD-09-003, http://grants.nih.gov/grants/guide/notice-
files/not-od-09-003.html) are instructed to select
“Resubmission.” For additional information, see NIH Guide
Notice
NOT-OD-10-052,
http://grants.nih.gov/grants/guide/notice-files/not-od-10-
052.html).
• Renewal is equivalent to NIH and other PHS agencies
Competing Continuation.
• Continuation is equivalent to NIH and other PHS agencies
Progress Report. For the purposes of NIH and other PHS
agencies, the box for Continuation will not be used and
should not be checked.
• Revision is somewhat equivalent to NIH and other PHS
agencies Supplement, but would also include other changes
as noted in the definition above. In general, changes to the
“terms and conditions of the existing award” (as noted in
example 2 above) would not require the submission of
another application through Grants.gov. Applicants should
contact the awarding agency for advice on submitting any
revision/supplement application.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-48
Field Name Instructions
This field also affects how you complete the Federal Identifier.
If “Type of Application” is “New”, you can leave the Federal
Identifier field blank unless otherwise specified in the funding
opportunity announcement.
If “Type of Application” is “Renewal,” “Revision,” or
“Resubmission,” enter the IC and serial number of the
previously assigned application/award number (e.g., use
CA987654 from 1R01CA987654-01A1).
If Revision, mark
appropriate box(es)
If Revision, mark appropriate box(es). May select more than one:
A. Increase Award
B. Decrease Award
C. Increase Duration
D. Decrease Duration
E. Other
If “Other” is selected, please specify in the text box provided.
For the purposes of NIH and other PHS agencies, the boxes for
options B, C, D, and E will generally not be used and should not
be selected unless specifically addressed in a particular FOA.
Other If “Other” is selected for Revision, add text to explain.
Is this application being
submitted to other agencies?
Check box if applicable. One selection is required.
In the field “Is this application being submitted to other
agencies?,” please check the box “yes” if one or more of the
specific aims submitted in your application are also contained in
a similar, identical, or essentially identical application submitted
to another Federal agency. Indicate the agency or agencies to
which the application has been submitted. For additional
information, please see NIH Guide Notice NOT-OD-09-100,
Reminder and Clarification of NIH Policies on Similar,
Identical, or Essentially Identical Applications, Submission of
Applications Following RFA Review, and Submission of
Applications with a Changed Activity Code,
http://grants.nih.gov/grants/guide/notice-files/NOT-OD-09-
100.html.
What Other Agencies? Enter Agency name.
9. Name of Federal Agency
Name the Federal agency from which assistance is being requested with this application. This field is pre-
populated from the opportunity package.
PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-49
10. Catalog of Federal Domestic Assistance (CFDA) Number and Title (CFDA)
This is the Catalog of Federal Domestic Assistance number of the program under which assistance is
requested. This field is pre-populated from the opportunity package.
This field may be blank if you are applying to an opportunity that references multiple CFDA
numbers. When this field is blank, leave it blank; the field will not allow any data entry. The
appropriate CFDA number will be automatically assigned by the agency once the application is
assigned to the appropriate awarding component.
11. Descriptive Title of Applicant’s Project
Enter a brief descriptive title of the project. This field is required.
A “new” application must have a different title from any other PHS project with the same PD/PI.
A “resubmission” or “renewal” application should normally have the same title as the previous
grant or application. If the specific aims of the project have significantly changed, choose a new
title.
A “revision” application must have the same title as the currently funded grant.
NIH and other PHS agencies limit title character length to 81 characters, including the spaces
between words and punctuation. Titles in excess of 81 characters will be truncated. Be sure to
only use standard characters in the descriptive title: A through Z, a through z, 0 through 9, space
( ), and underscore (_).
12. Proposed Project
Start Date: Enter the proposed start date of the project. This field is required.
Ending Date: Enter the proposed end date of the project. This field is required.
13. Congressional District of Applicant
Enter the Congressional District in the format: 2 character State Abbreviation – 3 character District
Number. Examples: CA-005 for California’s 5th district, CA-012 for California’s 12th district.
If outside the U.S., enter 00-000.
To locate your congressional district, visit the Grants.gov website.
For States and U.S. territories with only a single congressional district enter “001” for the district
code. For jurisdictions with no representative, enter “099”. For jurisdictions with a nonvoting
delegate, enter “098” for the district number. Example: DC-098, PR-098.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-50

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-51
14. Program Director/Principal Investigator (PD/PI) Contact Information
If submitting an application reflecting Multiple PD/PIs, the individual designated as the Contact
PI must be affiliated in the Commons with the applicant organization and should be entered here.
See Section 4.5 Senior/Key Person Profile Forms
for additional instructions for Multiple PD/PIs.
To avoid potential errors and delays in processing, please ensure that the information provided in
this section is identical to the PD/PI profile information contained in the eRA Commons.
Field Name Instructions
Prefix The Project Director/Principal Investigator (PD/PI) is the individual
responsible for the overall scientific and technical direction of the
project. Enter the prefix (e.g., Mr., Mrs., Rev.) for the name of the
PD/PI.
First Name Enter the first name of the PD/PI. This field is required.
Middle Name Enter the middle name of the PD/PI.
Last Name Enter the last (family) name of the PD/PI. This field is required.
Suffix Enter the suffix (e.g., Jr., Sr., Ph.D) of the PD/PI.
Do not use this field to record degrees. Degrees for the PD/PI
are requested separately in the Senior/Key Person Profile.
Position/Title Enter the position/title of the PD/PI.
Organization Name Enter the name of organization for the PD/PI. This field is required.
Department Enter the name of primary organizational department, service,
laboratory, or equivalent level within the organization of the PD/PI.
Division Enter the name of primary organizational division, office, or major
subdivision of the PD/PI.
Street1 Enter first line of the street address for the PD/PI in the “Street1” field.
This field is required.
Street2 Enter the second line of the street address for the PD/PI in the “Street2”
field. This field is optional.
City Enter the City for address of the PD/PI. This field is required.
County/Parish Enter the county/parish for address of the PD/PI.
State Enter the State where the PD/PI is located. This field is required if the
PD/PI is located in the United States.
PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-52
Field Name Instructions
Province Enter the province for PD/PI.
If “Country” is not Canada, please leave blank.
Country Select the country for the PD/PI address.
ZIP/Postal Code Enter the Postal Code (e.g., ZIP code) of the PD/PI. This field is
required if the PD/PI is located in the United States.
A nine-digit Zip code is required.
Phone Number Enter the daytime phone number for the PD/PI. This field is required.
Fax Number Enter the fax number for the PD/PI.
E-mail Enter the e-mail address for the PD/PI. This field is required.
15. Estimated Project Funding
Field Name Instructions
a. Total Federal Funds
Requested
Enter total Federal funds requested for the entire project period. This
field is required.
Please ensure number(s) complies with application
requirements.
b. Total Non-Federal Funds Enter total non-Federal funds proposed for the entire project period. This
field is required.
For NIH and other PHS agencies, enter “0” in this field unless
cost sharing is a requirement for the specific announcement.
c. Total Federal & Non-
Federal Funds
Enter total estimated funds for the entire project period, including both
Federal and non-Federal funds. This field is required.
For NIH and other PHS agencies applicants, this field will be the
same as Total Federal Funds Requested above unless the
specific announcement indicates that cost sharing is a
requirement.
d. Estimated Program
Income
Identify any Program Income estimated for this project period if
applicable. This field is required.
16. Is Application Subject to Review by State Executive Order 12372 Process?
If yes, check box. If the announcement indicates that the program is covered under Executive Order
12372, applicants should contact the State Single Point of Contact (SPOC) for Federal Executive Order
12372. If no, check appropriate box. One selection is required.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-53
If block 16a is checked, insert date application was submitted to State.
For NIH and other PHS agencies submissions using the SF424 (R&R), applicants should check
“No, Program is not covered by E.O. 12372.”
17. Certification
Check “I agree” to provide the required certifications and assurances. This field is required.
The list of NIH and other PHS agencies Assurances, Certifications, and other Policies is found in
Supplemental Instructions Part III, Policies, Assurances, Definitions, and Other Information
.
The applicant organization is responsible for verifying its eligibility and the accuracy, validity,
and conformity with the most current institutional guidelines of all the administrative, fiscal, and
scientific information in the application, including the Facilities and Administrative rate.
Deliberate withholding, falsification, or misrepresentation of information could result in
administrative actions, such as withdrawal of an application, suspension and/or termination of an
award, debarment of individuals, as well as possible criminal penalties. The signer further
certifies that the applicant organization will be accountable both for the appropriate use of any
funds awarded and for the performance of the grant-supported project or activities resulting from
this application. The grantee institution may be liable for the reimbursement of funds associated
with any inappropriate or fraudulent conduct of the project activity.
18. SFLLL (Disclosure of Lobbying Activities) or Other Explanatory Documentation
If applicable, attach the SFLLL or other explanatory document per agency instructions.
If unable to certify compliance in with the Certification above attach an explanation.
Additionally, as applicable, attach the SFLLL (Standard Form LLL, Disclosure of Lobbying
Activities) or other documents in this item. A fillable version of the SFLLL form is available at
http://www.whitehouse.gov/omb/assets/omb/grants/sflllin.pdf
.
19. Authorized Representative
This is equivalent to the individual with the organizational authority to sign for an application;
otherwise known as the Authorized Organization Representative or the Signing Official.
Field Name Instructions
Prefix Enter the prefix (Mr., Mrs., Rev.) for the name of the Authorized
Representative.
First Name Enter the first (given) name of the Authorized Representative. This field
is required.
Middle Name Enter the middle name of the Authorized Representative.
Last Name Enter the last (family) name of the Authorized Representative. This field
is required.
Suffix Enter the suffix (e.g., Jr., Sr., Ph.D.) for the name of the Authorized
Representative.
Position/Title Enter the title of the Authorized Representative. This field is required.
Organization Enter the name of the organization for the Authorized Representative.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-54
Field Name Instructions
This field is required.
Department Enter the name of the primary organizational department, service,
laboratory, or equivalent level within the organization of the Authorized
Representative.
Division Enter the name of the primary organizational division, office, or major
subdivision of the Authorized Representative.
Street1 Enter the first line of the street address for the Authorized Representative
in the “Street1” field. This field is required.
Street2 Enter the second line of the street address for the Authorized
Representative in the “Street2” field. This field is optional.
City City for address of the Authorized Representative. This field is required.
County/Parish Enter the county/parish for address of the Authorized Representative.
State Enter the state where the Authorized Representative is located. This field
is required if the Authorized Representative is located in the United
States.
Province Enter the Province for the Authorized Representative.
If “Country” is not Canada, please leave blank.
Country Select the country for the Authorized Representative address.
ZIP/Postal Code Enter Postal Code (e.g., ZIP code) of the Authorized Representative.
This field is required if the Authorized Representative is located in the
United States.
A nine-digit Zip code is required.
Phone Number Enter the daytime phone number for the Authorized Representative. This
field is required.
Fax Number Enter the fax number for the Authorized Representative.
E-mail Enter the e-mail address for the Authorized Representative. This field is
required.
Signature of Authorized
Representative
It is the organization’s responsibility to assure that only properly
authorized individuals sign in this capacity and/or submit the application
to Grants.gov. If this application is submitted through Grants.gov, leave

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-55
Field Name Instructions
blank. If a hard copy is submitted, the AOR must sign this block.
Date Signed If this application is submitted through Grants.gov, the system will
generate this date. If submitting a hard copy, enter the date the AOR
signed the application.
20. Pre-Application
If submitting a pre-application, provide a summary description of the project in accordance with the
announcement and/or agency specific instructions, and save the file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and then click Open.
Unless specifically noted in a program announcement, NIH and other PHS agencies do not use
Pre-applications.
21. Cover Letter Attachment
Attach the cover letter in accordance with the announcement and/or the agency specific instructions.
Applicants are encouraged to include a cover letter with the application. The cover letter is only
for internal use and will not be shared with peer reviewers. The letter should contain any of the
following information that applies to the application:
1. Application title.
2. Funding Opportunity (PA or RFA) title of the NIH initiative.
3. Request of an assignment (referral) to a particular awarding component(s) or Scientific
Review Group (SRG). The PHS makes the final determination.
4. List of individuals (e.g., competitors) who should not review your application and why.
5. Disciplines involved, if multidisciplinary.
6. For late applications (see Late Application policy in Section 2.14) include specific
information about the timing and nature of the cause of the delay.
7. When submitting a Changed/Corrected Application after the submission date, a cover letter is
required explaining the reason for the Changed/Corrected Application. If you already
submitted a cover letter with a previous submission and are now submitting a
Changed/Corrected Application, you must include all previous cover letter text in the revised
cover letter attachment. The system does not retain any previously submitted cover letters;
therefore, you must repeat all information previously submitted in the cover letter as well as
any additional information.
8. Explanation of any subaward budget components that are not active for all periods of the
proposed grant.
9. Statement that you have attached any required agency approval documentation for the type of
application submitted. This may include approval for applications $500,000 or more,
approval for Conference Grant or Cooperative Agreement (R13 or U13), etc.
Suggested Cover Letter Format
The Division of Receipt and Referral (DRR), Center for Scientific Review (CSR) is responsible
for assigning applications to ICs and to Scientific Review Groups (SRGs). DRR will be utilizing

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-56
knowledge management approaches as an adjunct to the work of referral experts as part of an
overall plan to shorten the time from submission to review. Analysis has shown that requests
made by investigators are a valuable source of information in this process. In order to facilitate
the use of these requests in conjunction with knowledge management analysis of the content of
the application, applicants are requested to use the following format when assignment requests
are contained in a cover letter.
• List one request per line.
• Place Institute/Center (IC) and SRG review requests (if both are made) on separate lines.
• Place positive and negative requests (if both are made) on separate lines.
• Include name of IC or SRG, followed by a dash and the acronym. Do not use
parentheses.
• Provide explanations for each request in a separate paragraph.
Examples:
Please assign this application to the following:
Institutes/Centers
National Cancer Institute - NCI
National Institute for Dental and Craniofacial Research – NIDCR
Scientific Review Groups
Molecular Oncogenesis Study Section – MONC
Cancer Etiology Study Section – CE
Please do not assign this application to the following:
Scientific Review Groups
Cancer Genetics Study Section – CG
The reasons for this request are [provide a narrative explanation for the request(s)].

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-57
4.3 Project/Performance Site Locations Form
Indicate the primary site where the work will be performed. If a portion of the project will be performed
at any other site(s), identify the site location(s) in the blocks provided.
Project/Performance Site Primary Location
Generally, the Primary Location should be that of the applicant organization or identified as off-
site in accordance with the conditions of the applicant organization’s negotiated Facilities and
Administrative (F&A) agreement. This information must agree with the F&A information on the
Cover Page Supplement Form of the application. If there is more than one performance site,
including any Department of Veterans Affairs (VA) facilities and foreign sites, list them in the

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-58
fields provided for Location 1 - # below. Applicants should also provide an explanation of
resources available from each Project/Performance Site, the Facilities and Resources attachment
of the Other Project Information form, and describe any consortium/contractual arrangements in
the Consortium/Contractual Arrangements attachment of the PHS 398 Research Plan.
Unless otherwise instructed in a FOA, do not check the “I am submitting an application as an
individual, and not on behalf of a company, state, local or tribal government, academia, or other
type of organization” box.
If a Project/Performance Site is engaged in research involving human subjects, the applicant
organization is responsible for ensuring that the Project/Performance Site operates under an
appropriate Federal Wide Assurance for the protection of human subjects and complies with 45
CFR part 46 and other NIH human subject related policies described in Supplemental Instructions
Part II of this Application Guide and in the NIH Grants Policy Statement.
For research involving live vertebrate animals, the applicant organization must ensure that all
Project/Performance Sites hold an OLAW-approved Animal Welfare Assurance. If the applicant
organization has neither an animal care and use program, facilities to house animals and conduct
research on site, nor an IACUC, and the animal work will be conducted at an institution with an
Animal Welfare Assurance, the applicant must obtain an Inter-institutional Assurance from
OLAW prior to an award.
Field Name Instructions
Organization Name Indicate the organization name of the primary site where the work will
be performed. If a portion of the project will be performed at any other
sites(s), identify the site location(s) in the block(s) provided.
DUNS Number Enter the DUNS number associated with the organization where the
project will be performed.
The DUNS Number is a required field for the Primary
Performance Site.
Street1 Enter first line of the street address in "Street 1" field of the primary
performance site location. This field is required.
Street2 Enter second line of the street address in "Street 2" field for the primary
performance site location. This field is optional.
City Enter the City for address of the primary performance site location. This
field is required.
County Enter the County of the primary performance site location.
State Enter the State where the primary performance site location is located.
This field is required if the Project Performance Site is located in the
United States..

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-59
Field Name Instructions
Province Enter the province of the primary performance site location.
If “Country” is not Canada, please leave blank.
Country Select the Country of the Primary Performance Site location. This field
is required.
ZIP Code Enter the nine-digit Postal Code (e.g., ZIP code) of the performance site
location. This field is required if the performance site location is in the
United States.
A nine-digit Zip code is required.
Project/Performance Site
Congressional District
Enter the Congressional District in the format: 2 character State
Abbreviation – 3 character District Number. Examples: CA-005 for
California’s 5th district, CA-012 for California’s 12th district.
If all districts in a state are affected, enter “all” for the district number.
Example MD-all for all congressional districts in Maryland.
If nationwide (all districts in all states), enter US-all.
If the program/project is outside the U.S., enter 00-000.
To locate your congressional district, visit the Grants.gov website. Note
it is likely this field will be identical to the “Congressional District of
Applicant” field provided elsewhere in the application.
For States and U.S. territories with only a single congressional
district enter “001” for the district code. For jurisdictions with
no representative, enter “099”. For jurisdictions with a
nonvoting delegate, enter “098” for the district number.
Example: DC-098, PR-098.
Project/Performance Site Location 1
Field Name Instructions
Organization Name Indicate the organization name of the primary site where the work will
be performed. If a portion of the project will be performed at any other
sites(s), identify the site location(s) in the block(s) provided.
DUNS Number Enter the DUNS number associated with the organization where the
project will be performed. This field is optional.
Street1 Enter first line of the street address in "Street 1" field of the primary
performance site location. This field is required.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-60
Field Name Instructions
Street2 Enter second line of the street address in "Street 2" field for the primary
performance site location. This field is optional.
City Enter the City of the performance site location. This field is required.
County Enter the County of the performance site location.
State Enter the State where the primary performance site location is located.
This field is required if the Project Performance Site is located in the
United States.
Province Enter the Province where the primary performance site location is
located.
If “Country” is not Canada, please leave blank.
Country Select the name of the country for the primary project performance site.
ZIP Code Enter the nine-digit Postal Code (e.g., ZIP code) of the primary
performance site location. This field is required if the Project
Performance Site is located in the United States.
Project/Performance Site
Congressional District
Enter the Congressional District in the format: 2 character State
Abbreviation – 3 character District Number. Examples: CA-005 for
California’s 5th district, CA-012 for California’s 12th district.
If all districts in a state are affected, enter “all” for the district number.
Example MD-all for all congressional districts in Maryland.
If nationwide (all districts in all states), enter US-all.
If the program/project is outside the U.S., enter 00-000.
To locate your congressional district, visit the Grants.gov website. Note
it is likely this field will be identical to the “Congressional District of
Applicant” field provided elsewhere in the application.
For States and U.S. territories with only a single congressional
district enter “001” for the district code. For jurisdictions with
no representative, enter “099”. For jurisdictions with a
nonvoting delegate, enter “098” for the district number.
Example: DC-098, PR-098.
For additional performance site locations, click Next Site to display the fields for Project/Performance
Site Locations 2 through 300.
If you need to add more than 300 locations, enter the information in a separate file. In the Additional
Locations section at the bottom of the form, click Add Attachment, select the file, and then click Open.
A sample Additional Performance Sites format page for greater than eight locations is found under
“Additional Format Pages” at: http://grants.nih.gov/grants/funding/424/index.htm
.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-61
4.4 Other Project Information Form
1. Are Human Subjects Involved?
If activities involving human subjects are planned at any time during the proposed project at any
performance site, check yes. Check yes even if the proposed project is exempt from Regulations for the
Protection of Human Subjects. If no, skip the rest. This field is required.
Applications proposing human subjects research may be required to submit additional
information, forms, or attachments with the application, in accordance with NIH and PHS
policies covering human subjects research. Refer to
Part II, Supplemental Instructions for
Preparing the Human Subjects Section of the Research Plan.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-62
1.a. If YES to Human Subjects
Is the Project Exempt from Federal Regulations? Yes/No
Yes: If the project is exempt from Federal regulations, check Yes.
No: If the project is not exempt from Federal regulations, check No.
If yes, check appropriate exemption number 1, 2, 3, 4, 5, 6
Select the appropriate exemption number from 1, 2, 3, 4, 5, 6.
If human subject activities are exempt from Federal regulations, provide the exemption numbers
corresponding to one or more of the exemption categories. The six categories of research that qualify for
exemption from coverage by the regulations are defined in the Common Rule for the Protection of Human
Subjects. These regulations can be found at
http://www.hhs.gov/ohrp/humansubjects/guidance/45cfr46.html
.
OHRP guidance states that appropriate use of Exemptions described in 45 CFR 46 should be
determined by an authority independent from the investigators
(http://answers.hhs.gov/ohrp/categories/1564
). Institutions often designate their IRB to make this
determination. Because NIH does not require IRB approval at the time of application, the
exemptions designated often represent the opinion of the PD/PI, and the justification provided for
the exemption by the PD/PI is evaluated during peer review.
Proposed research may include more than one research project; thus the application may include
individual projects that meet the requirements for non-exempt or exempt human subjects
research, or are not defined as human subjects research. Human subjects research should be
designated as exempt if all of the proposed research meets the criteria for one or more of the six
exemptions.
If no, is the IRB review Pending? Yes/No
If IRB review is pending, check Yes. If IRB review is not pending, check No.
IRB Approval Date
Enter the latest Institutional Review Board (IRB) approval date (if available). Leave blank if Pending.
Applicants should check “Yes” to the question “Is the IRB review Pending?” even if the IRB
review/approval process has not yet begun at the time of submission. Also note that an IRB
Approval Date is not required at the time of submission. This may be requested later in the pre-
award cycle as a Just-In-Time
requirement.
Human Subject Assurance Number
Enter the approved Federal Wide Assurance (FWA), Multiple Project Assurance (MPA), Single Project
Assurance (SPA) Number or Cooperative Project Assurance Number that the applicant has on file with
the Office for Human Research Protections, if available. If the applicant has a FWA number, enter the 8-
digit number. Do not enter the FWA before the number.
Insert “None” if the applicant organization does not have an approved assurance on file with
OHRP. In this case, the applicant organization, by the signature in the Certification signature
section on the SF424 (R&R) Cover form, is declaring that it will comply with 45 CFR part 46 and
proceed to obtain a human subjects assurances (see http://www.hhs.gov/ohrp
). Do not insert the
human subjects assurance number of any collaborating institution in the space provided.
2. Are Vertebrate Animals Used?
If activities involving vertebrate animals are planned at any time during the proposed project at any
performance site, check yes. If no, skip the rest of block 2. This field is required.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-63
Note that the generation of custom antibodies constitutes an activity involving vertebrate animals.
If animal involvement is anticipated within the period of award but plans are indefinite, check
"Yes" and add a Vertebrate Animals attachment to, provide an explanation and indicate when it is
anticipated that animals will be used. If an award is made, prior to the involvement of animals the
grantee must provide all of the information required by adding a Vertebrate Animals attachment
in the Research Plan and verifying of IACUC approval to the awarding component.
2.a. If YES to Vertebrate Animals
Is the IACUC review Pending?
Indicate if an Institutional Animal Care and Use Committee (IACUC) review is pending.
Click Yes if an IACUC review is pending. Click No, if no review is pending.
Check “Yes” even if the IACUC review and approval process has not yet begun.
IACUC Approval Date
Enter the latest IACUC approval date (if available). Leave blank if Pending.
IACUC approval must have been granted within three years to be valid. Note that an IACUC
Approval Date is not required at the time of submission. NIH does not require verification of
review and approval of the proposed research by the IACUC before peer review of the
application. However, this information is required under Just-In-Time Policy
.
Animal Welfare Assurance Number
Enter the Federally approved assurance number, if available.
Enter “None” if the applicant organization does not have an OLAW-approved Animal Welfare
Assurance. To determine if the applicant organization holds an Animal Welfare Assurance, see
the lists of Domestic and Foreign
Assured institutions. Do not enter the Animal Welfare
Assurance number for a Project/Performance Site of a collaborating institution. When an
applicant organization does not have an Animal Welfare Assurance, the Authorized Organization
Representative’s signature on the application constitutes declaration that the applicant
organization will submit an Animal Welfare Assurance when requested by OLAW.
3. Is proprietary/privileged information included in the application?
Patentable ideas, trade secrets, privileged or confidential commercial or financial information, disclosure
of which may harm the applicant, should be included in applications only when such information is
necessary to convey an understanding of the proposed project. If the application includes such
information, check yes and clearly mark each line or paragraph on the pages containing the
proprietary/privileged information with a legend similar to: "The following contains
proprietary/privileged information that (name of applicant) requests not be released to persons outside the
Government, except for purposes of review and evaluation. " This field is required.
If a grant is awarded as a result of or in connection with the submission of this application, the
Government shall have the right to use or disclose the information to the extent authorized by
law. Although the grantee institution and the PD/PI will be consulted about any such disclosure,
the PHS will make the final determination. Any indication by the applicant that the application
contains proprietary or privileged information does not automatically shield the information from
release in response to a Freedom of Information Act (FOIA) request should the application result
in an award (see 45 CFR Part 5). If an applicant fails to identify proprietary information at the
time of submission as instructed in the application guide, a significant substantive justification
will be required to withhold the information if requested under FOIA.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-64
4. Environmental Questions
Most NIH research grants are not expected to individually or cumulatively have a significant
effect on the environment, and NIH has established several categorical exclusions allowing most
applicants to answer “No” to this question unless a specific FOA indicates that the National
Environmental Policy Act (NEPA) applies. However, if an applicant expects that the proposed
project will have an actual or potential impact on the environment, or if any part of the proposed
research and/or project includes one or more of the following categorical exclusions listed below,
the box marked “Yes” should be checked and an explanation provided.
1. The potential environmental impacts of the proposed research may be of greater scope or
size than other actions included within a category.
2. The proposed research threatens to violate a Federal, State, or local law established for the
protection of the environment or for public health and safety.
3. Potential effects of the proposed research are unique or highly uncertain.
4. Use of especially hazardous substances or processes is proposed for which adequate and
accepted controls and safeguards are unknown or not available.
5. The proposed research may overload existing waste treatment plants due to new loads
(volume, chemicals, toxicity, additional hazardous wasted, etc.)
6. The proposed research may have a possible impact on endangered or threatened species.
7. The proposed research may introduce new sources of hazardous/toxic wastes or require
storage of wastes pending new technology for safe disposal.
8. The proposed research may introduce new sources of radiation or radioactive materials.
9. Substantial and reasonable controversy exists about the environmental effects of the
proposed research.
4.a. Does the project have an actual or perceived impact—positive or negative—on the
environment?
Indicate if this project has an actual or perceived impact on the environment? Click No here if this is not
the case. This field is required.
4.b. If yes, please explain
Explanation of the actual or perceived impact - positive or negative - on the environment.
4.c. If this project has an actual or perceived impact on the environment, has an exemption been
authorized or an Environmental Assessment (EA) or an Environmental Impact Statement (EIS)
been performed?
Check yes or no. This field is required.
4.d. If yes, please explain
Enter additional details about the EA or EIS.
5. Is the research performance site designated, or eligible to be designated, as a historic place?
Yes/No
If any research performance site is designated, or eligible to be designated, as a historic place, if Yes,
check the Yes box and then provide an explanation in the box provided in 5.a. Otherwise, check the No
box. This field is required.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-65
5.a. If yes, please explain:
If you checked the Yes box indicating any performance site is designated, or eligible to be designated, as
a historic place, provide the explanation here.
6. Does this project involve activities outside of the United States or partnerships with
International Collaborators?
Indicate whether this project involves activities outside of the United States or partnerships with
international collaborators. Check yes or no. This field is required.
Applicants to NIH and other PHS agencies must check “Yes” if the applicant organization is a
foreign institution or if the project includes a foreign component. For a definition of a
foreign
component, see “Definitions” section of Supplemental Instructions Part III: Policies, Assurances,
Definitions, and Other Information.
6.a. If yes, identify countries
Enter the countries with which international cooperative activities are involved.
6.b. Optional Explanation
Enter an explanation for involvement with outside entities (optional).
If you have checked “Yes” to 6, applicants to the NIH and other PHS agencies must describe
special resources or characteristics of the research project (e.g., human subjects, animals, disease,
equipment, and techniques), whether similar research is being done in the United States and
whether there is a need for additional research in this area. Provide this information in a separate
file, attaching it on the Other Project Information form. In the body of the text, begin the section
with a heading indicating “Foreign Justification.” When saving this file, please name it “Foreign
Justification” as well.
7. Project Summary/Abstract
The Project Summary must contain a summary of the proposed activity suitable for dissemination to the
public. It should be a self-contained description of the project and should contain a statement of
objectives and methods to be employed. It should be informative to other persons working in the same or
related fields and insofar as possible understandable to a scientifically or technically literate lay reader.
This Summary must not include any proprietary/confidential information. Please click the Add
Attachment button to the right of this field to complete this entry.
The Project Summary is meant to serve as a succinct and accurate description of the proposed
work when separated from the application. State the application’s broad, long-term objectives and
specific aims, making reference to the health relatedness of the project (i.e., relevance to the
mission of the agency). Describe concisely the research design and methods for achieving the
stated goals. This section should be informative to other persons working in the same or related
fields and insofar as possible understandable to a scientifically or technically literate reader.
Avoid describing past accomplishments and the use of the first person. Finally, please make every
effort to be succinct. This section must be no longer than 30 lines of text, and follow the required
font and margin specifications
. An abstract which exceeds this allowable length may be flagged
as an error by the agency upon submission. This would require a corrective action before the
application will be accepted.
As noted above, do not include proprietary, confidential information or trade secrets in the
description section. If the application is funded, the Project Description will be entered into an
NIH database and made available on the NIH Research Portfolio Online Reporting Tool
(RePORT, available at http://report.nih.gov
) and will become public information.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-66
The attachment must be in PDF format. (See Section 2.6 for additional information on preparing
attachments.)
8. Project Narrative
Provide Project Narrative in accordance with the announcement and/or agency-specific instructions.
Please click the Add Attachment button to the right of this field to complete this entry.
For NIH and other PHS agencies applications, using no more than two or three sentences,
describe the relevance of this research to public health. In this section, be succinct and use plain
language that can be understood by a general, lay audience.
A separate Research Plan form is required for NIH and other PHS agencies applications. Refer to
Section 5.5, Research Plan
Form, for separate file uploads and instructions.
9. Bibliography & References Cited
Provide a bibliography of any references cited in the Project Narrative. Each reference must include the
names of all authors (in the same sequence in which they appear in the publication), the article and
journal title, book title, volume number, page numbers, and year of publication. Include only
bibliographic citations. Applicants should be especially careful to follow scholarly practices in providing
citations for source materials relied upon when preparing any section of the application. To attach a
document for Bibliography and References Cited, click Add Attachment.
Unless otherwise noted in an FOA, this section is required for submissions to NIH and other PHS
agencies. This section (formerly “Literature Cited”) should include any references cited in the
PHS 398 Research Plan form (see Section 5.5
for details on completing that form). When citing
articles that fall under the Public Access Policy, were authored or co-authored by the applicant
and arose from NIH support, provide the NIH Manuscript Submission reference number (e.g.,
NIHMS97531) or the PubMed Central (PMC) reference number (e.g., PMCID234567) for each
article. If the PMCID is not yet available because the Journal submits articles directly to PMC on
behalf of their authors, indicate “PMC Journal – In Process.” A list of these journals is posted at:
http://publicaccess.nih.gov/submit_process_journals.htm.
Citations that are not covered by the Public Access Policy, but are publicly available in a free,
online format may include URLs or PubMed ID (PMID) numbers along with the full reference
(note that copies of publicly available publications are not accepted as appendix material). The
references should be limited to relevant and current literature. While there is not a page
limitation, it is important to be concise and to select only those literature references pertinent to
the proposed research.
10. Facilities & Other Resources
This information is used to assess the capability of the organizational resources available to perform the
effort proposed. Identify the facilities to be used (Laboratory, Animal, Computer, Office, Clinical and
Other). If appropriate, indicate their capacities, pertinent capabilities, relative proximity and extent of
availability to the project. Describe only those resources that are directly applicable to the proposed work.
Provide any information describing the Other Resources available to the project (e.g., machine shop,
electronic shop) and the extent to which they would be available to the project. Please click the Add
Attachment button to the right of this field to complete this entry.
No special form is required but this section must be completed and attached for submissions to
NIH and other PHS agencies unless otherwise noted in an FOA. Describe how the scientific
environment in which the research will be done contributes to the probability of success (e.g.,
institutional support, physical resources, and intellectual rapport). In describing the scientific
environment in which the work will be done, discuss ways in which the proposed studies will

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-67
benefit from unique features of the scientific environment or subject populations or will employ
useful collaborative arrangements.
For Early Stage Investigators (ESIs), describe institutional investment in the success of the
investigator, e.g., resources for classes, travel, training; collegial support such as career
enrichment programs, assistance and guidance in the supervision of trainees involved with the
ESI’s project, and availability of organized peer groups; logistical support such as administrative
management and oversight and best practices training; and financial support such as protected
time for research with salary support. See, http://grants.nih.gov/grants/new_investigators/
.
If there are multiple performance sites, describe the resources available at each site.
Describe any special facilities used for working with biohazards or other potentially dangerous
substances. Note: Information about select agents must be described in the Research Plan, Section
11 (Select Agent Research).
11. Equipment
List major items of equipment already available for this project and, if appropriate identify location and
pertinent capabilities. Please click the Add Attachment button to the right of this field to complete this
entry.
12. Other Attachments
Attach a file only to provide any other project information not provided above or in accordance with the
announcement and/or agency-specific instruction.
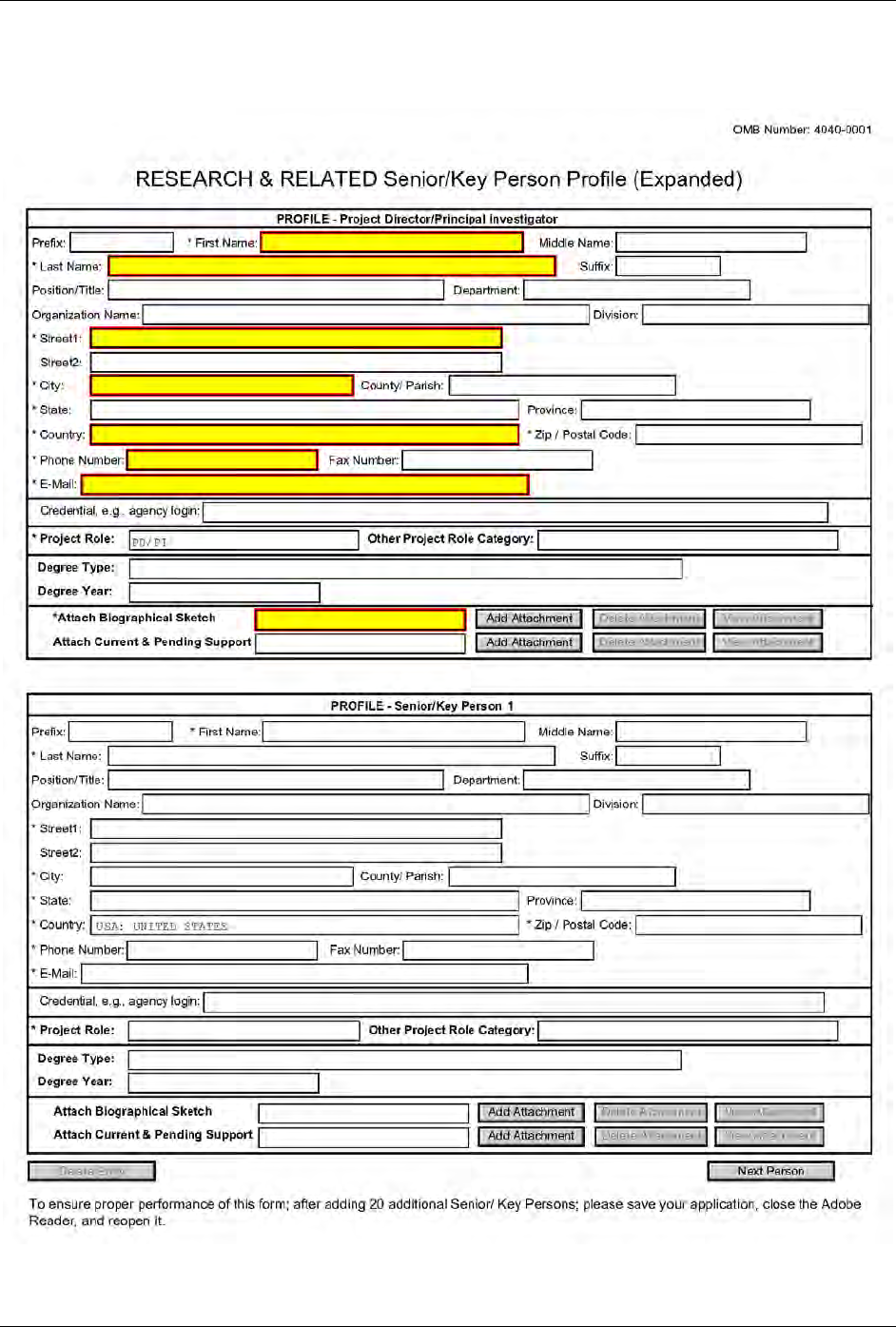
PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-68
4.5 Senior/Key Person Profile (Expanded) Form

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-69
This form provides the ability to collect structured data for up to 100 senior/key persons. Data must be
entered for the first 100 individuals (PD/PI + 99 others) before the Additional Senior/Key Person Form
Attachments section becomes available. The information for the PD/PI continues to be pre-populated
from the SF424 (R&R) Cover form. See instructions in section 4.2 Cover Form if these fields are empty.
Unless otherwise specified in an agency announcement, senior/key personnel are defined as all
individuals who contribute in a substantive, meaningful way to the scientific development or execution of
the project, whether or not salaries are requested. Consultants should be included if they meet this
definition.
Multiple PD/PIs
NIH is now accepting applications reflecting Multiple PD/PIs for all grant activity codes using
the SF424 (R&R) application. When submitting an application involving Multiple PD/PIs, the
Contact PD/PI must be affiliated in the Commons with the applicant organization and should be
listed as the PD/PI in the SF424 R&R Cover Form (see Section 4.2.14
). That information
automatically prepopulates the first senior/key person profile record in this form. For the
additional PD/PIs, complete all the requested information. Each PD/PI must be assigned the
PD/PI role, even those at subaward/consortium sites when applicable. (Do not use the “Co-
PD/PI” or Co-Investigator role.) For more information, please see
Section 4.8 Special Instructions
for Preparing Applications with a Subaward/Consortium.
Each PD/PI must also be registered in the eRA Commons and must be assigned the PI Role
in that system (note other roles such as SO or IAR will not give PD/PIs the appropriate
access to the application records). Each PD/PI must include their respective eRA Commons
ID in the Credential field. For more information on NIH Implementation of Multiple PD/PIs,
see: http://grants.nih.gov/grants/multi_pi/index.htm
.
When completing the detailed budget form for either the prime organization or a subaward/consortium
organization, the project roles listed in the budget form should be consistent with those used in the
Senior/Key Person Form.
Profile – Program Director/Principal Investigator (PD/PI)
Field Name Instructions
Prefix Pre-populated from the SF 424 (R&R). The prefix (e.g., Mr., Mrs.,
Rev.) for the name of the PD/PI.
First Name Pre-populated from the SF 424 (R&R). The first (given) name of the
PD/PI. This field is required.
Middle Name Pre-populated from the SF 424 (R&R). The middle name of the PD/PI.
Last Name Pre-populated from the SF 424 (R&R). The last (family) name of the
PD/PI. This field is required.
Suffix Pre-populated from the SF 424 (R&R). The suffix (e.g., Jr, Sr, PhD) for
the name of the PD/PI.
Position/Title Pre-populated from the SF 424 (R&R). The title of the PD/PI.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-70
Field Name Instructions
Department Pre-populated from the SF 424 (R&R). The name of primary
organizational department, service, laboratory, or equivalent level within
the organization of the PD/PI.
Organization Name Pre-populated from the SF 424 (R&R). The name of organization of the
PD/PI.
Division Pre-populated from the SF 424 (R&R). The name of primary
organizational division, office, or major subdivision of the PD/PI.
Street1 Pre-populated from the SF 424 (R&R). The first line of the street address
for the PD/PI in the "Street 1" field. This field is required.
Street2 Pre-populated from the SF 424 (R&R). The second line of the street
address for the PD/PI in the "Street 2" field. This field is optional.
City Pre-populated from the SF 424 (R&R). The city for address of PD/PI.
This field is required.
County/Parish Pre-populated from the SF 424 (R&R). The county/parish for address of
PD/PI.
State Pre-populated from the SF 424 (R&R). The state where the PD/PI is
located. This field is required if the PD/PI is located in the United
States.
Province Pre-populated from the SF 424 (R&R). The Province where the PD/PI is
located.
If “Country” is not Canada, this will be blank.
Country Pre-populated from the SF 424 (R&R). The country for the PD/PI
address. This field is required.
ZIP Code Pre-populated from the SF 424 (R&R). The postal Code (e.g., ZIP code)
of PD/PI. This field is required if the PD/PI is located in the United
States.
A nine-digit Zip code is required.
Phone Number Pre-populated from the SF 424 (R&R). The daytime phone number for
the PD/PI. This field is required.
Fax Number Pre-populated from the SF 424 (R&R). The fax number for the PD/PI.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-71
Field Name Instructions
E-mail Pre-populated from the SF 424 (R&R). The e-mail address for the
PD/PI. This field is required for PD/PI.
Credential, e.g., agency
login
If you are submitting to an agency (e.g., NIH) where you have an
established personal profile, enter the agency ID. If not, leave blank.
For NIH and other PHS agencies, registration in the eRA
Commons for all PD/PIs is required. The assigned Commons
UserName (the unique name used to log into the system) for
anyone assigned the PD/PI role must be entered here and
must have the PI role in eRA Commons. This is a required
field for applications submitted to NIH and other PHS
agencies. Applications will not pass agency validation
requirements without this field.
Project Role Select one. Use "Other" if a category is not listed in the pick list.
Select PD/PI for this person.
Other Project Role Category Complete if you selected “Other Professional” or “Other” as a project
role; e.g., Engineer, Chemist.
Degree Type Enter the highest academic or professional degree or other credentials
(e.g., R.N.). This is optional information.
Degree Year Enter the year the highest degree or other credential was obtained. This
is optional information.
Attach Biographical Sketch Provide a biographical sketch for the senior/key person. Recommended
information includes: Education and Training, Research and Professional
Experience, Collaborators and Affiliations (for conflicts of interest),
Publications and Synergistic Activities. Save the information in a single
file and attach here. This is required information.
Biographical sketches should follow the format described below
.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-72
Field Name Instructions
Attach Current & Pending
Support
Provide a list of all current and pending support for the PD/PI (even if
they receive no salary support from the project(s)) for ongoing projects
and pending applications. Show the total award amount for the entire
award period (including indirect costs) as well as the number of person-
months per year to be devoted to the project by the senior/key person,
regardless of source of support. Concurrent submission of an application
to other organizations for simultaneous consideration will not prejudice
its review.
Unless otherwise required in a specific FOA, do not use this
attachment upload for NIH and other PHS agency submissions.
This information is no longer required at the time of application
submission. This information may be requested later in the pre-
award cycle. When this occurs, you will be instructed to refer to
Other Support
in Supplemental Instructions Part III, Policies,
Assurances, Definitions and Other Information.
Profile – Senior/Key Person [n]
The remaining senior/key person profiles should be listed in alphabetical order. While
alphabetical order is preferred, it is not required. However, be aware that these profiles will
appear in the application in the order provided by the applicant. Therefore, peer reviewers will
see them in the order presented. Those with a postdoctoral role should be included if they meet
the definition of senior/key personnel. Also use this section to list any Other Significant
Contributors (OSCs). OSCs should be listed after all senior/key persons. OSCs are individuals
who have committed to contribute to the scientific development or execution of the project, but
are not committing any specified measurable effort (in person months) to the project. These
individuals are typically presented at “effort of zero person months” or “as needed” (individuals
with measurable effort cannot be listed as Other Significant Contributors). Consultants should be
included if they meet this definition.
A biosketch, including Research Support information, will be required for these individuals as
this highlights their accomplishments as scientists. Reviewers use these pages to address the
“investigator” review criterion. However, if an award is to be made, Other Support information
will not be required or accepted since considerations of overlap do not apply to these individuals.
Should the level of involvement change for an individual listed as an OSC, the individual should
be redesignated as “senior/key personnel.” This change should be made before any compensation
is charged to the project.
After providing data for each individual senior/key person, click the Next Person button at the
bottom of the form to enter data for the next senior/key person. Continue in this manner until data
has been provided for up to 100 senior/key persons. To ensure proper performance of this form,
after adding 20 additional senior/key persons please save your application, close the Adobe
reader, and reopen it. For applications involving more than 100 senior/key persons, the
“Additional Senior/Key Person Profiles” fields will become available once data for the first 100
senior/key persons has been provided.
Field Name Instructions

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-73
Field Name Instructions
Prefix Enter the prefix (e.g., Mr., Mrs., Rev.) for the name of the Senior/Key
Person.
First Name Enter the first (given) name of the Senior/Key Person. This field is
required.
Middle Name Enter the middle name of the Senior/Key Person, if applicable.
Last Name Enter the last (family) name of the Senior/Key Person. This field is
required.
Suffix Enter the suffix (e.g., Jr., Sr., Ph.D.) for the name of the Senior/Key
Person.
Position/Title Enter the title of the Senior/Key Person.
Department Enter the name of primary organizational department, service, laboratory,
or equivalent level within the organization of the Senior/Key Person.
Organization Name Enter the name of organization of the Senior/Key Person.
This is a required field for applications submitted to NIH and
other PHS agencies.
Division Enter the name of primary organizational division, office, or major
subdivision of the Senior/Key Person.
Street1 Enter first line of the street address for the Senior/Key Person in the
"Street 1" field. This field is required.
Street2 Enter second line of the street address for the Senior/Key Person in the
"Street 2" field. This field is optional.
City City for address of Senior/Key Person. This field is required
County/Parish County/ Parish for address of Senior/Key Person.
State Enter the State where the Senior/Key Person is located. This field is
required if the senior/key person is located in the United States.
Province Enter the Province where the Senior/Key Person is located.
If “Country” is not Canada, please leave blank.
Country Select the country for the Senior/Key Person address. This field is
required.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-74
Field Name Instructions
ZIP Code Enter the Postal Code (e.g., ZIP code) of Senior/Key Person. This field is
required if the Senior/Key Person is located in the United States.
A nine-digit Zip code is required.
Phone Number Enter the daytime telephone number for the Senior/Key Person. This field
is required.
Fax Number Enter the fax number for the Senior/Key Person.
E-mail Enter the e-mail address for the Senior/Key Person. This field is required
for the Senior/Key Person.
Credential, e.g., agency
login
If you are submitting to an agency (e.g., NIH) where you have an
established personal profile, enter the agency ID. If not, leave blank.
For NIH and other PHS agencies, registration in the eRA
Commons for all PD/PIs is required. The assigned Commons
UserName (the unique name used to log into the system) for
anyone assigned the PD/PI role must be entered here and
must have the PI role in eRA Commons. This is a required
field for applications submitted to NIH and other PHS
agencies. Applications will not pass agency validation
requirements without this field.
Note for applications reflecting Multiple PD/PIs, the Commons
UserName must be provided for all individuals assigned the
PD/PI Role on the application.
Project Role Select one. Use "Other" if a category is not listed in the pick list.
For applications reflecting Multiple PD/PIs, all such individuals
must be assigned the PD/PI role, even those at organizations
other than the applicant organization. The role of “Co-PD/PI” is
not currently used by NIH and other PHS agencies. Assigning an
individual(s) the role of "Co-PD/PI" will not identify the
application as a Multiple PD/PI application. If applicants wish to
use a different role, select “Other” for the Project Role field and
then insert the appropriate role descriptor in the Other Project
Role Category field.
If including individuals classified as “Other Significant
Contributors (OSCs),” use the “Other” category and indicate
“Other Significant Contributor” as the role in the “Other Project
Role Category.” OSCs should be listed last after all other
senior/key persons have been listed.
Other Project Role
Category
Complete if you selected “Other Professional” or “Other” as a project
role; e.g., Engineer, Chemist.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-75
Field Name Instructions
Degree Type Enter the highest academic or professional degree or other credentials
(e.g., R.N.). This is optional information.
Degree Year Enter the year the highest degree or other credential was obtained. This is
optional information.
Applicants should ensure that their degree information is current
in their Commons Profile.
Attach Biographical Sketch Provide a biographical sketch for the Senior/Key Person. Recommended
information includes: Education and Training, Research and Professional
Experience, Collaborators and Affiliations (for conflicts of interest),
Publications and Synergistic Activities. Save the information in a single
file and attach here. This field is required.
Biographical sketches should follow the format described below
.
Attach Current & Pending
Support
Provide a list of all current and pending support for the senior/key person
(even if they receive no salary support from the project(s)) for ongoing
projects and pending proposals. Show the total award amount for the
entire award period (including indirect costs) as well as the number of
person-months per year to be devoted to the project by the senior/key
person, regardless of source of support. Concurrent submission of a
proposal to other organizations will not prejudice its review.
Unless otherwise required in a specific FOA, do not use this
attachment upload for NIH and other PHS agency submissions.
This information is no longer required at the time of application
submission. This information may be requested later in the pre-
award cycle. When this occurs, refer to Other Support
in
Supplemental Instructions Part III, Policies, Assurances,
Definitions, and Other Information.
Additional Senior/Key Person Profile(s)
If more than 99 senior/key person profiles are proposed, enter the information in a separate file and attach
it here.
A sample Additional Senior/Key Person Profiles format page for greater than 100 profiles is
found under “Additional Format Pages” at: http://grants.nih.gov/grants/funding/424/index.htm
.
Additional Biographical Sketch(es) (Senior/Key Person)
Provide a biographical sketch for each senior/key person. Recommended information includes: Education
and Training, Research and Professional Experience, Collaborators and Affiliations (for conflicts of
interest), Publications and Synergistic Activities. Save the information in a single file and attach here.
Biographical Sketches should follow the format described below
.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-76
Additional Current and Pending Support(s)
Provide a list of all current and pending support for the PD/PI and each senior/key person (even if they
receive no salary support from the project(s) for ongoing projects and pending proposals). Show the total
award amount for the entire award period (including indirect costs) as well as the number of person
months per year to be devoted to the project by the senior/key person, regardless of source of support.
Concurrent submission of a proposal to other organizations will not prejudice its review.
Unless otherwise required in a specific FOA, do not use this attachment upload for NIH and other
PHS agency submissions. This information is no longer required at the time of application
submission. This information may be requested later in the pre-award cycle. When this occurs,
refer to Other Support
in Supplemental Instructions Part III, Policies, Assurances, Definitions,
and Other Information.
Additional NIH and Other PHS Agencies Instructions for a Biographical Sketch
Use the sample format on the Biographical Sketch Format Page
to prepare this section for all (modular
and other) grant applications. Include biographical sketches of all senior/key personnel and Other
Significant Contributors. The Biographical Sketch may not exceed four pages per person. This 4-page
limit includes the table at the top of the first page. See the sample of a
completed Biographical Sketch. If
the individual is registered in the eRA Commons, include the Commons User Name. This data item is
required for the PD/PI but is currently optional for all other senior/key persons. In other federal forms this
information is referred to as “Credential, e.g., agency login.” For information on the eRA Commons, see
https://commons.era.nih.gov/commons/index.jsp.
Complete the educational block at the top of the format page beginning with baccalaureate or other initial
professional education, such as nursing, and include postdoctoral training, separately referencing
residency training when applicable. For each entry provide the name and location of the institution; the
degree received (if applicable); the month and year the degree was received, and the field of study. For
residency entries, the field of study section should reflect the area of residency.
Following the educational block, complete sections A, B, C, and D as described below.
A. Personal Statement. Briefly describe why your experience and qualifications make you particularly
well-suited for your role (e.g., PD/PI, mentor, participating faculty) in the project that is the subject
of the application. Within this section you may, if you choose, briefly describe factors such as family
care responsibilities, illness, disability, and active duty military service that may have affected your
scientific advancement or productivity.
B. Positions and Honors. List in chronological order previous positions, concluding with your present
position. List any honors. Include present membership on any Federal Government public advisory
committee.
C. Selected Peer-reviewed Publication and Patent Citations. NIH encourages applicants to limit the
list of selected peer-reviewed publications, manuscripts in press, and patent citations to no more than
15. Do not include manuscripts submitted or in preparation. The individual may choose to include
selected publications based on recency, importance to the field, and/or relevance to the proposed
research. When citing articles that fall under the Public Access Policy, were authored or co-authored
by the applicant and arose from NIH support, provide the NIH Manuscript Submission reference
number (e.g., NIHMS97531) or the PubMed Central (PMC) reference number (e.g., PMCID234567)
for each article. If the PMCID is not yet available because the Journal submits articles directly to
PMC on behalf of their authors, indicate “PMC Journal – In Process.” A list of these journals is
posted at: http://publicaccess.nih.gov/submit_process_journals.htm
. Citations that are not covered by
the Public Access Policy, but are publicly available in a free, online format may include URLs or

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-77
PubMed ID (PMID) numbers along with the full reference (note that copies of publicly available
publications are not acceptable as appendix material).
D. Research Support. List both selected ongoing and completed (during the last three years) research
projects (Federal or non-Federal support). Begin with the projects that are most relevant to the
research proposed in this application. Briefly indicate the overall goals of the projects and
responsibilities of the senior/key person identified on the Biographical Sketch. Do not include
number of person months or direct costs.
Don’t confuse “Research Support” with “Other Support.” Though they sound similar, these parts of
the application are very different. As part of the biosketch section of the application, “Research
Support” highlights your accomplishments, and those of your colleagues, as scientists. This
information will be used by the reviewers in the assessment of each individual’s qualifications for a
specific role in the proposed project, as well as to evaluate the overall qualifications of the research
team. In contrast, “Other Support” information is required for all applications that are selected to
receive grant awards. NIH staff will request complete and up-to-date “other support” information
from you after peer review. This information will be used to check that the proposed research has not
already been Federally-funded.
4.6 Selecting the Appropriate Budget Form
The application forms package associated with most NIH funding opportunities includes two
optional budget forms—(1) R&R Budget Form; and, (2) PHS 398 Modular Budget Form. NIH
applications will include either the R&R Budget Form or the PHS 398 Modular Budget Form, but
not both. (Note AHRQ does not accept modular budgets.)
To determine which budget form to use for NIH applications, consult the modular budget and
foreign grantee guidelines below. Additional guidance may also be provided in the specific
funding opportunity announcement.
Modular Budget Guidelines. Modular budgets are applicable to certain research grant
applications requesting $250,000 or less per year for direct costs. Note, consortium/contractual
F&A costs are not factored into the direct cost limit. Consortium F&A costs may be requested in
addition to the $250,000 limit. Modular budgets are simplified; therefore, detailed categorical
information is not to be submitted with the application. The modular budget is applicable only to
applications for R01, R03, R15, R21, and R34 and their corresponding cooperative agreement
activity codes.
Instructions for completing a Modular Budget Form can be found in Section 5.4. Instructions for
completing the R&R Budget Form are provided in the next section.
Foreign Grantee Budget Guidelines. All competing (new, renewal, resubmission, and revision)
grant applications from foreign (non-U.S.) institutions must include only detailed (non-modular)
budgets. For additional information, see NIH Guide Notice NOT-OD-06-096,
http://grants.nih.gov/grants/guide/notice-files/NOT-OD-06-096.html.
Applications from foreign (non-U.S.) institutions submitted via Grants.gov must follow the
Research & Related Budget Form instructions and must complete and submit the Research &
Related Budget forms. Applications from foreign organizations must request budgets in U.S.
dollars.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-78
4.7 R&R Budget Form
The R&R Budget form includes three separate data entry screens: (1) Sections A and B; (2) Sections C
through E; and (3) Sections F through K. To navigate between the various screens, use the Previous and
Next buttons at the top of the form or use the scroll bar on the side of the screen. Complete the R&R
Budget form following the instructions provided. You must complete a separate detailed budget for each
year of support requested. The form will generate a cumulative budget for the total project period. You
must complete all the required information (i.e., those fields that are highlighted and outlined in red)
before the Next Period button is activated. If no funds are requested for a required field, enter “0.”
While the dollar fields allow cents to be entered, all dollar fields should be presented in whole numbers.
Please round to the nearest whole number.
NIH and other PHS agencies use the concept of person months as a metric for determining percent of
effort. To assist applicants unfamiliar with this concept, resources are available on the web at:
http://grants.nih.gov/grants/policy/person_months_faqs.htm
. Frequently asked questions and a conversion
calculator are available.
If funds are being requested for more than one budget period, click the Next Period button at the top of
the third budget screen (Sections F through K) to navigate to screens for the next budget period.
Revision (Supplemental) Application. For a “Revision” (Supplemental) application, show only those
items for which additional funds are requested. If the initial budget period of the supplementation
application is less than 12 months, prorate the personnel costs and other appropriate items of the detailed
budget.
4.7.1 Section A and B
Organizational DUNS

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-79
Enter the DUNS or DUNS+4 number of the applicant organization. For project applicant, this field is pre-
populated from the R&R SF424 Cover Page. For subaward applicants, this field is a required enterable
field.
Enter name of Organization
Pre-populated from the R&R SF424. Enter the name of the organization.
Budget Type
Project, Subaward/Consortium: Check the appropriate block. This field is required.
Project: The budget requested for the primary applicant organization.
Subaward/Consortium: The budget requested for subawardee/consortium organization(s). Note, separate
budgets are required only for subawardee/consortium organizations that perform a substantive portion of
the project.
If creating Subaward Budget, use the R&R Subaward Budget Attachment and attach as a separate file on
the R&R Budget Attachment(s) form.
If you are preparing an application that includes a subaward/consortium, see
Section 4.8 Special
Instructions for Preparing Applications with a Subaward/Consortium.
Start Date
Pre-populated from the R&R SF424. Enter the requested/proposed start date of each budget period. This
field is required.
End Date
Enter the requested/proposed end date of each budget period. This field is required.
Budget Period
Identify the specific budget period (for example, 1, 2, 3, 4, 5). If submitting through Grants.gov, the
system will automatically generate a cumulative budget for the total project period. This is a required
field.
(If the Reset Entries button is pressed, please navigate to previous year to enable the submission of the
form.)
A. Senior/Key Person
This section should include the names of all senior/key persons at the applicant organization who
are involved on the project in a particular budget year. Include all collaborating investigators, and
other individuals meeting the senior/key person definition if they are from the applicant
organization. Details of collaborators at other institutions will be provided in the Subaward
budget for each subaward/consortium organization. Personnel listed as Other Significant
Contributors who are not committing any specific measurable effort to the project should not be
included in the Personnel section of the budget since no associated salary and/or fringe benefits
should be requested for their contribution. Consultants designated as senior/key persons in the
Senior/Key Person Profile Form can be included in Budget Section A only if they are also
employees of the applicant organization. Otherwise, consultant costs should be included in
Consultant Services.
Field Name Instructions

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-80
Field Name Instructions
Prefix Enter the prefix (e.g., Mr., Mrs., Rev.) for the name of each Senior/Key
Person.
First Name Enter the first (given) name of each senior/key person. Senior/Key
Person.
Middle Name Enter the middle name of each Senior/Key Person, if applicable.
Last Name Enter the last (family) name of each Senior/Key Person. This field is
required.
Suffix Enter the suffix (e.g., Jr., Sr., PhD) of each Senior/Key Person.
Base Salary ($) Enter the annual compensation paid by the employer for each Senior/Key
Person. This includes all activities such as research, teaching, patient care,
or other. You may choose to leave this column blank.
An applicant organization may choose to leave this blank;
however, PHS staff will request this information prior to award.
Cal. Months Identify the number of months devoted to the project for each senior/key
person (i.e., calendar, academic, summer).
If effort does not change throughout the year, it is OK to use only
the calendar months column. However, you may use both
academic and summer months columns if your institutional
business process requires noting each separately even if effort
remains constant. If effort varies between academic and summer
months, leave the calendar months column blank and use only the
academic and summer months columns. Please use either
calendar months OR a combination of academic and summer
months. Some measurable effort is required for every Senior/Key
Person entry.
Acad. Months Identify the number of months devoted to the project for each senior/key
person (for example, calendar, academic, summer).
If your institution does not use a 9-month academic year, indicate
your institution’s definition of academic year in the budget
justification. Some measurable effort is required for every
Senior/Key Person entry.
Sum. Months Identify the number of months devoted to the project for each senior/key
person (for example, calendar, academic, summer).
If your institution does not use a 3-month summer period,
indicate your institution’s definition of summer in the budget
justification. Some measurable effort is required for every
Senior/Key Person entry.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-81
Field Name Instructions
Requested Salary ($) Regardless of the number of months being devoted to the project, indicate
only the amount of salary being requested for this budget period for each
senior/key person. This field is required.
Some PHS grant programs are currently subject to a legislatively
imposed salary limitation. Any adjustment for salary limits will
be made at the time of award; therefore requested salary should
be based on institutional base salary at the time the application is
submitted and not adjusted for any limitation. For guidance on
current salary limitations, see the Salary Cap Summary
on the
NIH grants website or contact your office of sponsored programs.
NIH grants also limit the compensation for graduate students.
Compensation includes salary or wages, fringe benefits and
tuition remission. While actual institutional-based compensation
should be requested and justified, this may be adjusted at the time
of the award. For more guidance on this policy, see:
http://grants.nih.gov/grants/ guide/notice-files/NOT-OD-02-
017.html.
Fringe Benefits ($) Enter applicable fringe benefits, if any, for each senior/key person.
Funds Requested ($) The requested salary and fringe benefits for each senior/key person. This
field is auto-calculated.
Project Role Identify the project role of each senior/key person in this section. This
section could also include such roles as Co-PD/PI, Postdoctoral
Associates, and Other Professionals.
Roles should correspond to the roles included on the Senior/Key
Person Profile (Expanded) Form.
Additional Senior/Key
Persons
If funds are requested for more than eight senior/key persons, include all
pertinent budget information as identified in this section and attach as a
file here. Enter the total funds requested for all additional senior/key
persons in line 9 of Section A. This attachment is required if funds are
entered in line 9 of Section A.
Use the same format as the budget form and include all required
information.
Total Funds requested for
all persons in the attached
file
Enter the total funds requested for all senior/key persons. This is required
information.
Total Senior/Key Persons The total funds requested for all senior/key persons.
Special Instructions: Joint University and Department of Veterans Affairs (V.A.)
Appointments
Individuals with joint university and V.A. appointments may request the university’s share of

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-82
their salary in proportion to the effort devoted to the research project. The individual’s salary with
the university determines the base for computing that request. Signature by the institutional
official on the application certifies that: (1) the individual is applying as part of a joint
appointment specified by a formal Memorandum of Understanding between the university and
the V.A.; and (2) there is no possibility of dual compensation for the same work, or of an actual
or apparent conflict of interest regarding such work. Additional information may be requested by
the awarding components.
B. Other Personnel
Field Name Instructions
Number of Personnel For each project role category identify the number of personnel proposed.
In most circumstances, the salaries of administrative or clerical staff at
educational institutions and nonprofit organizations are included as part of
indirect costs. Examples, however, of situations where direct charging of
administrative or clerical staff salaries may be appropriate may be found
at: http://www.whitehouse.gov/omb/circulars/a021/a21_2004.html#exc.
The circumstances for requiring direct charging of these services must be
clearly described in the budget justification.
For all Postdoctoral Associates and Graduate Students not already
named in Section A. Senior/Key Person, individually list names,
roles (e.g., PostDoc or Graduate Student), associated months, and
salary & fringe benefits requested in the Budget Justification.
Project Role For each project role category identify the number of personnel proposed.
List any additional project role(s) in the blank(s) provided, e.g., Engineer,
IT Professionals, etc.
Do not include consultants in this section. Consultants are
included below in Section F. Other Direct Costs.
Cal. Months Identify the number of months devoted to the project in the applicable box
for each project role category (i.e., calendar, academic, summer).
Acad. Months Identify the number of months devoted to the project in the applicable
box for each project role category (i.e., calendar, academic, summer).
If your institution does not use a 9-month academic year, indicate
your institution’s definition of academic year in the budget
justification.
Sum. Months Identify the number of months devoted to the project in the applicable
box for each project role category (i.e., calendar, academic, summer).
If your institution does not use a 3-month summer period,
indicate your institution’s definition of summer in the budget
justification.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-83
Field Name Instructions
Requested Salary ($) Regardless of the number of months being devoted to the project, indicate
only the amount of salary/wages being requested for each project role.
Some PHS grant programs are currently subject to a legislatively
imposed salary limitation. Any adjustment for salary limits will
be made at the time of award. For guidance on current salary
limitations, see the Salary Cap Summary
on the NIH grants
website or contact your office of sponsored programs.
NIH grants also limit the compensation for graduate students.
Compensation includes salary or wages, fringe benefits and
tuition remission. While actual institutional-based compensation
should be requested and justified, this may be adjusted at the time
of the award. For more guidance on this policy, see:
http://grants.nih.gov/grants/ guide/notice-files/NOT-OD-02-
017.html.
Fringe Benefits ($) Enter applicable fringe benefits, if any, for this project role category.
Funds Requested This field is auto-calculated.
Total Number of Other
Personnel
This total will auto-calculate. Total Number of Personnel.
Total Other Personnel Total Funds requested for all other Personnel.
Total Salary, Wages and
Fringe Benefits (A+B)
Total Funds requested for all Senior/Key persons and all Other Personnel.
This total will auto-calculate.
To navigate to the next page (Sections C through E), click the Next button at the top of the form or use
the scroll bar on the left-hand side of the screen.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-84
4.7.2 Sections C through E
The information for Organizational DUNS, Budget Type, Name of Organization, and Start and End Dates
is automatically filled in based on the information entered on the first budget screen. To edit this
information, return to the initial budget screen (Sections A and B) by clicking the Previous button.
C. Equipment Description
List of items and dollar amount for each item exceeding $5,000.
Field Name Instructions
Equipment Item Equipment is defined as an item of property that has an acquisition cost of
$5,000 or more (unless the organization has established lower levels) and
an expected service life of more than one year. List each item of
equipment separately and justify each in the budget justification section.
Allowable items ordinarily will be limited to research equipment and
apparatus not already available for the conduct of the work. General-
purpose equipment, such as a personal computer, is not eligible for
support unless primarily or exclusively used in the actual conduct of
scientific research.
Funds Requested List the estimated cost of each item of equipment including shipping and
any maintenance costs and agreements. This is required information.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-85
Field Name Instructions
Additional Equipment If this section cannot accommodate all the equipment proposed, attach a
file in the block provided. List each additional item and the funds
requested. For all additional items in the attached file, list the total funds
requested in the following field
Total funds requested for
all equipment listed in the
attached file
Total funds requested for all equipment listed in the attached file. Dollar
amount for each item should exceed $5000.
Total Equipment Total Funds requested for all equipment.
D. Travel
Field Name Instructions
Domestic Travel Costs
(Incl. Canada, Mexico, and
U.S. Possessions)
Identify the total funds requested for domestic travel. Domestic travel
includes Canada, Mexico, and U.S. possessions. In the budget
justification section, include the purpose, destination, dates of travel (if
known), and number of individuals for each trip. If the dates of travel are
not known, specify estimated length of trip (e.g., 3 days).
Foreign Travel Costs Identify the total funds requested for foreign travel. Foreign travel
includes any travel outside of North America and/or U.S. possessions. In
the budget justification section, include the purpose, destination, dates of
travel (if known) and number of individuals for each trip. If the dates of
travel are not known, specify estimated length of trip (e.g., 3 days).
Total Travel Cost Total Funds requested for all travel.
E. Participant/Trainee Support Costs
Unless specifically stated otherwise in an announcement, NIH and other PHS agencies applicants
should leave blank Section E. Note: Tuition remission for graduate students should continue to be
included in Section F. Other Direct Costs when applicable.
Field Name Instructions
Tuition/Fees/Health
Insurance
List total funds requested for Participant/Trainee Tuition / Fees / Health
insurance.
Stipends List total funds requested for Participant/Trainee stipends.
Travel List total funds requested for Participant/Trainee travel.
Subsistence List total funds requested for Participant/Trainee subsistence.
Other Describe any other participant trainee funds requested. List total funds
requested for any other Participant/Trainee costs described.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-86
Field Name Instructions
Number of
Participants/Trainees
List total number of proposed Participants/Trainees. Value cannot be
greater than 999.
Total Participant/Trainee
Support Costs
Total Funds requested for all trainee costs. This field is required if any
data has been entered in section E.
4.7.3 Sections F through K
The information for Organizational DUNS, Budget Type, Name of Organization, and Start and End Dates
is automatically filled in based on the information entered on the first budget screen. To edit this
information, return to the initial budget screen (Sections A and B) by clicking the Previous button.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-87
Special Instructions: Foreign Organizations (Non-domestic [non-U.S. Entities])
Foreign institutions and international organizations may request funds for limited F&A costs (8
percent of modified total direct costs less equipment) to support the costs of compliance with
DHHS and NIH requirements including, but not limited to, protection of human subjects, animal
welfare, invention reporting, financial conflict of interest and research misconduct.
Foreign organizations may not include any charge-back of customs and import fees, such as
consular fees, customs surtax, value-added taxes (VAT) and other related charges.
F. Other Direct Costs
Field Name Instructions
1. Materials and Supplies List total funds requested for materials and supplies. In the budget
justification, indicate general categories such as glassware, chemicals,
animal costs, including an amount for each category. Categories less than
$1,000 are not required to be itemized.
2. Publication Costs List the total publication funds requested. The proposal budget may
request funds for the costs of documenting, preparing, publishing, or
otherwise making available to others the findings and products of the
work conducted under the award. In the budget justification include
supporting information.
3. Consultant Services List the total costs for all consultant services. In the budget justification,
identify each consultant, the services he/she will perform, total number of
days, travel costs, and the total estimated costs.
In the budget justification also provide the names and
organizational affiliations of all consultants, other than those
involved in consortium/contractual arrangements. Include
consultant physicians in connection with patient care and persons
who are confirmed to serve on external monitoring boards or
advisory committees to the project. Describe the services to be
performed.
4. ADP/Computer Services List total funds requested for ADP/computer services. The cost of
computer services, including computer-based retrieval of scientific,
technical and education information may be requested. In the budget
justification, include the established computer service rates at the
proposing organization if applicable.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-88
Field Name Instructions
5. Subawards/Consortium/
Contractual Costs
List total funds requested for 1) all subaward/consortium organization(s)
proposed for the project and 2) any other contractual costs proposed for
the project.
This line item should include both direct and indirect costs for all
subaward/consortium organizations. Contractual costs for support
services, such as the laboratory testing of biological materials,
clinical services, or data processing, are occasionally sufficiently
high to warrant a categorical breakdown of costs. When this is the
case, provide detailed information as part of the budget
justification.
NIH policy provides for exclusion of consortium/contractual
F&A costs when determining if an applicant is in compliance
with a direct cost limitation. Please see the
Supplemental
Instructions, Part III, Section 1.1 for additional information
regarding this exclusion Policy
6. Equipment or Facility
Rental/User Fees
List total funds requested for equipment or facility Rental/Use fees. In the
budget justification, identify each rental user fee and justify.
7. Alterations and
Renovations
List total funds requested for alterations and renovations. In the budget
justification, itemize by category and justify the costs of alterations and
renovations including repairs, painting, removal or installation of
partitions, shielding, or air conditioning. Where applicable, provide the
square footage and costs.
Under certain circumstances the public policy requirements that
apply to construction activities may also apply to A&R activities.
Please refer to the NIH Grants Policy Statement section on
“
Construction Grants – Public Policy Requirements and
Objectives” for more information.
Note, costs for any Alterations and Renovations (A&R) were
previously unallowable on applications from foreign institutions,
international organizations and domestic applications with foreign
subawards. However, an HHS policy change now allows for
minor A&R (≤$500,000) on these applications.
When requesting minor A&R costs under this policy, please
provide detailed information on the planned A&R in the budget
justification.
8-10 Other Add text to describe any “other” direct costs not requested above. Use the
budget justification to further itemize and justify.
List total funds requested for items 8-10 “Other.”
Use lines 8-10 for such costs as patient care and tuition remission.
If requesting patient care costs, request inpatient and outpatient
costs separately using lines 8 and 9.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-89
Field Name Instructions
Total Other Direct Costs Total Funds requested for all other direct costs.
Special Instructions for Patient Care Costs
If inpatient and/or outpatient costs are requested, provide the names of any hospitals and/or
clinics and the amounts requested for each in the budget justification.
State whether each hospital or clinic has a currently effective DHHS-negotiated research patient
care rate agreement and, if not, what basis is used for calculating costs. If an applicant does not
have a DHHS-negotiated rate, the PHS awarding component can approve a provisional rate.
Indicate, in detail, the basis for estimating costs in this category, including the number of patient
days, estimated cost per day, and cost per test or treatment. If both inpatient and outpatient costs
are requested, provide information for each separately. If multiple sites are to be used, provide
detailed information by site.
Include information regarding projected patient accrual for the project/budget periods and relate
this information to the budget request for patient care costs. If patient accrual is anticipated to be
lower at the start or during the course of the project, plan budget(s) accordingly.
Provide specific information regarding anticipated sources of Other Support for patient care costs,
e.g., third party recovery or pharmaceutical companies. Include any potential or expected
utilization of General Clinical Research Centers.
G. Total Direct Costs (A through F)
Total Funds requested for all direct costs.
H. Indirect Costs
Field Name Instructions
Indirect Cost Type Indicate the type of cost e.g.; Salary & Wages, Modified Total Direct
Costs, or Other explain. Also indicate if Off-site. If more than one
rate/base is involved, use separate lines for each. If you do not have a
current indirect rate(s) approved by a Federal agency, indicate, “None—
will negotiate” and include information for a proposed rate. Use the
budget justification if additional space is needed.
Indirect Cost Rate (%) Indicate the most recent indirect cost rate(s) (also known as Facilities &
Administrative Costs [F&A]) established with the cognizant Federal
office, or in the case of for-profit organizations, the rate(s) established
with the appropriate agency. If you have a cognizant/oversight agency and
are selected for an award, you must submit your indirect rate proposal to
that office for approval. If you do not have a cognizant/oversight agency,
contact the awarding agency.
If this field does not allow a figure greater than 100% to be
entered, use two lines to show the entire calculation. This field
should be entered using a rate such as “55.5.”
Indirect Cost Base ($) Enter the amount of the base for each indirect cost type.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-90
Field Name Instructions
Funds Requested Enter funds requested for each indirect cost type.
Total Indirect Costs Total Funds requested for indirect costs.
Cognizant Federal Agency Enter the name of the cognizant Federal Agency, name and phone number
of the individual responsible for negotiating your rate. If no cognizant
agency is known, enter “None.”
I. Total Direct and Indirect Institutional Costs (G + H)
Total Funds requested for direct and indirect costs.
J. Fee
Generally, a fee is not allowed on a grant or cooperative agreement. Do not include a fee in your budget,
unless the program announcement specifically allows the inclusion of a “fee” (e.g., SBIR/STTR). If a fee
is allowable, enter the requested fee.
K. Budget Justification
Use the budget justification to provide the additional information requested in each budget category
identified above and any other information the applicant wishes to submit to support the budget request.
The following budget categories must be justified, where applicable: equipment, travel, participant/trainee
support and other direct cost categories. Only one file may be attached. The attachment is required.
Use this section to list the names, role (e.g., PostDoc or Graduate Student), associated months,
salary and fringe benefits for all Postdoctoral Associates and Graduate Students included in
Budget Section B. Other Personnel.
Include a justification for any significant increases or decreases from the initial year budget.
Justify budgets with more than a standard escalation from the initial to the future year(s) of
support. Also use this section to explain any exclusions applied to the F&A base calculation.
If the application includes a subaward/consortium budget, a separate budget justification is
submitted for that budget. See
Section 4.8 Special Instructions for Preparing Applications with a
Subaward/Consortium.
Completing Budget Periods 2-5
If funds are being requested for more than one budget period, you must complete a separate detailed
budget for each year of support requested. To navigate to screens for the next budget period, click the
Next Period button at the top of the 3
rd
budget screen (Sections F through K). You must complete all the
required information (i.e., those fields that are highlighted and outlined in red) before the Next Period
button is activated. If no funds are requested for a required field, enter “0.” Note the Budget Justification
is also a required item and must be attached before the Next Period button is activated.
Supplemental/Revision Application
For a supplemental/revision application, show only those items for which additional funds are
requested. If the initial budget period of the supplemental/revision application is less than 12
months, prorate the personnel costs and other appropriate items of the detailed budget.
Submitting Budgets With More Than 5 Budget Periods
When authorized or requested by the appropriate NIH IC, applicants may submit applications

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-91
with more than 5 budget periods. In these situations complete the detailed budget for periods 1-5
as usual. However, include the same level of detail for Period 6 in the Budget Justification along
with an explanation of the situation. Also, be sure to include a cover letter that addresses these
extra budget periods, and include the IC Program Official’s preapproval as part of the Cover
Letter PDF.
4.7.4 Cumulative Budget
All values on this form are calculated automatically. They present the summations of the amounts that
you have entered previously, under Sections A through K, for each of the individual budget periods.
Therefore, no data entry is allowed or required, in order to complete this “Cumulative Budget” section.
If any of the amounts displayed on this form appears to be incorrect, you may correct it by adjusting one
or more of the values that contribute to that total. To make any such adjustments, you will need to revisit
the appropriate budget period form(s), to enter corrected values.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-92

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-93
4.8 Special Instructions for Preparing Applications with a
Subaward/Consortium

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-94

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-95
A complete subaward/consortium budget form (including the budget justification section) should be
completed by each consortium grantee organization. Separate budgets are required only for
subawardee/consortium organizations that perform a substantive portion of the project.
Note, a complete subaward/consortium budget form is only required when the prime grantee is submitting
a detailed budget using the R&R Budget Form. Do not use this subaward/consortium budget form for
applications using the PHS 398 Modular Budget Form. Applicants using the Modular Budget Form
should see Section 5.4 for instructions concerning information on consortium budgets.
For any subaward or consortium sites, it is appropriate and expected that someone may be designated as
the consortium lead investigator responsible for ensuring proper conduct of the project or program at that
site. However, when completing the Project Role for the consortium lead investigator, the project role of
“PD/PI” should only be used if the entire application is being submitted under the Multiple PI policy.
Otherwise, this individual should be assigned some other project role in the senior/key personnel section
of the application. Also, the role of Co-PD/PI is not currently used by NIH and other PHS agencies.
Assigning an individual(s) the role of "Co-PD/PI" will not identify the application as a Multiple PD/PI
application. Although NIH now recognizes the role of “Co-Investigator,” if applicants wish to use the role
of “Consortium PI” or some other similar role, select “Other” for the Project Role field and then insert the
appropriate role descriptor in the Other Project Role Category field.
NIH continues to support the policy established in April 2004, (revised in November 2004) regarding
applications that involve consortium/contractual F&A costs (See NOT-OD-05-004
). This policy allows
applicants to exclude consortium/contractual F&A costs when determining compliance for any
application where a direct cost limit applies. The use of the SF424 (R&R) application with separately
submitted subaward/consortium budgets allows NIH to take advantage of a system validation for this
policy. When an application is submitted in response to a program with a direct cost limit, the eRA
system will perform the calculation by taking the total direct costs requested by the prime/parent
organization in their detailed budget, and subtracting all subaward/consortium F&A from each and every
subaward budget attached. When the validation calculation equals or exceeds the respective direct cost
limit, the application will receive a warning. There are circumstances, when the system does not have
sufficient information to exclude all allowable F&A costs. Applicants should document in their budget
justification, how their budget falls below the direct cost limit.
This form accommodates a set number of separate subaward budgets (30). If you are submitting an
application with more subaward budgets than the form allows, the remaining budgets should be converted
to PDF and included as part of Section K. Budget Justification of the parent budget. Reminder, the sum of
all subaward budgets; e.g., those attached separately and those provided as part of the budget justification,
must be included in Line F.5 Subawards/Consortium/Contractual Costs of the parent budget.
To start the process, the applicant organization should:
• Select the Subaward Budget Attachment Form from the Optional Documents in the Grant
Application Package.
• Open the form, and click the Click here to extract the R&R Subaward Budget Attachment
button in the middle of the form. A “SAVE” dialog box appears.
• Save the file locally using the first ten letters of the consortium organization’s name and use
“.pdf” as the file extension. (The extracted file is an Adobe PDF file.) Once you have saved the
file there is no need to extract another budget attachment. Doing so may cause you to lose any
data already stored in the saved file.
• E-mail the extracted, saved form to the consortium grantee. Note: consortium grantees must have
installed a compatible version of Adobe Reader before they can complete the form. The
consortium grantee should complete all the budget information as instructed in the R&R Budget

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-96
form instructions in Section 4.7. The Budget Type should be set to Subaward/Consortium.
Organizational DUNS and Name of Organization fields must reflect that of the
subaward/consortium grantee.
• The consortium grantee must complete the budget form and e-mail it back to the applicant
organization.
• Return to the Subaward Budget Attachment Form and attach the consortium grantee’s budget to
one of the blocks provided on the form.
Submitting Subaward Budgets that are not Active for all Periods of the Prime Grant
When submitting subaward budgets that are not active for all periods of the prime grant, fill out the
subaward R&R Budget form and include only the number of periods for which the subaward is active.
The budget period start/end dates reflected in each period should reflect the corresponding prime budget
period start/end dates. This approach is the most workable solution to the limitations in existing forms
that do not allow an “empty” budget period and do not allow submission of a subaward budget with zero
effort to skip a budget period.
For example, suppose the prime has filled out a budget form with the following periods:
• period 1 Jan 1, 2013 – Dec 31, 2013
• period 2 Jan 1, 2014 – Dec 31, 2014
• period 3 Jan 1, 2015 – Dec 31, 2015
• period 4 Jan 1, 2016 – Dec 31, 2016
• period 5 Jan 1, 2017 – Dec 31, 2017
Now, suppose there is a subaward that performs in support year 1 and does not become active again until
support year 4. The subaward can fill out the first two periods of their budget form as follows:
• period 1 Jan 1, 2013 – Dec 31, 2013 (dates correspond to prime period 1)
• period 2 Jan 1, 2016 – Dec 31, 2016 (dates correspond to prime period 4)
It is not necessary that the budget period numbers between the prime and subaward match; the correlation
is reflected in the dates. Do be careful, however, that the dates exactly match what is listed for the period
in the prime budget.
Note this approach may cause a validation warning regarding the NIH $500,000 per year limit on direct
costs, therefore you should document in both the cover letter and the subaward budget justification that
the subaward is only active for specific periods of the prime. Appropriate NIH staff has access to the
cover letter and reviewers have access to the budget justification. This documentation will make the date
correlation immediately apparent and will help avoid any confusion.
Once all data have been entered use the scroll bar to scroll up. You will be returned to the Grant
Application Package screen. To remove a form from your application, uncheck the box next to the form
name in the Optional document section.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-97
5. Completing PHS 398 Form
5.1 Overview
In conjunction with the SF424 (R&R) forms, NIH and other PHS agencies grants applicants should also
complete and submit additional forms titled “PHS 398.” Note the PHS 398 forms include additional data
required by the agency for a complete application. While these are not identical to the PHS 398
application form pages, the PHS 398 reference is used to distinguish these additional data requirements
from the data collected in the SF424 (R&R) forms. A complete application to NIH and other PHS
agencies will include SF424 (R&R) and PHS 398 forms. The PHS 398 forms include:
• PHS 398 Cover Page Supplement (this supplements the data requirements in the R&R Cover
form)
• PHS 398 Modular Budget Form (use only when a modular budget is submitted instead of a
detailed budget)
• PHS 398 Research Plan Form
• Planned Enrollment Report
• PHS 398 Cumulative Inclusion Enrollment Report
Complete each form using the instructions provided below.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-98
5.2 (Reserved)
5.3 Cover Page Supplement Form

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-99
1. Program Director/Principal Investigator (PD/PI)
Field Name Instructions
Prefix Pre-populated from the SF424 (R&R). The prefix (for example, Mr.,
Mrs., Rev.) for the name of the PD/PI.
First Name Pre-populated from the SF424 (R&R). The first (given) name of the
PD/PI. This field is required.
Middle Name Pre-populated from the SF424 (R&R). The middle name of the PD/PI.
Last Name Pre-populated from the SF424 (R&R). The last (family) name of the
PD/PI. This field is required.
Suffix Pre-populated from the SF424 (R&R). The suffix (for example, Jr., Sr.,
PhD) for the name of the PD/PI.
2. Human Subjects
Field Name Instructions
Clinical Trial? Check "yes" or "no" to indicate whether the project includes a clinical
trial. See Supplemental Grant Application Instructions, Part III.3.
for the
specific definition.
Agency-Defined Phase III
Clinical Trial
Check “Yes” or “No” to indicate whether the project is an NIH-defined
Phase III clinical trial.
An NIH-defined Phase III clinical trial is a broadly based prospective
Phase III clinical investigation, usually involving several hundred or more
human subjects, for the purpose of either evaluating an experimental
intervention in comparison with a standard or control intervention or of
comparing two or more existing treatments. Often the aim of such
investigation is to provide evidence leading to a scientific basis for
consideration of a change in health policy or standard of care. The
definition includes pharmacologic, non-pharmacologic, and behavioral
interventions given for disease prevention, prophylaxis, diagnosis, or
therapy. Community trials and other population-based intervention trials
are also included.
3. *Disclosure Permission Statement
Field Name Instructions

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-100
Field Name Instructions
If this application does not
result in an award, is the
Government permitted to
disclose the title of your
proposed project, and the
name, address, telephone
number and e-mail address
of the official signing for
the applicant organization,
to organizations that may
be interested in contacting
you for further information
(e.g., possible
collaborations,
investment)?
Select "yes" or "no" to indicate whether disclosure permission is granted.
This field is required.
Your response will not affect any peer review or funding decisions.
4. Program Income
Field Name Instructions
Is program income
anticipated during the
periods for which the grant
support is requested?
If program income is anticipated during the periods for which the grant
support is requested, check “Yes,” and then complete the section below. If
no program income is anticipated, check “No” and leave the following
section blank.
Budget Period If program income is anticipated, enter the budget periods in this column.
If the application is funded, the Notice of Grant Award will provide
specific instructions regarding the use of such income.
Anticipated Amount ($) If program income is anticipated, enter the amount anticipated for each
budget period listed.
Source(s) If program income is anticipated, enter the source for each budget period
listed.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-101
5. Human Embryonic Stem Cells
Field Name Instructions
Does the proposed project
involve human embryonic
stem cells?
If the proposed project involves human embryonic stem cells, check Yes
and complete the section below.
If the proposed project does not involve human embryonic stem cells,
check No.
Specific stem cell line
cannot be referenced at this
time. One from the registry
will be used.
If a specific line cannot be referenced at the time of application
submission, check this box. Additionally, provide a strong justification for
why an appropriate cell line is not available from the Registry at this time.
The justification should be included as part of the Research Strategy or
Program Plan as appropriate..
Cell Line(s) List in this section the registration number of the specific cell line(s) from
the NIH Human Embryonic Stem Cell Registry

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-102
6. Inventions and Patents (For renewal applications only)
Field Name Instructions
Inventions and Patents This block need only be completed if submitting an R&R “Renewal”
application or a Resubmission of a Renewal application. If no inventions
were conceived or reduced to practice during the course of work under
this project, check “No.” The remaining parts of the item are then not
applicable. If any inventions were conceived or reduced to practice during
the previous period of support, check “Yes.”
Note: NIH recipient organizations must promptly report inventions to the
Extramural Inventions and Technology Resources Branch of the Office of
Policy for Extramural Research Administration, OER, NIH, Bethesda,
MD 20892-2750, (301) 435-1986. Invention reporting compliance
according to regulations at 37 CFR 401.14 is described at
http://www.iedison.gov
. The grantee is encouraged to submit reports
electronically using Interagency Edison (http://www.iedison.gov).
Previously Reported If the item above is checked "Yes", indicate whether this information has
been reported previously to the PHS or to the applicant organization
official responsible for patent matters.
7. Change of Investigator/Change of Institution Questions
Field Name Instructions

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-103
Field Name Instructions
Change of Program
Director/Principal
Investigator
Check here, if this application reflects a change in principal
investigator/program director from that indicated on a previous
application. This is not generally applicable to a "New" application
Prefix If this application reflects a change in PD/PI, enter the name prefix (for
example, Mr., Mrs., Rev.) of the former PD/PI.
First Name If this application reflects a change in PD/PI, enter the first name of the
former PD/PI.
Middle Name If this application reflects a change in PD/PI, enter the middle name of the
former PD/PI.
Last Name If this application reflects a change in PD/PI, enter the last name of the
former PD/PI.
Suffix If this application reflects a change in PD/PI, provide the suffix (for
example, Jr., Sr., PhD) of the former PD/PI.
Change of Grantee
Institution
Check here, if this application reflects a change in grantee institution from
that indicated on a previous application. This is not generally applicable
to a "New" application.
Name of Former Institution If this application reflects a change in grantee institution, insert the name
of the former institution here.
5.4 Modular Budget Form
Selecting the Appropriate Budget Form
The application forms package associated with most NIH funding opportunities includes two optional
budget forms—(1) R&R Budget Form; and, (2) PHS 398 Modular Budget Form. NIH applications will
include either the R&R Budget Form or the PHS 398 Modular Budget Form but not both. (Note AHRQ
does not accept modular budgets.)
To determine which budget form to use for NIH applications, consult the modular budget guidelines
below. Additional guidance may also be provided in the specific funding opportunity announcement.
Modular Budget Guidelines
Modular budgets are applicable to certain research grant applications from domestic organizations
requesting $250,000 or less per year for direct costs. International organizations and others that do not fall
under this definition should use the detailed budget forms described in Section 4.7. Note,
consortium/contractual F&A costs are not factored into the direct cost limit. They may be requested in
addition to the $250,000 limit. Modular budgets are simplified; therefore, detailed categorical information
is not to be submitted with the application.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-104
For all modular budgets, request total direct costs (in modules of $25,000), reflecting appropriate support
for the project. There will be no future year escalations. A typical modular grant application will request
the same number of modules in each year. Provide an additional narrative budget justification for any
variation in the number of modules requested.
NIH may request (prior to award) additional budget justification in exceptional circumstances. For further
information, see http://grants.nih.gov/grants/funding/modular/modular.htm
and
http://grants.nih.gov/grants/funding/modular/modular_review.htm.
Using the Modular Budget Form
The Modular Budget Form provides budget fields for up to 5 years of support (e.g., budget periods 1 - 5).
If requesting less than 5 years of support, complete only those years requested and leave the others blank.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-105
5.4.1 Budget Period Form

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-106
NOTE: The form allows for up to five budget periods followed by a cumulative budget for all budget
periods. The fields are the same for all budget periods. The following instructions can be used for each.
Budget Period
Field Name Instructions
Start Date Enter the requested/proposed start date of the budget period. Use the
following format: MM/DD/YYY.
End Date Enter the requested/proposed end date of the budget period. Use the
following format: MM/DD/YYY.
A. Direct Costs
Field Name Instructions
Direct Cost less
Consortium F&A
Enter the amount of Direct Costs, less actual consortium F&A costs for
this budget period. This figure must be in $25,000 increments, and it may
not exceed $250,000. Any consortium F&A costs are excluded from this
figure. This field is required.
Consortium F&A If this project involves a consortium, enter the actual consortium F&A
costs for this budget period. If this project does not involve a consortium,
leave blank.
Total Direct Costs This field auto-calculates.
B. Indirect Costs
Field Name Instructions
Indirect Cost Type Indicate the type of base (for example, Salary & Wages, Modified Total
Direct Costs, Other [explain]), and indicate if Off-site. If more than one
rate/base is involved, use separate lines for each. If you do not have a
current indirect rate(s) approved by a Federal agency, indicate, “None—
will negotiate” and include information for a proposed rate. Use the
budget justification if additional space is needed.
Indirect Cost Rate (%) Indicate the most recent Indirect Cost rate(s) (also known as Facilities &
Administrative Costs [F&A]) established with the cognizant Federal
office, or in the case of for-profit organizations, the rate(s) established
with the appropriate agency. If you have a cognizant/oversight agency and
are selected for an award, you must submit your indirect rate proposal to
that office for approval. If you do not have a cognizant/oversight agency,
contact the awarding agency.
Indirect Cost Base ($) Enter the amount of the base for each indirect cost type.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-107
Field Name Instructions
Funds Requested ($) Enter funds requested for each indirect cost type.
Cognizant Agency (Agency
Name, POC Name and
Phone Number)
Enter the name of the cognizant Federal Agency, name, and phone
number of the individual responsible for negotiating your rate. If no
cognizant agency is known, enter “None.”
Indirect Cost Rate
Agreement Date
If you have a negotiated rate agreement, enter the agreement date.
Total Indirect Costs This field auto-calculates.
C. Total Direct and Indirect Costs (A+B) Funds Requested ($)
The total funds requested for direct and indirect costs. This field is required.
Once you have entered all required information for budget period 1, press the Next Period button to enter
information for the subsequent budget period.
5.4.2 Cumulative Budget Information
Cumulative Budget Information
All values for the Cumulative Budget Information are calculated automatically. They equal the
summations of the amounts that you have entered previously for each of the individual budget periods.
Therefore, no data entry is allowed or required, in order to complete this “Cumulative Budget” section.
If any of the amounts displayed on this form appears to be incorrect, you may correct it by adjusting one
or more of the values that contribute to that total. To make any such adjustments, you will need to revisit
the appropriate budget period form(s), to enter corrected values.
Modular Budget Justifications
Field Name Instructions
Personnel Justification List all personnel, including names, percent of effort and roles on the
project. No individual salary information should be provided. Since the
modules should be a reasonable estimate of costs allowable, allocable,
and appropriate for the proposed project, applicants must use the current
legislatively imposed salary limitation when estimating the number of
modules. For guidance on current salary limitations contact your office of
sponsored programs.
NIH grants also limit the compensation for graduate students.
Compensation includes salary or wages, fringe benefits and tuition
remission. This limit should also be used when estimating the number of
modules. See:
http://grants1.nih.gov/grants/guide/notice-files/NOT-OD-
02-017.html
Save the information in a single file and click the add attachment button

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-108
Field Name Instructions
to complete this entry.
Consortium Justification Provide an estimate of total costs (direct plus Facilities and
Administrative) for each year, rounded to the nearest $1,000. List the
individuals/ organizations with whom consortium or contractual
arrangements have been made. List all personnel, including percent of
effort and roles on the project. No individual salary information should be
provided. Indicate whether the collaborating institution is foreign or
domestic. While only the direct cost for a consortium/contractual
arrangement is factored into eligibility for using the modular budget
format, the total consortium/contractual costs must be included in the
overall requested modular direct cost amount.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
Additional Narrative
Justification
If the requested budget requires any additional justification; e.g, variations
in the number of modules requested, save the information in a single file
and click the add attachment button to complete this entry.
Once all data have been entered use the scroll bar to scroll up. You will be returned to the Grant
Application Package screen. To remove a form from your application, uncheck the box next to the form
name in the Optional document section.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-109
5.5 PHS 398 Research Plan Form
The Research Plan should include sufficient information needed for evaluation of the project, independent
of any other document (e.g., previous application). Be specific and informative, and avoid redundancies.
Research Plan Attachments (See also Section 2.3.2 Creating PDFs for Text Attachments
)
Text attachments should be generated using word processing software and then converted to PDF using
PDF generating software. Avoid scanning text attachments to convert to PDF since that causes problems
for the agency handling the application. In addition, be sure to save files with descriptive file names.
Do not include any information in a header or footer of the attachments. A header will be system-
generated that references the name of the PD/PI. Page numbers for the footer will be system-generated in
the complete application, with all pages sequentially numbered.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-110
Since a number of reviewers will be reviewing applications as an electronic document and not a paper
version, applicants are strongly encouraged to use only a standard, single-column format for the text.
Avoid using a two-column format since it can cause difficulties when reviewing the document
electronically.
Full-sized glossy photographs of material such as electron micrographs or gels must only be included
within the page limits of the Research Strategy. The maximum size of images to be included should be
approximately 1200 x 1500 pixels using 256 colors. Figures must be readable as printed on an 8.5 x 11
inch page at normal (100%) scale.
Investigators must use image compression such as JPEG or PNG. Do not include figures or photographs
as separate attachments either in the Appendix or elsewhere in the application.
Separate Attachments
Separate attachments have been designed for the Research Plan sections to maximize automatic
validations conducted by the eRA system. When the application is received by the agency, all of the
Research Plan sections will be concatenated in the appropriate order so that reviewers and agency staff
will see a single cohesive Research Plan.
When attaching a PDF document to the actual forms, please note you are attaching an actual document,
not just pointing to the location of an externally stored document. Therefore, if you revise the document
after it has been attached, you must delete the previous attachment and then reattach the revised
document to the application form. Use the View Attachment button to determine if the correct version
has been attached.
Page Limits
Applicants must follow the page limits described in table 2.6.1 unless the FOA specifies otherwise.
All tables, graphs, figures, diagrams, and charts must be included within the Research Strategy page limit.
If PAs or RFAs contain specific page limits, those instructions always supersede these instructions.
All applications and proposals for NIH funding must be self-contained within specified page limits.
Agency validations will include many checks for page limits. Note that while these computer validations
will help minimize incomplete and/or non-compliant applications, they may not address all page limit
requirements for a specific FOA and do not replace the validations conducted by NIH staff. Applications
found not to comply with the requirements may be delayed in the review process. Unless otherwise
specified in an NIH solicitation, internet website addresses (URLs) may not be used to provide
information necessary to the review because reviewers are not obligated to view the internet sites.
Moreover, reviewers are cautioned that they should not directly access an website (except to review
publications cited in the Biographical Sketch or Progress Report publication list) as it could compromise
their anonymity.
Applicants are prohibited from using the Appendix to circumvent page limitations in any section of the
application for which a page limit applies. For additional information regarding Appendix material and
page limits, please refer to the NIH Guide Notice NOT-OD-11-080,
http://grants.nih.gov/grants/guide/notice-files/NOT-OD-11-080.html
.
Notice of Proprietary Information
Applicants are discouraged from submitting information considered proprietary unless it is deemed
essential for proper evaluation of the application. However, when the application contains information
that constitutes trade secrets, or information that is commercial or financial, or information that is
confidential or privileged, make sure you have checked “Yes” for the proprietary/privileged information
question in the Other Project Information form.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-111
When information in the application constitutes trade secrets or information that is commercial or
financial, or information that is confidential or privileged, it is furnished to the Government in confidence
with the understanding that the information shall be used or disclosed only for evaluation of this
application. However, if a grant is awarded as a result of or in connection with the submission of this
application, the Government shall have the right to use or disclose the information to the extent
authorized by law. This restriction does not limit the Government’s right to use the information if it is
obtained without restriction from another source.
Although the grantee institution and the PD/PI will be consulted about any such release, the PHS will
make the final determination. Any indication by the applicant that the application contains proprietary or
privileged information does not automatically shield the information from release in response to a
Freedom of Information Act (FOIA) request should the application result in an award (see
45 CFR Part
5). If an applicant fails to identify proprietary information at the time of submission as instructed in the
application guide, a significant substantive justification will be required to withhold the information if
requested under FOIA.
Begin each text section of the Research Plan with a section header (e.g., Introduction, Specific
Aims, Research Strategy, etc).
Field Name Instructions
1. Introduction to
Application (for
Resubmission or Revision
only)
Use only if Type of Application is Resubmission or Revision. See specific
instructions in Part I Section 2.7, Resubmission Applications and Part I
Section 2.8, Revision Applications on the content of the Introduction.
First time (new) applications should not include an Introduction.
The Introduction is a required attachment for Resubmissions and
Revisions. Follow the page limits for the Research Strategy in the table of
page limits (Table 2.6-1
), unless specified otherwise in the FOA.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
2. Specific Aims State concisely the goals of the proposed research and summarize the
expected outcome(s), including the impact that the results of the proposed
research will exert on the research field(s) involved.
List succinctly the specific objectives of the research proposed, e.g., to
test a stated hypothesis, create a novel design, solve a specific problem,
challenge an existing paradigm or clinical practice, address a critical
barrier to progress in the field, or develop new technology.
The Specific Aims attachment is required unless otherwise specified in
the FOA. Follow the page limits for the Research Strategy in the table of
page limits (Table 2.6-1
), unless specified otherwise in the FOA.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
3. Research Strategy Organize the Research Strategy in the specified order and using the
instructions provided below. Start each section with the appropriate
section heading – Significance, Innovation, Approach. Cite published

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-112
Field Name Instructions
experimental details in the Research Strategy section and provide the full
reference in the Bibliography and References Cited section (Part I Section
4.4.9).
Follow the page limits for the Research Strategy in the table of page limits
(Table 2.6-1), unless specified otherwise in the FOA. Note that the page
limit for this attachment will be validated as a single file.
(a) Significance
• Explain the importance of the problem or critical barrier to
progress in the field that the proposed project addresses.
• Explain how the proposed project will improve scientific
knowledge, technical capability, and/or clinical practice in
one or more broad fields.
• Describe how the concepts, methods, technologies,
treatments, services, or preventative interventions that drive
this field will be changed if the proposed aims are achieved.
(b) Innovation
• Explain how the application challenges and seeks to shift
current research or clinical practice paradigms.
• Describe any novel theoretical concepts, approaches or
methodologies, instrumentation or interventions to be
developed or used, and any advantage over existing
methodologies, instrumentation, or interventions.
• Explain any refinements, improvements, or new applications
of theoretical concepts, approaches or methodologies,
instrumentation, or interventions.
(c) Approach
• Describe the overall strategy, methodology, and analyses to
be used to accomplish the specific aims of the project. Unless
addressed separately the Resource Sharing Plan, attachment
include how the data will be collected, analyzed, and
interpreted as well as any resource sharing plans as
appropriate.
• Discuss potential problems, alternative strategies, and
benchmarks for success anticipated to achieve the aims.
• If the project is in the early stages of development, describe
any strategy to establish feasibility, and address the
management of any high risk aspects of the proposed work.
• Point out any procedures, situations, or materials that may be
hazardous to personnel and precautions to be exercised. A full
discussion on the use of select agents should appear in the
Select Agent Research attachment, below.
• If research on Human Embryonic Stem Cells (hESCs) is
proposed but an approved cell line from the NIH hESC

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-113
Field Name Instructions
Registry cannot be identified, provide a strong justification
for why an appropriate cell line cannot be chosen from the
Registry at this time.
If an applicant has multiple Specific Aims, then the applicant may address
Significance, Innovation and Approach for each Specific Aim
individually, or may address Significance, Innovation and Approach for
all of the Specific Aims collectively.
As applicable, also include the following information as part of the
Research Strategy, keeping within the three sections listed above:
Significance, Innovation, and Approach.
Preliminary Studies for New Applications: For new applications,
include information on Preliminary Studies. Discuss the PD/PI’s
preliminary studies, data, and or experience pertinent to this application.
Except for Exploratory/Developmental Grants (R21/R33), Small Research
Grants (R03), and Academic Research Enhancement Award (AREA)
Grants (R15), preliminary data can be an essential part of a research grant
application and help to establish the likelihood of success of the proposed
project. Early Stage Investigators should include preliminary data
(however, for R01 applications, reviewers will be instructed to place less
emphasis on the preliminary data in application from Early Stage
Investigators than on the preliminary data in applications from more
established investigators).
Progress Report for Renewal and Revision Applications. For
renewal/revision applications, provide a Progress Report. Provide the
beginning and ending dates for the period covered since the last
competitive review. Summarize the specific aims of the previous project
period and the importance of the findings, and emphasize the progress
made toward their achievement. Explain any significant changes to the
specific aims and any new directions including changes to the specific
aims and any new directions including changes resulting from significant
budget reductions. A list of publications, patents, and other printed
materials should be included in the Progress Report Publication List
attachment; do not include that information here.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
4. Progress Report
Publication List (Renewal
Applications Only)
List the titles and complete references to all appropriate publications,
manuscripts accepted for publication, patents, and other printed materials
that have resulted from the project since it was last reviewed
competitively. When citing articles that fall under the Public Access
Policy, were authored or co-authored by the applicant and arose from NIH
support, provide the NIH Manuscript Submission reference number (e.g.,
NIHMS97531) or the PubMed Central (PMC) reference number (e.g.,
PMCID234567) for each article. If the PMCID is not yet available
because the Journal submits articles directly to PMC on behalf of their

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-114
Field Name Instructions
authors, indicate “PMC Journal – In Process.” A list of these journals is
posted at: http://publicaccess.nih.gov/submit_process_journals.htm.
Citations that are not covered by the Public Access Policy, but are
publicly available in a free, online format may include URLs or PubMed
ID (PMID) numbers along with the full reference (note that copies of
these publications are not accepted as appendix material, see Part I
Section 5.5.15 for more information).
Human Subjects Sections
Field Name Instructions
5. Protection of Human
Subjects
Refer to Part II, Supplemental Instructions for Preparing the Human
Subjects Section of the Research Plan.
Complete this section if you answered “yes” to the question “are human
subjects involved?” on the R&R Other Project Information Form. If you
answered “no” to the question but your proposed research involves human
specimens and/or data from subjects you must provide a justification in
this section or your claim that no human subjects are involved. Follow the
instructions provided in the Application Guide and the FOA regarding the
Protection of Human Subject attachment.
Do not use the protection of human subjects section to circumvent the
page limits of the Research Strategy.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
6. Inclusion of Women and
Minorities
Refer to Part II, Supplemental Instructions for Preparing the Human
Subjects Section of the Research Plan. Complete this section if you
answered “yes” to the question “are human subjects involved?” on the
R&R Other Project Information Form and the research does not fall under
Exemption 4.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
7. Inclusion of Children Refer to Supplemental Instructions for Preparing the Human Subjects
Section of the Research Plan, Sections 4.4 and 5.7
Complete this section
if you answered “yes” to the question “are human subjects involved?” on
the R&R Other Project Information Form and the research does not fall
under Exemption 4.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
Other Research Plan Sections

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-115
Field Name Instructions
8. Vertebrate
Animals
Complete this section if you answered “yes” to the question “are Vertebrate
Animals Used?” on the R&R Other Project Information Form.
If Vertebrate Animals are involved in the project, address each of the five points
below. This section should be a concise, complete description of the animals
and proposed procedures. While additional details may be included in the
Research Strategy, the responses to the five required points below must be
cohesive and include sufficient detail to allow evaluation by peer reviewers and
NIH staff. If all or part of the proposed research involving vertebrate animals
will take place at alternate sites (such as project/performance or collaborating
site(s)), identify those sites and describe the activities at those locations.
Although no specific page limitation applies to this section of the application,
be succinct. Failure to address the following five points will result in the
application being designated as incomplete and will be grounds for the PHS to
defer the application from the peer review round. Alternatively, the
application’s impact/priority score may be negatively affected.
If the involvement of animals is indefinite, provide an explanation and indicate
when it is anticipated that animals will be used. If an award is made, prior to the
involvement of animals the grantee must submit to the NIH awarding office
detailed information as required in points 1-5 below and verification of IACUC
approval. If the grantee does not have an Animal Welfare Assurance, then an
applicable Animal Welfare Assurance will be required (see Part III
Section 2.2
Vertebrate Animals for more information).
The five points are as follows:
1. Provide a detailed description of the proposed use of the animals in the
work outlined in the Research Strategy section. Identify the species,
strains, ages, sex, and numbers of animals to be used in the proposed work.
2. Justify the use of animals, the choice of species, and the numbers to be
used. If animals are in short supply, costly, or to be used in large numbers,
provide an additional rationale for their selection and numbers.
3. Provide information on the veterinary care of the animals involved.
4. Describe the procedures for ensuring that discomfort, distress, pain, and
injury will be limited to that which is unavoidable in the conduct of
scientifically sound research. Describe the use of analgesic, anesthetic, and
tranquilizing drugs and/or comfortable restraining devices, where
appropriate, to minimize discomfort, distress, pain, and injury.
5. Describe any method of euthanasia to be used and the reasons for its
selection. State whether this method is consistent with the
recommendations of the American Veterinary Medical Association
(AVMA) Guidelines on Euthanasia. If not, include a scientific justification
for not following the recommendations.
For additional information, see
http://grants.nih.gov/grants/olaw/VASchecklist.pdf
.
Do not use the vertebrate animal section to circumvent the page limits of the

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-116
Field Name Instructions
Research Strategy.
Save this information in a single file in a location you remember. Click Add
Attachment, browse to where you saved the file, select the file, and then click
Open.
9. Select Agent
Research
Select agents are hazardous biological agents and toxins that have been
identified by DHHS or USDA as having the potential to pose a severe threat to
public health and safety, to animal and plant health, or to animal and plant
products. CDC and the Animal APHIS Select Agent Programs jointly maintain
a list of these agents. See http://www.selectagents.gov/
.
If the activities proposed in the application involve only the use of a strain(s) of
select agents which has been excluded from the list of select agents and toxins
as per 42 CFR 73.3, the select agent requirements do not apply. Use this section
to identify the strain(s) of the select agent that will be used and note that it has
been excluded from this list. The list of exclusions is available at
http://www.selectagents.gov/Select%20Agents%20and%20Toxins%20Exclusio
ns.html.
If the strain(s) is not currently excluded from the list of select agents and toxins
but you have applied or intend to apply to DHHS for an exclusion from the list,
use this section to indicate the status of your request or your intent to apply for
an exclusion and provide a brief justification for the exclusion.
If any of the activities proposed in your application involve the use of select
agents at any time during the proposed project period, either at the applicant
organization or at any other performance site, address the following three points
for each site at which select agent research will take place. Although no specific
page limitation applies to this section, be succinct.
1. Identify the select agent(s) to be used in the proposed research.
2. Provide the registration status of all entities* where select agent(s) will be
used.
• If the performance site(s) is a foreign institution, provide the
name(s) of the country or countries where select agent research will
be performed.
*An “entity” is defined in 42 CFR 73.1 as “any government agency
(Federal, State, or local), academic institution, corporation, company,
partnership, society, association, firm, sole proprietorship, or other legal
entity.”
3. Provide a description of all facilities where the select agent(s) will be used.
• Describe the procedures that will be used to monitor possession, use
and transfer of the select agent(s).
• Describe plans for appropriate biosafety, biocontainment, and
security of the select agent(s).
• Describe the biocontainment resources available at all performance
sites.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-117
Field Name Instructions
If you are responding to a specific funding opportunity announcement (e.g., PA
or RFA), address any requirements specified by the FOA.
Reviewers will assess the information provided in this Section, and any
questions associated with select agent research will need to be addressed prior
to award.
Save this file in a location you remember. Click Add Attachment, browse to
where you saved the file, select the file, and then click Open.
10. Multiple PD/PI
Leadership Plan
For applications designating multiple PD/PIs, a leadership plan must be
included. For applications designating multiple PD/PIs, all such individuals
must be assigned the PD/PI role on the Senior/Key Profile form, even those at
organizations other than the applicant organization. A rationale for choosing a
multiple PD/PI approach should be described. The governance and
organizational structure of the leadership team and the research project should
be described, including communication plans, process for making decisions on
scientific direction, and procedures for resolving conflicts. The roles and
administrative, technical, and scientific responsibilities for the project or
program should be delineated for the PD/PIs and other collaborators. Do not
submit a leadership plan if you are not submitting a Multiple PD/PI application.
If budget allocation is planned, the distribution of resources to specific parts of
the project or the individual PD/PIs should be delineated in the Leadership Plan.
In the event of an award, the requested allocations may be reflected in a
footnote on the Notice of Grant Award.
Save this file in a location you remember. Click Add Attachment, browse to
where you saved the file, select the file, and then click Open.
11.
Consortium/Contract
ual Arrangements
Explain the programmatic, fiscal, and administrative arrangements to be made
between the applicant organization and the consortium organization(s). If
consortium/contractual activities represent a significant portion of the overall
project, explain why the applicant organization, rather than the ultimate
performer of the activities, should be the grantee. The signature of the
Authorized Organization Representative on the SF424 (R&R) cover form
signifies that the applicant and all proposed consortium participants understand
and agree to the following statement:
The appropriate programmatic and administrative personnel of each
organization involved in this grant application are aware of the
agency’s consortium agreement policy and are prepared to establish the
necessary inter-organizational agreement(s) consistent with that policy.
Save this information in a single file in a location you remember. Click Add
Attachment, browse to where you saved the file, select the file, and then click
Open.
12. Letters of Support
(e.g., Consultants)
Provide all appropriate letters of support, including any letters necessary to
demonstrate the support of consortium participants and collaborators such as
Senior/Key Personnel and Other Significant Contributors included in the grant
application. Letters are not required for personnel (such as research assistants)

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-118
Field Name Instructions
not contributing in a substantive, measurable way to the scientific development
or execution of the project. Letters should stipulate expectations for co-
authorship, and whether cell lines, samples or other resources promised in the
letter are freely available to other investigators in the scientific community or
will be provided to the particular investigators only. For consultants, letters
should include rate/charge for consulting services and level of effort/number of
hours per year anticipated. In addition, letters ensuring access to core facilities
and resources should stipulate whether access will be provided as a fee-for-
service. Do not place these letters in the Appendix. Consultant biographical
sketches should be in the Biographical Sketch section.
Save this information in a single file in a location you remember. Click Add
Attachment, browse to where you saved the file, select the file, and then click
Open.
13. Resource Sharing
Plan(s)
NIH considers the sharing of unique research resources developed through NIH-
sponsored research an important means to enhance the value and further the
advancement of the research. When resources have been developed with NIH
funds and the associated research findings published or provided to NIH, it is
important that they be made readily available for research purposes to qualified
individuals within the scientific community. See Supplemental Instructions
Part
III, 1.5 Sharing Research Resources.
1. Data Sharing Plan: Investigators seeking $500,000 or more in direct costs
(exclusive of consortium F&A) in any year are expected to include a brief 1-
paragraph description of how final research data will be shared, or explain why
data-sharing is not possible. Specific Funding Opportunity Announcements may
require that all applications include this information regardless of the dollar
level. Applicants are encouraged to read the specific opportunity carefully and
discuss their data-sharing plan with their program contact at the time they
negotiate an agreement with the Institute/Center (IC) staff to accept assignment
of their application. See Data-Sharing Policy
or
http://grants.nih.gov/grants/guide/notice-files/NOT-OD-03-032.html.
2. Sharing Model Organisms: Regardless of the amount requested, all
applications where the development of model organisms is anticipated are
expected to include a description of a specific plan for sharing and distributing
unique model organisms or state why such sharing is restricted or not possible.
See Sharing Model Organisms Policy, and NIH Guide NOT-OD-04-042
.
3. Genome Wide Association Studies (GWAS): Applicants seeking funding for a
genome-wide association study are expected to provide a plan for submission of
GWAS data to the NIH-designated GWAS data repository, or an appropriate
explanation why submission to the repository is not possible. GWAS is defined
as any study of genetic variation across the entire genome that is designed to
identify genetic associations with observable traits (such as blood pressure or
weight) or the presence or absence of a disease or condition. For further
information see Policy for Sharing of Data Obtained in NIH Supported or
Conducted Genome-Wide Association Studies, NIH Guide NOT-OD-07-088
,
and http://gwas.nih.gov/.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-119
Field Name Instructions
Save this information in a single file in a location you remember. Click Add
Attachment, browse to where you saved the file, select the file, and then click
Open.
15. Appendix Only one copy of appendix material is necessary. Use the Add Attachments
button to the right of this field to complete this entry.
A maximum of 10 PDF attachments is allowed in the Appendix. If more than 10
appendix attachments are needed, combine the remaining information into
attachment #10. Note that this is the total number of appendix items, not the
total number of publications. When allowed there is a limit of 3 publications
that are not publicly available (see below for further details and check the FOA
for any specific instructions), though not all grant activity codes allow
publications to be included in the appendix.
Do not use the appendix to circumvent the page limits of the Research Strategy
or any other section of the application for which a page limit applies. For
additional information regarding Appendix material and page limits, please
refer to the NIH Guide Notice NOT-OD-11-080,
http://grants.nih.gov/grants/guide/notice-files/NOT-OD-11-080.html
.
Appendix material may not appear in the assembled application in the order
attached, so it is important to use filenames for attachments that are descriptive
of the content. A summary sheet listing all of the items included in the appendix
is also encouraged but not required. When including a summary sheet, it should
be included in the first appendix attachment. Applications that do not follow the
appendix requirements may be delayed in the review process.
New, resubmission, renewal, and revision applications may include the
following materials in the Appendix (note, however, that some FOAs do not
permit publications):
• Publications – No longer allowed as appendix materials except
in the circumstances noted below. Applicants may submit up to 3
of the following types of publications:
o Manuscripts and/or abstracts accepted for publication but
not yet published: The entire article should be submitted as a
PDF attachment.
o Manuscripts and/or abstracts published, but a free, online,
publicly available journal link is not available: The entire
article should be submitted as a PDF attachment.
o Patents directly relevant to the project: The entire document
should be submitted as a PDF attachment.
(Do not include unpublished theses, or abstracts/manuscripts submitted
(but not yet accepted) for publication.)
• Surveys, questionnaires, and other data collection instruments;
clinical protocols and informed consent documents may be
submitted in the Appendix as necessary.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-120
Field Name Instructions
• For materials that cannot be submitted electronically or materials
that cannot be converted to PDF format (e.g., medical devices,
prototypes, DVDs, CDs), applicants should contact the Scientific
Review Officer for instructions following notification of
assignment of the application to a SRG. Applicants are encouraged
to be as concise as possible and submit only information essential
for the review of the application.
Items that must not be included in the appendix:
• Photographs or color images of gels, micrographs, etc., are no
longer accepted as Appendix material. These images must be
included in the Research Strategy PDF. However, images
embedded in publications are allowed.
• Publications that are publicly accessible. For such publications, the
URL or PMC submission identification numbers along with the full
reference should be included as appropriate in the Bibliography and
References cited section, the Progress Report Publication List
section, and/or the Biographical Sketch section.
Once all data have been entered use the scroll bar to scroll up. You will be returned to the Grant
Application Package screen. To remove a form from your application, uncheck the box next to the form
name in the Optional document section.
5.6 (Reserved)
5.7 (Reserved)
5.8 Planned Enrollment Report and Cumulative Inclusion
Enrollment Report
NOTE: These report formats should NOT be used for collecting data from study participants. To
ensure proper performance, please save frequently.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-121
5.8.1 Planned Enrollment Report
Field Name Instructions

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-122
Field Name Instructions
Study Title Enter a unique title that describes the study that the participants will be
involved in. If there is more than one study, provide a separate Study Title
for each. Follow the instructions provided in the Application Guide and
the FOA regarding the Inclusion of Women and Minorities. Maximum
250 characters. This is a required field.
Domestic/Foreign Select whether the participants described in the planned enrollment report
are domestic or foreign. At a minimum, domestic and foreign participants
must be reported separately even if for the same study. This is a required
field.
Comments Enter information you wish to provide about this planned enrollment
report. This includes but is not limited to addressing information about
distinctive subpopulations if relevant to the scientific hypotheses being
studied and/or a study that will have a delayed onset. Maximum 500
characters.
American Indian/Alaska
Native
Enter the expected number of females and males (in the respective fields)
who are American Indian/Alaska Native and Not Hispanic or Latino, and;
enter the expected number of females and males (in the respective fields)
who are American Indian/Alaska Native and Hispanic or Latino. These
are required fields.
Asian Enter the expected number of females and males (in the respective fields)
who are Asian and Not Hispanic or Latino, and; enter the expected
number of females and males (in the respective fields) who are Asian and
Hispanic or Latino. These are required fields.
Native Hawaiian or Other
Pacific Islander
Enter the expected number of females and males (in the respective fields)
who are Native Hawaiian or Other Pacific Islander and Not Hispanic or
Latino, and; enter the expected number of females and males (in the
respective fields) who are Native Hawaiian or Other Pacific Islander and
Hispanic or Latino. These are required fields.
Black or African American Enter the expected number of females and males (in the respective fields)
who are Black or African American and Not Hispanic or Latino, and;
Enter the expected number of females and males (in the respective fields)
who are Black or African American and Hispanic or Latino. These are
required fields.
White Enter the expected number of females and males (in the respective fields)
who are White and Not Hispanic or Latino, and; enter the expected
number of females and males (in the respective fields) who are White and
Hispanic or Latino. These are required fields.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-123
Field Name Instructions
More than One Race Enter the expected number of females and males (in the respective fields)
who identify with more than one racial category and are Not Hispanic or
Latino, and; enter the expected number of females and males (in the
respective fields) who identify with more than one racial category and are
Hispanic or Latino. These are required fields.
Total The total fields at the bottom are auto-calculated to total all racial
categories for females and males who are Not Hispanic or Latino and all
racial categories for females and males who are Hispanic or Latino. The
total fields at the right are auto-calculated to total all males and females of
both Not Hispanic or Latino and Hispanic or Latino ethnicity in each
racial category.
5.8.2 Cumulative Inclusion Enrollment Report

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-124
Field Name Instructions
Study Title Enter a unique title that describes the study that the participants will be
involved in. The title should be the same as submitted on the original
Planned Enrollment form for this study. Follow the instructions provided
in the Application Guide and the FOA regarding the Inclusion of Women
and Minorities. Maximum 250 characters. This is a required field.
Comments Enter information you wish to provide about this Cumulative Inclusion
Enrollment Report. This includes but is not limited to information if
distinctive subpopulations are relevant to the scientific hypotheses being
studied. Maximum 500 characters.
American Indian/Alaska
Native
Enter the number of females, males, and individuals of unknown/not
reported sex/gender (in the respective fields) who are American
Indian/Alaska Native and Not Hispanic or Latino, and; enter the number
of females, males, and individuals of unknown/not reported sex/gender (in
the respective fields) who are American Indian/Alaska Native and
Hispanic or Latino; and enter the number of females, males, and
individuals of unknown/not reported sex/gender (in the respective fields)
who are American Indian/Alaska Native and of unknown/not reported
ethnicity. These are required fields.
Asian Enter the number of females, males, and individuals of unknown/not
reported sex/gender (in the respective fields) who are Asian and Not
Hispanic or Latino, and; enter the number of females, males, and
individuals of unknown/not reported sex/gender (in the respective fields)
who are Asian and Hispanic or Latino; and enter the number of females,
males, and individuals of unknown/not reported sex/gender (in the
respective fields) who are Asian and of unknown/not reported ethnicity.
These are required fields.
Native Hawaiian or Other
Pacific Islander
Enter the number of females, males, and individuals of unknown/not
reported sex/gender (in the respective fields) who are Native Hawaiian or
Other Pacific Islander and Not Hispanic or Latino, and; enter the number
of females, males, and individuals of unknown/not reported sex/gender (in
the respective fields) who are Native Hawaiian or Other Pacific Islander
and Hispanic or Latino; and enter the number of females, males, and
individuals of unknown/not reported sex/gender (in the respective fields)
who are Native Hawaiian or Other Pacific Islander and of unknown/not
reported ethnicity. These are required fields.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-125
Black or African American Enter the number of females, males, and individuals of unknown/not
reported sex/gender (in the respective fields) who are Black or African
American and Not Hispanic or Latino, and; enter the number of females,
males, and individuals of unknown/not reported sex/gender (in the
respective fields) who are Black or African American and Hispanic or
Latino; and enter the number of females, males, and individuals of
unknown/not reported sex/gender (in the respective fields) who are Black
or African American and of unknown/not reported ethnicity. These are
required fields.
White Enter the number of females, males, and individuals of unknown/not
reported sex/gender (in the respective fields) who are White and Not
Hispanic or Latino, and; enter the number of females, males, and
individuals of unknown/not reported sex/gender (in the respective fields)
who are White and Hispanic or Latino; and enter the number of females,
males, and individuals of unknown/not reported sex/gender (in the
respective fields) who are White and of unknown/not reported ethnicity.
These are required fields.
More than One Race Enter the number of females, males, and individuals of unknown/not
reported sex/gender (in the respective fields) who identify with more than
one racial category and are Not Hispanic or Latino, and; enter the number
of females, males, and individuals of unknown/not reported sex/gender (in
the respective fields) who identify with more than one racial category and
are Hispanic or Latino; and enter the number of females, males, and
individuals of unknown/not reported sex/gender (in the respective fields)
who identify with more than one racial category and of unknown/not
reported ethnicity. These are required fields.
Unknown or Not Reported Enter the number of females, males, and individuals of unknown/not
reported sex/gender (in the respective fields) whose race is unknown/not
reported and who are Not Hispanic or Latino, and; enter the number of
females, males, and individuals of unknown/not reported sex/gender (in
the respective fields) whose race is unknown/not reported and who are
Hispanic or Latino; and enter the number of females, males, and
individuals of unknown/not reported sex/gender (in the respective fields)
who are of unknown/not reported race and of unknown/not reported
ethnicity. These are required fields.
Total The total fields at the bottom are auto-calculated to total all racial
categories for females, males, and individuals of unknown/not reported
sex/gender who are Not Hispanic or Latino; all racial categories for
females, males, and individuals of unknown/not reported sex/gender who
are Hispanic or Latino, and all racial categories for females, males, and
individuals of unknown/not reported sex/gender who are of unknown/not
reported ethnicity. The total fields at the right are auto-calculated to total
all individuals in a given racial category.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-126
6. Peer Review Process
Overview
NIH policy is intended to ensure that applications for funding submitted to the NIH are evaluated on the
basis of a process that is fair, equitable, timely, and conducted in a manner free of bias. The NIH dual
peer review system is mandated by statute in accordance with section 492 of the Public Health Service
Act and federal regulations governing "Scientific Peer Review of Research Grant Applications and
Research and Development Contract Projects" (42 CFR part 52h
).
The first level of review is carried out by a Scientific Review Group (SRG) composed primarily of non-
federal scientists who have expertise in relevant scientific disciplines and current research areas. The
second level of review is performed by Institute and Center (IC) National Advisory Councils or Boards.
Councils composed of both scientific and lay members are chosen for their expertise, interest, or activity
in matters related to health and disease. Only applications that are favorably recommended by both the
SRG and the Advisory Council (or the IC in the case of fellowships) are considered for funding. Only the
NIH Institute or Center may make funding decisions.
NIH has implemented Special Council Review procedures for the additional review of grant and
cooperative agreement applications from Program Director(s)/Principal Investigator(s) [PD(s)/PI(s)] who
already receive in excess of $1 million per year in total costs to determine if additional funds should be
provided to already well-supported investigators. It is important to note that the Special Council Review
does not represent a cap on total NIH funding for any one investigator. For additional information, please
see NOT-OD-12-110 and NOT-OD-12-140
.
A detailed description of what happens to a research project grant application at NIH after it is received
for peer review can be found at the following location:
http://grants.nih.gov/grants/peer_review_process.htm
.
Additional information about charters and membership of SRGs, Councils, and Boards can be obtained
from the appropriate Institute, Center, or Office. Information on CDC review procedures is located at
http://www.cdc.gov/phpr/science/erp_policies.htm
.
Streamlining
The initial scientific peer review of most applications will also include a process in which only those
applications deemed by the reviewers to have the highest scientific and technical merit, generally the
better half of the applications under review, will be discussed at the SRG meeting, assigned an impact
score, and receive a second level review. Applications in the lower half are reviewed by SRG members
but they are not discussed or assigned overall impact scores at the SRG meeting. This process allows the
reviewers to focus their discussion on the most meritorious applications.
Before the review meeting, each reviewer and discussant assigned to an application will give a separate
score for each of (at least) five review criteria and a preliminary impact score for that application (see
below). The preliminary impact scores help the SRG to determine which applications will be discussed.
Scoring
Each FOA specifies all of the review criteria and considerations that will used in the evaluation of
applications submitted for that FOA; RFAs and other types of funding opportunities (e.g., for construction
or fellowship applications) may include different and/or additional review criteria and considerations.
SRG members are instructed to evaluate research applications by addressing the scored review criteria
(see below) and additional review criteria as applicable for the application.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-127
For each application that is discussed, a final overall impact/priority score will be given by each eligible
committee member (without conflicts of interest) following the panel discussion. Each member’s impact
score will reflect his/her evaluation of the potential overall impact of the project in its entirety, rather than
an arithmetic formula applied to the reviewer’s scores given to each criterion. The final impact score for
each discussed application will be determined by calculating the arithmetic average of all the eligible
members’ impact scores, and multiplying the average by 10.
As part of the initial merit review, and regardless of whether an application is discussed or not discussed
(streamlined), all applicants will receive a written critique, called a Summary Statement. Unless stated
otherwise in the FOA, the Summary Statement represents a combination of the reviewers' written
comments and scores for individual criteria. The Summary Statement for discussed applications includes
the Scientific Review Officer's summary of the members' discussion during the SRG meeting; the final
impact score; the recommendations of the SRG, including budget recommendations; and administrative
notes of special considerations. For applications that are not discussed by the full committee, the scores of
the assigned reviewers and discussants for the five scored criteria will be reported individually on the
Summary Statement. Final, numerical impact scores are not given for applications that are not discussed.
Research Project Evaluation Criteria
Overall Impact. Reviewers will provide an overall impact/priority score to reflect their assessment of
the likelihood for the project to exert a sustained, powerful influence on the research field(s) involved, in
consideration of the following scored review criteria, and additional review criteria (as applicable for the
project proposed).
Scored Review Criteria. Reviewers will consider each of the five review criteria below in the
determination of scientific and technical merit, and give a separate score for each. An application does not
need to be strong in all categories to be judged likely to have major scientific impact. For example, a
project that by its nature is not innovative may be essential to advance a field.
Significance: Does the project address an important problem or a critical barrier to progress in the
field? If the aims of the project are achieved, how will scientific knowledge, technical capability,
and/or clinical practice be improved? How will successful completion of the aims change the
concepts, methods, technologies, treatments, services, or preventative interventions that drive this
field?
Investigator(s): Are the PD/PIs, collaborators, and other researchers well suited to the project? If
Early Stage Investigators or New Investigators, or in the early stages of independent careers, do they
have appropriate experience and training? If established, have they demonstrated an ongoing record
of accomplishments that have advanced their field(s)? If the project is collaborative or multi-PD/PI,
do the investigators have complementary and integrated expertise; are their leadership approach,
governance and organizational structure appropriate for the project?
Innovation: Does the application challenge and seek to shift current research or clinical practice
paradigms by utilizing novel theoretical concepts, approaches or methodologies, instrumentation, or
interventions? Are the concepts, approaches or methodologies, instrumentation, or interventions
novel to one field of research or novel in a broad sense? Is a refinement, improvement, or new
application of theoretical concepts, approaches or methodologies, instrumentation, or interventions
proposed?
Approach: Are the overall strategy, methodology, and analyses well-reasoned and appropriate to
accomplish the specific aims of the project? Are potential problems, alternative strategies, and
benchmarks for success presented? If the project is in the early stages of development, will the
strategy establish feasibility and will particularly risky aspects be managed?

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-128
If the project involves human subjects and/or NIH-defined clinical research, are the plans to address
1) the protection of human subjects from research risks, and 2) inclusion (or exclusion) of
individuals on the basis of sex/gender, race, and ethnicity, as well as the inclusion or exclusion of
children, justified in terms of the scientific goals and research strategy proposed?
Environment: Will the scientific environment in which the work will be done contribute to the
probability of success? Are the institutional support, equipment and other physical resources
available to the investigators adequate for the project proposed? Will the project benefit from unique
features of the scientific environment, subject populations, or collaborative arrangements?
Additional Review Criteria. As applicable for the project proposed, reviewers will consider the
following additional items in the determination of scientific and technical merit, and in providing an
overall impact/priority score, but will not give separate scores for these items.
Appropriate Representation (for Conference Grant Applications): How well do the plans for
inclusion of women, racial/ethnic minorities, persons with disabilities, and other individuals who
traditionally have been underrepresented in science provide for their appropriate representation in
the planning, organization, and execution of the proposed conference/scientific meeting? For more
information, visit Inclusion of Women, Minorities and Persons with Disabilities in NIH-Supported
Conference Grants
Protections for Human Subjects: For research that involves human subjects but does not involve
one of the six categories of research that are exempt under 45 CFR part 46, the committee will
evaluate the justification for involvement of human subjects and the proposed protections from
research risk relating to their participation according to the following five review criteria: 1) risk to
subjects, 2) adequacy of protection against risks, 3) potential benefits to the subjects and others, 4)
importance of the knowledge to be gained, and 5) data and safety monitoring for clinical trials.
For research that involves human subjects and meets the criteria for one or more of the six categories
of research that are exempt under 45 CFR part 46, the committee will evaluate: 1) the justification
for the exemption, 2) human subjects involvement and characteristics, and 3) sources of materials.
For additional information on review of the Human Subjects section, please refer to the
Human
Subjects Protection and Inclusion Guidelines.
Inclusion of Women, Minorities, and Children: When the proposed project involves human subjects
and/or NIH-defined clinical research, the committee will evaluate the proposed plans for the
inclusion (or exclusion) of individuals on the basis of sex/gender, race, and ethnicity, as well as the
inclusion (or exclusion) of children to determine if it is justified in terms of the scientific goals and
research strategy proposed. For additional information see the
Human Subjects Protection and
Inclusion Guidelines.
Vertebrate animals: The committee will evaluate the involvement of live vertebrate animals as part
of the scientific assessment according to the following five points: 1) proposed use of the animals,
and species, strains, ages, sex, and numbers to be used; 2) justifications for the use of animals and
for the appropriateness of the species and numbers proposed; 3) adequacy of veterinary care; 4)
procedures for limiting discomfort, distress, pain and injury to that which is unavoidable in the
conduct of scientifically sound research including the use of analgesic, anesthetic, and tranquilizing
drugs and/or comfortable restraining devices; and 5) methods of euthanasia and reason for selection
if not consistent with the AVMA Guidelines on Euthanasia. For additional information, see
http://grants.nih.gov/grants/olaw/VASchecklist.pdf
.
Biohazards: Reviewers will assess whether materials or procedures proposed are potentially
hazardous to research personnel and/or the environment, and if needed, determine whether adequate
protection is proposed.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-129
Resubmission Applications: When reviewing a Resubmission application the committee will
evaluate the application as now presented, taking into consideration the response to comments from
the previous scientific review group and changes made to the project.
Renewal Applications: When reviewing a Renewal application, the committee will consider the
progress made in the last funding period.
Revision Applications: When reviewing a Revision application, the committee will consider the
appropriateness of the proposed expansion of the scope of the project. If the Revision application
relates to a specific line of investigation presented in the original application that was not
recommended for approval by the committee, then the committee will consider whether the
responses to comments from the previous scientific review group are adequate and whether
substantial changes are clearly evident.
Additional Review Considerations. As applicable for the project proposed, reviewers will address each
of the following items, but will not give scores for these items and should not consider them in providing
an overall impact score.
Provision of Family Care Facilities (for Conference Grant Applications). Are the plans to inform
attendees about family care resources adequate?
Applications from Foreign Organizations. Reviewers will assess whether the project presents
special opportunities for furthering research programs through the use of unusual talent, resources,
populations, or environmental conditions that exist in other countries and either are not readily
available in the United States or augment existing U.S. resources.
Select Agent Research. Reviewers will assess the information provided in this section of the
application, including 1) the select agent(s) to be used in the proposed research, 2) the registration
status of all entities where select agent(s) will be used, 3) the procedures that will be used to monitor
possession use and transfer of select agent(s), and 4) plans for appropriate biosafety, biocontainment,
and security of the select agent(s).
Resource Sharing Plans. Reviewers will comment on whether the following Resource Sharing
Plans, or the rationale for not sharing the following types of resources, are reasonable: 1) Data
Sharing Plan (http://grants.nih.gov/grants/policy/data_sharing/data_sharing_guidance.htm
); 2)
Sharing Model Organisms (http://grants.nih.gov/grants/guide/notice-files/NOT-OD-04-042.html);
and 3) Genome Wide Association Studies (GWAS) (http://grants.nih.gov/grants/guide/notice-
files/NOT-OD-07-088.html).
Budget and Period of Support. Reviewers will consider whether the budget and the requested period
of support are fully justified and reasonable in relation to the proposed research
Dual-Level Peer Review
The second level of review will usually be performed by the Advisory Council or Board of the potential
awarding component (Institute, Center, or other unit). Council or Board recommendations are based not
only on considerations of scientific merit, as judged by the SRGs, but also on the relevance of the
proposed study to an Institute/Center’s mission, programs and priorities.
NIH has implemented Special Council Review procedures for the additional review of grant and
cooperative agreement applications from Program Director(s)/Principal Investigator(s) [PD(s)/PI(s)] who
already receive in excess of $1 million per year in total costs to determine if additional funds should be
provided to already well-supported investigators. It is important to note that the Special Council Review
does not represent a cap on total NIH funding for any one investigator. For additional information, please
see NOT-OD-12-110 and NOT-OD-12-140
.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-130
7. Supplemental Instructions to the SF 424(R&R) for
Preparing an Individual Research Career
Development Award (CDA) Application
(“K” Series)
7.1 Introduction
All applicants must use the SF 424 R&R Application for Federal Assistance, following the instructional
information in this Application Guide. The supplemental instructions found in this section (I.7) are for
Individual Career Development Award (CDA) series applications and include guidance and instructional
information only when there is a difference in the required information to be submitted or there is a need
for more specificity for the individual K program. Therefore, these supplemental instructions must be
used along with the information found in Parts I.1 – I.6 of this document.
These instructions do not cover applications for K12 and other institutional career development programs.
Institutions planning such applications should consult the applicable Funding Opportunity Announcement
(FOA) concerning eligibility, award criteria, and application procedures. Some K-series funded through
Requests for Applications (RFAs) may have special instructions.
It is imperative that applicants become familiar with the K activity code for which support is being
requested. Before applying for a K award, applicants should carefully review the applicable FOA for the
career award of interest, noting especially the eligibility requirements, requirements for a mentor, review
criteria, award provisions, and any special application instructions. Each FOA contains more specific
information associated with the award mechanism and includes names of individuals that may be
contacted prior to submission of an application for additional or clarifying information.
The eligibility criteria, support levels, and other important aspects of specific career awards, including
availability, may vary among NIH Institutes or Centers and other PHS agencies. For this reason, it is
strongly recommended that applicants consult with the NIH Scientific/Research contact of the appropriate
awarding component prior to submitting an application. FOAs and other guidelines are available on the
NIH K-Kiosk website http://grants.nih.gov/training/careerdevelopmentawards.htm
. Announcements for
various career award opportunities are issued periodically in the NIH Guide for Grants and Contracts, a
weekly electronic publication (
http://grants.nih.gov/grants/guide/index.html).
Note: A few individual K-series programs supported by the NIH include a delayed-award activation
and/or two award phases (e.g., K22, K99/R00). NIH intramural researchers may be eligible to apply for
these awards. The FOA will include any additional and/or specific instructions that must be followed
when applying for such support.
7.2 Individual Career Development Award Programs
The following chart provides a summary of the existing Career Development programs. Since this
information is subject to change, prospective applicants are encouraged to review the K-Kiosk
for the
most current program information. The K-Kiosk includes information on NIH-wide Parent FOAs as well
as IC-specific FOAs for a particular K program.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-131
Summary of Research Career Development Award Programs
PROGRAM
DESCRIPTION MENTOR
REFERENCE
LETTERS
K01
Mentored Research Scientist Development Award (
see K Kiosk)
Yes Yes
K02
Independent Scientist Award (
see K Kiosk)
No No
K05
Senior Scientist Award (
see K Kiosk)
No No
K07
Academic Career Award (see K Kiosk)
* *
K08
Mentored Clinical Scientist Development Award (
see K Kiosk)
Yes Yes
K18
Career Enhancement Award (see K Kiosk)
Yes Yes
K22
Career Transition Award (
see K Kiosk)
* Yes
K23
K23 Mentored Patient-Oriented Research Career Development Award
(
see K Kiosk)
Yes Yes
K24
Mid-Career Investigator Award in Patient Oriented Research (
see K
Kiosk)
No No
K25
Mentored Quantitative Research Career Development Award (see K
Kiosk)
Yes Yes
K26
Midcareer Investigator Award in Mouse Pathobiology Research (
see K
Kiosk)
No No
K99/R00
NIH Pathways to Independence (PI) Award (
see K Kiosk)
Yes Yes
*Varies with career status and source of award. Check the Funding Opportunity Announcement (FOA).
7.3 Letters of Reference (must be submitted electronically
through the eRA Commons)
At least three (but no more than 5) Letters of Reference are required for all applications defined as New
and Resubmissions (see Note below) for mentored support as indicated in the table above. The letters
should be from individuals not directly involved in the application, but who are familiar with the
applicant’s qualifications, training, and interests. The mentor/co-mentor(s) of the application cannot be
counted toward the three required references. It is important for the applicant to include the names of
those individuals in the application so that the NIH staff will be aware of planned reference letter
submissions. Within the application, the list of referees (including name, departmental affiliation, and
institution) is included in Other Attachments on the Other Project Information Form (see special K
instructions in Section 7.4.3). In addition, applicants must include the same list and information on the
SF424 (R&R) Cover Form in the Cover Letter Attachment.
The reference letters are critically important and should address the candidate's competence and potential
to develop into an independent biomedical or behavioral investigator. Only those individuals who can
make the most meaningful comments about the candidate's professional training and qualifications for a
research career should be used as referees. Where possible, some referees who are not from the
candidate's current department or organization, but are knowledgeable about their qualifications, should
be selected.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-132
Letters of Reference are due by the application receipt deadline date. Although previously NIH provided a
5 business days grace period for the receipt of letters of reference after the application receipt due date,
the new policy eliminates the grace period. More information can be found in NIH Guide Notice
NOT-
OD-11-079.
The candidate should request reference letters only from individuals who will be able to submit them to
the NIH at the time of the application submission due date (see NOT-OD-11-079
).
Applications that are missing the required letters of reference may be delayed in review or may not be
accepted.
Note: For resubmission applications, it is critical that NEW Letters of Reference be submitted providing
up-to-date evaluation of the applicant’s potential to become an independent researcher, and the continued
need for additional supervised research experience.
Electronic submission of a letter of reference is a separate process from submitting an application
electronically. Reference letters are submitted directly through the eRA Commons and do not use
Grants.gov. Therefore, this process requires that the referee be provided information including (a) the PI’s
(candidate’s) eRA Commons user name, (b) the PI’s first and last name as they appear on the PI’s eRA
Commons account, and (c) the number assigned to this Funding Opportunity Announcement.
Confirmation e-mails will be sent to both the referee and the candidate following reference letter
submission. The confirmation sent to the candidate will include the referee’s name and the date the letter
was submitted. The confirmation sent to the referee will include the referee and applicant’s names, a
confirmation number, and the date the letter was submitted.
The candidate may check the status of submitted letters by logging into their Commons account and
accessing the “check status” screen for this application. The candidate is responsible for reviewing the
status of submitted reference letters and contacting referees to ensure that letters are submitted by the
receipt deadline. While the candidate is able to check on the status of the submitted letters, the letters are
confidential and he/she will not have access to the letters themselves. Note: Because e-mail can be
unreliable, it is the candidate’s responsibility to check the status of his/her letters of reference in the
Commons.
Candidates should provide the following instructions to their referees.
Instructions for Referees: (these instructions are also found at:
http://grants.nih.gov/grants/funding/424/Referee_Instructions_Mentored_Career_Awards.doc
)
Name of Candidate (First & Last Name as shown in the eRA Commons): _______________
Candidate’s eRA Commons UserName: _______________
FOA Number: __________________________
The candidate is applying to the NIH for a Career Development Award. The purpose of this award is to
develop the research capabilities and career of the candidate. These awards provide salary support and
guarantee them the ability to devote at least 9 person months (75% of their total professional effort) to
research for the duration of the award. Many of these awards also provide funds for research and career
development costs. The award is available to persons who have demonstrated considerable potential to
become independent researchers, but who need additional supervised research experience in a productive
scientific setting, as well as to newly independent researchers.
In two pages or less (PDF format), describe the qualities and potential of the candidate for the career
development award program for which support is being requested. This should include your evaluation
with special reference to:
• potential to become an independent research scientist;

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-133
• evidence of originality;
• adequacy of scientific background;
• quality of research endeavors or publications to date, if any;
• commitment to health-oriented research; and
• need for further research experience and training
• any additional related comments that the referee may wish to provide
Please put the name of the candidate at the top of the letter. Also, be sure to include your name and title in
the letter.
Submitting Reference Letters
Letters must be submitted directly to the eRA Commons at:
https://public.era.nih.gov/commons/public/reference/submitReferenceLetter.do?mode=new
and must be
submitted by the application receipt deadline date. More information can be found in NIH Guide Notice
NOT-OD-11-079.
You will be requested to enter the following information on-line at the time of submission:
Referee Information:
• Referee First Name (Required)
• Referee Last Name Required)
• Referee MI Name (Not Required)
• Referee e-mail (Required)
• Referee institution/affiliation (Required)
• Referee department (Required)
Candidate Information:
• PI Commons User ID (Required)
• PI’s last name, as it appears on the PI’s Commons account (Required) (will be validated to ensure
they match)
• Funding Opportunity Announcement (FOA) Number (Required)
• Reference letter confirmation number (Required only if resubmitting a letter; not required
otherwise)
• Reference letter – two pages maximum; PDF format
After you have submitted your letter, both you and the candidate will receive a confirmation of receipt by
e-mail. The confirmation sent to the candidate will include your name and the date your letter was
submitted. However, the letters are confidential and the candidate will not be able to access the letters
themselves. Your e-mail confirmation will include a Reference Letter Submission Confirmation Number.
The Confirmation Number will be required when resubmitting letters for the current round. Please print
the confirmation e-mail for your records.
Revised reference letters may be submitted at the time of the application receipt date.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-134
7.4 K- Specific Instructions for K Applications using the
SF424 (R&R) Application
Standard Instructions found in Parts I.1 – I.6 should be followed with the exceptions found in this section.
Section numbers referenced below (e.g. 4.2 – 5.6) reflect those found in Part I.
7.4.1 Special Instructions for 4.2 Cover Form
Type of Application: Unless stated in the applicable FOA, individual K awards are usually not
renewable nor are they supplemented/revised (contact awarding form staff if clarification is needed).
Therefore, the applicant should generally check “new” or “resubmission.” “Renewal” applications are
accepted only for a few K programs; thus this value should only be checked if a specific FOA states
Renewals are accepted.
Proposed Project (Start and Ending Dates): The requested period of support must be within specified
limits for the type of K award requested.
Project Director/Principal Investigator (PD/PI) Contact Information: Provide the name of the
individual candidate (considered the PD/PI for K award programs). If the candidate is not located at the
applicant organization at the time the application is submitted, the information should reflect where the
candidate can be reached prior to the requested award start date. If the PD/PI is not located at the
applicant organization at the time of submission, the Commons account for the PD/PI must be affiliated
with the applicant organization. For additional information on creating affiliations for users in the eRA
Commons, see: https://commons.era.nih.gov/commons-help/175.htm
.
Note: For some career transition award programs (e.g., K22) the applicant may apply WITHOUT an
institutional affiliation. These individuals should refer to the specific funding opportunity announcement
(FOA) for application instructions.
Cover Letter Attachment
Mentored CDA applicants must include a cover letter. Applicants for independent CDAs are encouraged
to include a cover letter with the application. The cover letter is only for internal use and will not be
shared with peer reviewers. For mentored CDA applications, the cover letter must contain the same list of
Referees (including name, departmental affiliation, and institution) that is included in the Other Project
Information Form. Other Attachment (see Part I Section 7.4.3
).
For both mentored and non-mentored K applications, the cover letter can also include the information
found in Part I.4.2
.
7.4.2 Special Instructions for 4.3 Research & Related
Project/Performance Site Locations Form
Indicate where the work described in the Research and Career Development Plans will be conducted.
7.4.3 Special Instructions for 4.4 Other Project Information Form
Project Summary/Abstract (Do not exceed 1 page): Provide an abstract of the entire application
(candidate, environment, and research). Include the candidate's immediate and long-term career goals,
key elements of the research career development plan, and a description of the research project, as
indicated in Part I.4.4.6.
Facilities & Other Resources: Provide in the Attachment a detailed description of the institutional
facilities and resources available to the candidate, following the instructions in Part I.4.4.10. The

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-135
information provided is of major importance in establishing the feasibility of the goals of the career
development plan.
Other Attachments: All mentored K applications must include a list of Referees here. The list should
include the name of the referee, departmental affiliation and institution. This same list must also be
provided in the Cover Letter.
7.4.4 Special Instructions for 4.5 Senior/Key Person Profile(s) Form
7.4.4.1 The Candidate
For all K applications the K candidate is considered the Project Director/Principal Investigator (PD/PI).
Therefore the candidate must be registered in the eRA Commons and be assigned the PI role within the
Commons. Follow the instructions in Part I.2 which provides information regarding required registration
in the eRA Commons.
Note that agency policies concerning “Multiple PIs” are not applicable to K applications. Therefore, do
not use the PD/PI role for any other senior/key personnel.
Candidate’s Biographical Sketch
A biographical sketch attachment is required for the K candidate. Follow the page limits for the biosketch
in the table of page limits (Table 2.6-1
), unless specified otherwise in the FOA.
A biosketch for the K applicant should follow the instructions below:
Position Title: If the candidate is not currently located at the applicant organization, include both
“current” and “projected” position titles, labeling each accordingly.
Education: Complete the educational block at the top of the format page beginning with the
baccalaureate or other initial professional education, such as nursing, and include postdoctoral training;
separately referencing residency training when applicable. For each entry provide the name and location
of the institution;, the degree received (if applicable); the month and year the degree was received, and the
field of study. For residency entries, the Field of Study section should reflect the area of residency. For
non-degree education, indicate the time period covered. List professional certifications received within
the last 10 years.
Personal Statement: Briefly describe why your experience and qualifications make you particularly
well-suited to receive the K award for which you are applying.
Research and/or Professional Experience:
Use the headings given below instead of the instructions on the Biographical Sketch Format Page.
Identify each heading.
Employment
Start with the first position held following the baccalaureate and give a consecutive record to date.
Indicate the department and organization, department head or supervisor, rank, tenured or non-tenured,
status (full- or part-time), and inclusive dates (month and year). When applicable, include information on
military service, and, if not referenced under Education above, internships, residencies, research
assistantships, fellowships, etc. If the candidate is not currently located at the applicant organization,
include the projected employment position in this section as well.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-136
Honors
List academic and professional honors chronologically, including research grants and competitive
fellowships awarded to the candidate.
Professional Societies and Public Advisory Committees
Identify professional societies and related organizations in which membership has been held within the
last 10 years, giving dates. Include present membership on any Federal Government public advisory
committee.
Publications and Patent Citations
NIH encourages applicants to limit the list of selected peer-reviewed publications or manuscripts in press
to no more than 15. The individual may choose to include selected publications based on recency,
importance to the field, and/or relevance to the candidate’s proposed research. Candidates without 15
publications may substitute the following in lieu of publications:
o Original research and theoretical treatises;
o Non-experimental articles, e.g., review of literature in field, book chapters, etc.;
o Books, pamphlets, etc.
For each publication, list the authors in published sequence, full title of article, journal, volume number,
page numbers, and year of publication. Indicate if you previously used another name that is reflected in
any of the citations. URLs or NIH PubMed Central (PMC) submission identification numbers may be
included along with the full reference. While there is no limit to the number of URLs or PMC submission
identification numbers that can be cited, applicants should be both judicious and concise.
Do not include manuscripts submitted or in preparation.
When citing articles that fall under the Public Access Policy, were authored or co-authored by the
applicant and arose from NIH support, provide the NIH Manuscript Submission reference number (e.g.,
NIHMS97531) or the PubMed Central (PMC) reference number (e.g., PMCID234567) for each article. If
the PMCID is not yet available because the Journal submits articles directly to PMC on behalf of their
authors, indicate "PMC Journal - In Process." A list of these Journals is posted at:
http://publicaccess.nih.gov/submit_process_journals.htm
. Citations that are not covered by the Public
Access Policy, but are publicly available in a free, online format may include URLs or PubMed ID
(PMID) numbers along with the full reference (note that copies of publicly available publications are not
accepted as appendix material.)
Research Support
List both selected ongoing and completed (during the last three years) research projects (Federal or non-
Federal support). Begin with the projects that are most relevant to the research proposed in this
application. Briefly indicate the overall goals of the projects and responsibilities of the senior/key person
identified on the Biographical Sketch. Do not include number of person months or direct costs.
Don’t confuse “Research Support” with “Other Support.” Though they sound similar, these parts of the
application are very different. As part of the biosketch section of the application, “Research Support”
highlights your accomplishments, and those of your colleagues, as scientists. This information will be
used by the reviewers in the assessment of each individual’s qualifications for a specific role in the
proposed project, as well as to evaluate the overall qualifications of the research team. In contrast, “Other
Support” information is required for all applications that are selected to receive grant awards. NIH staff

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-137
will request complete and up-to-date “other support” information from you after peer review. This
information will be used to check that the proposed research has not already been Federally-funded.
7.4.4.2 Mentor, Co-mentor, and Other Senior/Key Persons
The mentored K awards require a primary mentor, and there may be co-mentor(s), consultants and
contributors. All individuals who have committed to contribute to the scientific development and
execution of the project, including mentors and co-mentors, should be identified as senior/key personnel,
even if they are not committing any specified measurable effort to the proposed project. Mentors and co-
mentors should be assigned the Project Role of “Other Professional” and then enter “Mentor” or “Co-
mentor” in the Other Project Role Category field.
Consultants should also be assigned the “Other Professional” role even if they are not committing any
specified measurable effort. Then, enter the specific project role under “Other Project Role Category.”
Any individuals identified as senior/key personnel who are committing specified measurable effort should
be appropriately assigned under Project Role (and Other Project Role Category, if necessary). Additional
information can be found in Section 4.5.1.
Current and Pending Support for Mentors/Co-mentors: For Mentored Career Development Awards,
as part of the application submission modified Current and Pending Support pages must be submitted for
the mentor and co-mentor(s), but not for the candidate, on the R&R Senior/Key Person Profile
(Expanded) page. Provide information on the following selected items for the mentor’s and co-mentor’s
current and pending research support relevant to the candidate’s research plan. Each attachment is limited
to 4 pages. Note, Current and Pending Support for the Candidate will be requested on a Just-In-Time
basis.
Special Instructions for Selected Items of Current & Pending Support for Mentor/Co-Mentors
Project Number: If applicable, include a code or identifier for the project.
Source: Identify the agency, institute, foundation, or other organization that is providing the support.
Major Goals: Provide a brief statement of the overall objectives of the project, subproject, or
subcontract.
Dates of Approved/Proposed Project: Indicate the inclusive dates of the project as approved/proposed.
For example, in the case of NIH support, provide the dates of the approved/proposed competitive
segment.
Annual Direct Costs: In the case of an active project, provide the current year’s direct cost budget. For a
pending project, provide the proposed direct cost budget for the initial budget period.
Do not include information on overlap and level of effort.
For non-mentored CDAs: Candidates for non-mentored CDAs should not submit Other Support Pages
at the time of application unless specified to do so in the applicable FOA.
Updated information on all active support for the candidate, mentor(s), co-mentor(s), and senior/key
personnel may be requested by the awarding component prior to award.
Biographical Sketch for Mentor/Co-mentor and Other Senior/Key Person: For the biographical
sketch for all individuals other than the candidate, follow the biographical sketch instructions found in
Part I.4.5.
Note: K22 and K99/R00 candidates should follow instruction in the specific FOA regarding senior/key
personnel.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-138
7.4.5 Special Instructions for 4.6 Selecting the Appropriate Budget
Form
K award mechanisms are not modular; therefore, only the R&R budget form is applicable and only a few
budget categories are actually used. Information regarding allowable costs for the candidate and any
allowable research development or other costs is included in each K program FOA. Candidates are
advised to contact the targeted awarding component if uncertain about allowable amounts for the
applicable K award mechanism, keeping in mind that amounts vary with awarding components. The
application forms package associated with CDA funding opportunities includes the R&R Budget Form.
Instructions for completing the R&R Budget Form are provided below. Additional guidance may also be
provided in the specific funding opportunity announcement.
Note: NIH intramural candidates applying for transitional career award support (e.g., K22, K99/R00)
should follow instructions in the applicable FOA. For the mentored phase of these awards, budgets are
negotiated with the sponsoring intramural laboratory. For awardees who receive approval to transition to
the extramural phase, a budget will be required as part of the extramural sponsored application.
7.4.6 Special Instructions for 4.7 R&R Budget Form
Follow the instructions provided in Part I.4.7 with the following exceptions:
4.7.1. A. Senior/Key Person: In general this section should include the name of the candidate only. Do
not include the mentor(s) or any other senior/key persons. For the candidate, provide the base salary,
person months, and requested salary and fringe benefits. For person months, be reminded that K programs
include a minimum effort requirement, usually 75% or 9 academic person months. For the salary column,
most NIH ICs limit the amount of salary provided for K programs. However, applicants should include
information on actual institutional base salary and the actual amount of salary and fringe being requested.
ICs may request updated salary information prior to award. Any adjustments based on policy limitations
will be made at the time of the award.
4.7.1. B. Other Personnel: In general, leave this section blank.
4.7.2. C – E: Leave these sections blank.
4.7.3 F. Other Direct Costs: In the Material and Supplies field (F.1), enter the total research
development support being requested for the initial year of the K award. Usually, a specific total amount
is allowed for research development and other costs (tuition, fees, research supplies, equipment, computer
time, travel, etc) that do not require individual cost category identification. Unless instructed differently in
the applicable FOA, applicants should enter only the total requested research development support
amount in this box. All remaining budget fields in this section should be left blank.
Please note that while this method of entering only the total requested research development support
(RDS) costs in section F will be simplest for most applicants, some applicants, including some system-to-
system applicants, may instead choose to enter those costs in the applicable detailed budget categories.
Please note that when choosing this option it is still the applicant’s responsibility to make certain the total
RDS costs do not exceed the allowable total. If there are no costs within the research development support
costs that affect the Indirect Cost Base calculation, the total RDS should be entered in total in F.1.
4.7.3. H. Indirect Costs: For all K applications, F&A/indirect costs are reimbursed at 8% of modified
total direct costs (exclusive of tuition and fees and expenditures for equipment) rather than on the basis of
a negotiated rate agreement. Follow the instructions in the chart below for completing this section.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-139
Field Name Instructions
Indirect Cost Type Indicate the Indirect Cost type as Modified Total Direct Costs.
Indirect Cost Rate (%) Indicate the indirect cost rate (also known as Facilities & Administrative
Costs [F&A]) as 8%.
Indirect Cost Base ($) Enter the amount of the base for the indirect cost type.
Funds Requested Enter the funds requested for the indirect cost type.
Total Indirect Costs The total funds requested for indirect costs.
Cognizant Federal Agency Enter “Not Applicable.” Alternatively, applicants may provide the name
of the cognizant Federal agency, name, and phone number of the
individual responsible for negotiating your rate. Either response is
acceptable since indirect costs will be reimbursed as 8% of modified total
direct costs rather than on the basis of a negotiated rate agreement.
4.7.3.K Budget Justification: Use this to provide a detailed description and justification for specific
items within the Research Development Support costs; e.g., all equipment, supplies, and other personnel
that will be used to help achieve the career development and research objectives of this award.
7.4.7 Special Instructions for 5. Completing PHS 398 Forms
5.1 Overview
In conjunction with the SF424 (R&R) forms, NIH and other PHS agencies grants applicants should also
complete and submit additional forms titled “PHS 398.” Note the PHS 398 forms include additional data
required by the agency for a complete application. While these are not identical to the PHS 398
application form pages, the PHS 398 reference is used to distinguish these additional data requirements
from the data collected in the SF424 (R&R) forms. A complete application to NIH and other PHS
agencies will include SF424 (R&R) and PHS 398 forms. The PHS 398 forms for the individual K
programs include:
• PHS 398 Cover Page Supplement (I.5.3): this supplements the data requirements in the R&R
Cover Form. Follow the instructions found in Part I.5.3 and in the K specific instructions below.
• PHS 398 Career Development Award Supplemental Form (I.7.5)
Complete each form using the instructions found in Part I.5 and the K specific instructions provided
below.
5.3 Cover Page Supplement Form
7. Change of Investigator / Change of Institution Questions: A change in PD/PI is not allowed
for K awards.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-140
7.5 PHS 398 Career Development Award Supplemental Form

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-141
The PHS 398 Career Development Award Supplemental Form should include sufficient information
needed for evaluation of the project, independent of any other document (e.g., previous application). Be
specific and informative, and avoid redundancies. Some sections are required for all K award applications
and some sections are only to be used when required by the FOA. Be sure to read all instructions in the
FOA before completing this section since errors could lead to incomplete or rejected applications.
Career Development Award Attachments (See also Section 2.3.2 Creating PDFs for Text Attachments
)
Although many of the sections of this application are separate PDF attachments, page limits referenced in
the instructions and/or funding opportunity announcement must still be followed. Agency validations will
include checks for page limits (and use of appropriate font). Some accommodation will be made for
sections that, when combined, must fit within a specified limitation.
Text attachments should be generated using word processing software and then converted to PDF using
PDF generating software. Avoid scanning text attachments to convert to PDF since that causes problems
for the agency handling the application. In addition, be sure to save files with descriptive file names.
Do not include any information in a header or footer of the attachments. A header will be system-
generated that references the name of the PD/PI. Page numbers for the footer will be system-generated in
the complete application, with all pages sequentially numbered.
Since a number of reviewers will be reviewing applications as an electronic document and not a paper
version, applicants are strongly encouraged to use only a standard, single-column format for the text.
Avoid using a two-column format since it can cause difficulties when reviewing the document
electronically.
Full-sized glossy photographs of material such as electron micrographs or gels must only be included
within the page limits of the Career Development Award application. The maximum size of images to be
included should be approximately 1200 x 1500 pixels using 256 colors. Figures must be readable as
printed on an 8.5 x 11 inch page at normal (100%) scale.
Candidates must use image compression such as JPEG or PNG. Do not include figures or photographs as
separate attachments either in the Appendix or elsewhere in the application.
Separate Attachments
Separate attachments have been designed for the Career Development Award Supplemental Form
sections to maximize automatic validations conducted by the eRA system. When the application is
received by the agency, all of the CDA Supplemental Form sections will be concatenated in the
appropriate order so that reviewers and agency staff will see a single cohesive application.
When attaching a PDF document to the actual forms, please note you are attaching an actual document,
not just pointing to the location of an externally stored document. Therefore, if you revise the document
after it has been attached, you must delete the previous attachment and then reattach the revised document
to the application form. Use the View Attachment button to determine if the correct version has been
attached.
Page Limits
Follow the page limits for the Career Development Award Supplemental Form in the table of page
limits (Table 2.6-1
), unless specified otherwise in the FOA. All tables, graphs, figures, diagrams, and
charts must be included within the 12-page limit (note that this may span to 15 pages in the eRA
Commons application image due to white space inserted at the end of sections when separating files).
Follow page limitations as specified in Funding Opportunity Announcements (FOAs).

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-142
All applications and proposals for NIH funding must be self-contained within specified page limits.
Agency validations will include checks for page limits. Note that while these computer validations will
help minimize incomplete and/or non-compliant applications, they do not replace the validations
conducted by NIH staff. Applications found not to comply with the requirements may be delayed in the
review process. Unless otherwise specified in an NIH solicitation, internet website addresses (URLs) may
not be used to provide information necessary to the review because reviewers are under no obligation to
view the internet sites. Moreover, reviewers are cautioned that they should not directly access an internet
site as it could compromise their anonymity.
Applicants are prohibited from using the Appendix to circumvent page limitations in any section of the
application for which a page limit applies.” For additional information regarding Appendix material and
page limits, please refer to the NIH Guide Notice NOT-OD-11-080,
http://grants.nih.gov/grants/guide/notice-files/NOT-OD-11-080.html
.
Notice of Proprietary Information
Applicants are discouraged from submitting information considered proprietary unless it is deemed
essential for proper evaluation of the application. However, when the application contains information
that constitutes trade secrets, or information that is commercial or financial, or information that is
confidential or privileged, make sure you have checked “Yes” for proprietary/privileged information
question in the Other Project Information form.
When information in the application constitutes trade secrets or information that is commercial or
financial, or information that is confidential or privileged, it is furnished to the Government in confidence
with the understanding that the information shall be used or disclosed only for evaluation of this
application. However, if a grant is awarded as a result of or in connection with the submission of this
application, the Government shall have the right to use or disclose the information to the extent
authorized by law. This restriction does not limit the Government’s right to use the information if it is
obtained without restriction from another source.
Although the grantee institution and the PD/PI will be consulted about any such release, the PHS will
make the final determination. Any indication by the applicant that the application contains proprietary or
privileged information does not automatically shield the information from release in response to a
Freedom of Information Act (FOIA) request should the application result in an award (see
45 CFR Part
5). If an applicant fails to identify proprietary information at the time of submission as instructed in the
application guide, a significant substantive justification will be required to withhold the information if
requested under FOIA.
Research Plan
A Research Plan is required for all types of individual K awards. The Research Plan is a major part of the
research career development plan. It is important to relate the research to the candidate's scientific career
goals. Describe how the research, coupled with other developmental activities, will provide the
experience, knowledge, and skills necessary to achieve the objectives of the career development plan and
launch and conduct an independent research career, or enhance an established research career. For
mentored K awards, explain the relationship between the candidate’s research on the CDA and the
mentor’s ongoing research program.
For most types of research, the plan should include: a specific hypothesis; a list of the specific aims and
objectives that will be used to examine the hypothesis; a description of the
methods/approaches/techniques to be used in each aim; a discussion of possible problems and how they
will be managed; and, when appropriate, alternative approaches that might be tried if the initial
approaches do not work.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-143
The Research Plan of a CDA is expected to be appropriate for, and tailored to the experience level of the
candidate, and allow him/her to develop the necessary skills needed for further career advancement, and
reviewers will evaluate the plan accordingly. The plan should be achievable within the requested time
period. Pilot or preliminary studies and routine data gathering are generally not appropriate as the sole
part(s) of a CDA research plan. Although candidates for mentored K awards are expected to write the
Research Plan, the mentor should review a draft of the plan and discuss it in detail with the candidate.
Review by other knowledgeable colleagues is also helpful. Although it is understood that CDA
applications do not require the extensive detail usually incorporated into regular research applications, a
fundamentally sound Research Plan and a reasonably detailed Approach section should be provided.
In general, less detail will be expected in descriptions of research planned for the future years of the
proposed CDA. However, sufficient detail should be provided to enable the peer reviewers to determine
that the plans for those years, including the approach to be used, are worthwhile and are likely to enable
the candidate to achieve the objectives of the Research Plan.
The PHS 398 Career Development Award Supplemental Form is comprised of sections for: Candidate
Information; Statement of Support (Mentors); Environment & Institutional Commitment to the Candidate;
and the Research Plan (including Human Subjects and Other Research Plan Sections).
Begin each text section of the Candidate Information and Research Plan with a section header (e.g.,
Introduction, Specific Aims, Background & Significance, etc). See Specific FOA for additional
information.
Field Name Instructions
1. Introduction to
Application (for
RESUBMISSION
applications only)
NIH policy allows a thirty-seven month window for resubmissions (A1
applications) following the submission of a New, Renewal, or Revision
application (A0 application). See NIH Notice NOT-OD-12-128
for
additional information/clarification of NIH policy.
Use only if Type of Application is Resubmission. Resubmission
applications must include an Introduction to Resubmission Application,
not to exceed one page. The Introduction must include responses to the
criticisms and issues raised in the Summary Statement. Summarize the
substantial additions, deletions, and changes. In the body of the
application, highlight paragraphs with significant changes by bracketing
and changing typography.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
Candidate Information
Field Name Instructions
2. Candidate’s Background This attachment is required. Follow the page limits for the Career
Development Award Supplemental Form in the table of page limits
(Table 2.6-1
), unless specified otherwise in the FOA.
Use this section to provide any additional information not described in the
Biographical Sketch Format Page such as research and/or clinical training
experience.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-144
Field Name Instructions
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
3. Career Goals and
Objectives
This attachment is required. Follow the page limits for the Career
Development Award Supplemental Form in the table of page limits
(Table 2.6-1
), unless specified otherwise in the FOA.
Describe your past scientific history, indicating how the award fits into
past and future research career development. If there are consistent
themes or issues that have guided previous work, these should be made
clear; if your work has changed direction, the reasons for the change
should be indicated. It is important to justify the award and how it will
enable you to develop or expand your research career. You may include a
timeline, including plans to apply for subsequent grant support.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
4. Candidate’s Plan for
Career Development/
Training Activities During
Award Period
This attachment is required. Follow the page limits for the Career
Development Award Supplemental Form in the table of page limits
(Table 2.6-1
), unless specified otherwise in the FOA.
Stress the new enhanced research skills and knowledge you will acquire
as a result of the proposed award. If you have considerable research
experience in the same areas as the proposed research, reviewers may
determine that the application lacks potential to enhance your research
career. For mentored awards, describe structured activities, such as course
work or technique workshops, which are part of the developmental plan.
If course work is included, provide course numbers and descriptive titles.
Briefly discuss each of the activities, except research, in which you expect
to participate. Include a percentage of time involvement for each activity
by year, and explain how the activity is related to the proposed research
and the career development plan.
Note that recipients of mentored K awards may receive concurrent
support from an NIH research grant award or cooperative agreement only
under certain conditions (see NIH Notice NOT-OD-08-065
).
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
5. Training in the
Responsible Conduct of
Research
This attachment is required. Follow the page limits for the Career
Development Award Supplemental Form in the table of page limits
(Table 2.6-1
), unless specified otherwise in the FOA.
For mentored career development awards, describe a plan to acquire
instruction in the responsible conduct of research. For independent career
awards, describe a plan to obtain or provide instruction in the responsible
conduct of research. See Supplemental Instructions Part III Section 1.16

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-145
Field Name Instructions
for information on the NIH Policy on Training in the Responsible
Conduct of Research (RCR).
Attach a description of plans for obtaining instruction in the responsible
conduct of research. This section should document prior instruction or
participation in RCR training during the applicant’s current career stage
(including the date instruction was last completed) and propose plans to
either receive instruction or participate as a course lecturer, etc., in order
to meet the once every four-year requirement. The plan should address
how applicants plan to incorporate the five instructional parts outlined in
the NIH Policy on Instruction in the Responsible Conduct of Research:
format, subject matter, faculty participation, duration, and frequency. The
plan may include career stage-appropriate individualized instruction or
independent scholarly activities that will enhance the applicant’s
understanding of ethical issues related to their specific research activities
and the societal impact of that research. The role of the mentor in RCR
instruction must be described.
Where applicable, Renewal applications must describe the RCR
instruction activities undertaken during the project period as well as future
plans.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
6. Candidate’s Plan to
Provide Mentoring
(Include only when
required by the specific
FOA, e.g., K24 and K05)
The plan should provide information about the candidate’s commitment to
serve as a mentor to other investigators, and describe previous mentoring
activities. The plan should describe the setting and provide information
about the available pool of mentees with appropriate backgrounds and
interests in the same field of science. It should also include information on
the candidate’s past and proposed mentees sufficient to evaluate the
quality of prior mentoring experiences, including the professional levels
of mentees, and the frequency and kinds of mentoring interactions
between the candidate and the mentees. Describe the productivity of the
mentoring relationship for the scientific development of the new scientists
as judged by their publications and current research activities. Senior level
(K05) candidates should describe any financial and material support from
their own funded research and research resources that will be available to
their mentees. The candidate’s proposed percent effort commitment to the
mentoring plan should also be stated.
Follow the page limits for the Career Development Award Supplemental
Form in the table of page limits (Table 2.6-1
), unless specified otherwise
in the FOA. Save this information in a single file in a location you
remember. Click Add Attachment, browse to where you saved the file,
select the file, and then click Open.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-146
Statements and Letters of Support
Field Name Instructions
7. Plans and Statements of
Mentor and Co-mentor(s)
This section is to be completed by the mentor and co-mentor(s), as
appropriate. The letters must be appended together and uploaded as a
single pdf file. Follow the page limits for the Career Development Award
Supplemental Form in the table of page limits (Table 2.6-1
), unless
specified otherwise in the FOA.
For mentored awards (see Summary of Career Development Award
Mechanisms table), the mentor must explain how they will contribute to
the development of the candidate's research career. This statement should
include all of the following:
1. The plan for the candidate's training and research career
development. This description must include not only research, but
also other developmental activities, such as seminars, scientific
meetings, training in the responsible conduct of research, and
presentations. It should discuss expectations for publications over the
entire period of the proposed project and define what aspects of the
proposed research project the candidate will be allowed to take with
him/her to start their own research program.
2. The source of anticipated support for the candidate’s research project
for each year of the award period.
3. The nature and extent of supervision and mentoring of the candidate,
and commitment to the candidate's development that will occur
during the award period.
4. The candidate's anticipated teaching load for the period of the award
(number and types of courses or seminars), clinical responsibilities,
committee and administrative assignments, and the portion of time
available for research.
5. A plan for transitioning the candidate from the mentored stage of
his/her career to the independent investigator stage by the end of the
project period of the award. The mentor should describe previous
experience as a mentor, including type of mentoring (e.g., graduate
students, career development awardees, postdoctoral students),
number of persons mentored, and career outcomes.
All mentored career development applications should identify any and all
co-mentors involved with the proposed research and career development
program. Co-mentors must specifically address the nature of their role in
the career development plan and how the responsibility for the
candidate’s development is shared with the mentor. Describe respective
areas of expertise and how they will be combined to enhance the
candidate’s development. Also describe the nature of any resources that
will be committed to this CDA. Letters from the mentor(s) and co-
mentor(s) documenting their role and willingness to participate in the
project must be included in this section of the application. Do not place
these letters in the Appendix.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-147
Field Name Instructions
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
8. Letters of Support from
Collaborators, Contributors,
and Consultants
Attach all appropriate letters of support. Letters are not required for
personnel (such as research assistants) not contributing in a substantive,
measurable way to the scientific development or execution of the project.
For consultants, letters should include rates/charges for consulting
services.
All mentored career development award applications should identify
any and all collaborators, contributors, and consultants involved with the
proposed research and career development program not already included
in Item 7. Letters should briefly describe their anticipated contributions
and document their role and willingness to participate in the project.
Additionally, non-mentored career development award applications
should include letters from collaborators, consultants and contributors
listing their proposed roles and documenting their willingness to
participate in the project. The letters should also briefly describe research
materials, data, guidance, or advice each person will provide.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, and the click
Open.
Environment and Institutional Commitment to the Candidate
Field Name Instructions
9. Description of
Institutional Environment
The sponsoring institution must document a strong, well-established
research program related to the candidate's area of interest, including the
names of key faculty members relevant to the candidate's proposed
developmental plan. Referring to the resources description (See section
4.4.10 Facilities and Other Resources), indicate how the necessary
facilities and other resources will be made available for career
enhancement as well as the research proposed in this application.
Describe opportunities for intellectual interactions with other
investigators, including courses offered, journal clubs, seminars, and
presentations. This attachment is required and is limited to one page.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
10. Institutional
Commitment to
Candidate’s Research
Career Development
Introduction
The institution should provide a document on institutional letterhead that
describes its commitment to the candidate and the candidate’s career
development, independent of the receipt of the CDA. The document

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-148
Field Name Instructions
should include the institution’s agreement to provide adequate time and
support for the candidate to devote the proposed protected time to
research and career development for the entire period of the proposed
award. The institution should provide the equipment, facilities, and
resources necessary for a structured research career development
experience. It is essential to document the institution's commitment to the
retention, development and advancement of the candidate during the
period of the award.
Because of the diverse types of K awards, applicants should contact the
appropriate awarding component Scientific/Research contact listed in the
specific FOA to determine the level of commitment required for this
application.
Agreement
The applicant organization must:
a. Agree to release the candidate from other duties and activities to
devote the required percentage of time for development of a research
career. For most K awards, commitment of at least 75 percent of
time is required. Describe actions that will be taken to ensure this;
e.g., reduction of the candidate's teaching load, committee and
administrative assignments, and clinical or other professional
activities for the current academic year. (For example, describe the
actions that will be taken to compensate for the reduction in clinic
responsibilities of the candidate, e.g., hiring of additional staff).
Describe the candidate's academic appointment, bearing in mind that
it must be full-time, and that the appointment (including all rights
and privileges pertaining to full faculty status if in an academic
setting) and the continuation of salary should not be contingent upon
the receipt of this award. Describe the proportion of time currently
available for the candidate's research experience and what the
candidate's institutional responsibilities will be if an award is made.
b. Provide the candidate with appropriate office and laboratory space,
equipment, and other resources and facilities (including access to
clinical and/or other research populations) to carry out the proposed
Research Plan.
c. Provide appropriate time and support for any proposed mentor(s)
and/or other staff consistent with the career development plan.
Signatures
The institutional commitment must be dated and signed by the person
who is authorized to commit the institution to the agreements and
assurances listed above. In most cases, this will be the dean or the
chairman of the department. The signature must appear over the signer's
name and title at the end of the statement. If the candidate will be working
away from the home institution, signatures from both the home and the

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-149
Field Name Instructions
host institution are required.
The sponsoring institution, through the submission of the application and
in the institutional commitment section, certifies that all items outlined
above will be provided and that the institution will abide by the applicable
assurances and PHS policies. See: NOT-OD-06-054
.
This attachment is required and is limited to one page. Create a single file
of the institutional letter and save it in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
Research Plan
Field Name Instructions
11. Specific Aims State precisely the goals of the proposed research and summarize the
expected outcome(s) including the impact that the results of the proposed
research will exert on the research field(s) involved.
List succinctly the specific objectives of the research proposed, e.g., to
test a stated hypothesis, create a novel design, solve a specific problem,
challenge an existing paradigm or clinical practice, address a critical
barrier to progress in the field, or develop new technology.
The Specific Aims attachment is required. Follow the page limits for the
Career Development Award Supplemental Form in the table of page
limits (Table 2.6-1
), unless specified otherwise in the FOA.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
12. Research Strategy Follow the page limits for the Career Development Award Supplemental
Form in the table of page limits (Table 2.6-1
), unless specified otherwise
in the FOA.
Organize the Research Strategy in the specified order and using the
instructions provided below. Start each section with the appropriate
section heading – Significance, Innovation, Approach. Cite published
experimental details in the Research Strategy section and provide the full
reference in the Bibliography and References Cited section (
Part I Section
4.4.9).
1. Significance
• Explain the importance of the problem or critical barrier to
progress in the field that the proposed project addresses.
• Explain how the proposed project will improve scientific
knowledge, technical capability, and/or clinical practice in
one or more broad fields.
• Describe how the concepts, methods, technologies,

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-150
Field Name Instructions
treatments, services, or preventative interventions that drive
this field will be changed if the proposed aims are achieved.
2. Innovation
• Explain how the application challenges current research or
clinical practice paradigms.
• Describe any novel theoretical concepts, approaches or
methodologies, instrumentation or interventions to be
developed or used, and any advantage over existing
methodologies, instrumentation, or interventions.
3. Approach
• Describe the overall strategy, methodology, and analyses to
be used to accomplish the specific aims of the project. Unless
addressed separately in the Resource Sharing Plan, include
how the data will be collected, analyzed, and interpreted as
well as any resource sharing plans as appropriate.
• Discuss potential problems, alternative strategies, and
benchmarks for success anticipated to achieve the aims.
• If the project is in the early stages of development, describe
any strategy to establish feasibility, and address the
management of any high risk aspects of the proposed work.
• Point out any procedures, situations, or materials that may be
hazardous to personnel and precautions to be exercised. A full
discussion on the use of select agents should appear in Item
18 below.
• If research on Human Embryonic Stem Cells (hESCs) is
proposed but an approved cell line from the NIH hESC
Registry cannot be identified, provide a strong justification
for why an appropriate cell line cannot be chosen from the
Registry at this time.
If an applicant has multiple Specific Aims, then the applicant may address
Significance, Innovation and Approach for each Specific Aim
individually, or may address Significance, Innovation and Approach for
all of the Specific Aims collectively.
As applicable, also include the following information as part of the
Research Strategy, keeping within the three sections listed above:
Significance, Innovation, and Approach.
Preliminary Studies for New Applications: For new applications,
include information on Preliminary Studies. Discuss the PD/PI’s
preliminary studies, data, and or experience pertinent to this application.
Progress Report for Renewal and Revision Applications. For
renewal/revision applications, provide a Progress Report. Provide the
beginning and ending dates for the period covered since the last
competitive review. Summarize the specific aims of the previous project

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-151
Field Name Instructions
period and the importance of the findings, and emphasize the progress
made toward their achievement. Explain any significant changes to the
specific aims and any new directions including changes to the specific
aims and any new directions including changes resulting from significant
budget reductions. A list of publications, patents, and other printed
materials should be included in the Progress Report Publication List; do
not include that information here.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
13. Progress Report
Publication List
(for RENEWAL
applications only)
List the titles and complete references to all appropriate publications,
manuscripts accepted for publication, patents, and other printed materials
that have resulted from the project since it was last reviewed
competitively. When citing articles that fall under the Public Access
Policy, were authored or co-authored by the applicant and arose from NIH
support, provide the NIH Manuscript Submission reference number (e.g.,
NIHMS97531) or the PubMed Central (PMC) reference number (e.g.,
PMCID234567) for each article. If the PMCID is not yet available
because the Journal submits articles directly to PMC on behalf of their
authors, indicate “PMC Journal – In Process.” A list of these journals is
posted at: http://publicaccess.nih.gov/submit_process_journals.htm
.
Citations that are not covered by the Public Access Policy, but are
publicly available in a free, online format may include URLs or PubMed
ID (PMID) numbers along with the full reference (note that copies of
these publications are not accepted as appendix material).
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
Human Subjects Sections
Field Name Instructions

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-152
Field Name Instructions
14. Protection of Human
Subjects
Refer to Part II, Supplemental Instructions for Preparing the Human
Subjects Section of the Research Plan. Complete this section if you
answered “yes” to the question “are human subjects involved?” on the
R&R Other Project Information Form. If you answered “no” to the
question but your proposed research involves human specimens and/or
data from subjects you must provide a justification in this section for your
claim that no human subjects are involved.
Do not use the protection of human subjects section to circumvent the
page limits of the Research Strategy.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
15. Inclusion of Women
and Minorities
Refer to Part II, Supplemental Instructions for Preparing the Human
Subjects Section of the Research Plan. Complete this section if you
answered “yes” to the question “are human subjects involved?” on the
R&R Other Project Information Form and the research does not fall under
Exemption 4.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
16. Inclusion of Children Refer to Part II Supplemental Instructions for Preparing the Human
Subjects Section of the Research Plan, Sections 4.4 and 5.7 Complete this
section if you answered “yes” to the question “are human subjects
involved?” on the R&R Other Project Information Form and the research
does not fall under Exemption 4.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-153

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-154
Other Research Plan Sections
Field Name Instructions
17. Vertebrate Animals Complete this section if you answered “yes” to the question “are
Vertebrate Animals Used?” on the R&R Other Project Information Form.
If Vertebrate Animals are involved in the project, address each of the five
points below. This section should be a concise, complete description of
the animals and proposed procedures. While additional details may be
included in the Research Strategy, the responses to the five required
points below must be cohesive and include sufficient detail to allow
evaluation by peer reviewers and NIH staff. If all or part of the proposed
research involving vertebrate animals will take place at alternate sites
(such as project/performance or collaborating site(s)), identify those sites
and describe the activities at those locations. Although no specific page
limitation applies to this section of the application, be succinct. Failure to
address the following five points will result in the application being
designated as incomplete and will be grounds for the PHS to defer the
application from the peer review round. Alternatively, the application’s
impact/priority score may be negatively affected.
If the involvement of animals is indefinite, provide an explanation and
indicate when it is anticipated that animals will be used. If an award is
made, prior to the involvement of animals the grantee must submit to the
NIH awarding office detailed information as required in points 1-5 below
and verification of IACUC approval. If the grantee does not have an
Animal Welfare Assurance then an applicable Animal Welfare Assurance
will be required (See Supplemental Instructions
Part III Section 2.2
Vertebrate Animals for more information).
The five points are as follows:
1. Provide a detailed description of the proposed use of the animals in
the work outlined in the Research Strategy section. Identify the
species, strains, ages, sex, and numbers of animals to be used in the
proposed work.
2. Justify the use of animals, the choice of species, and the numbers to
be used. If animals are in short supply, costly, or to be used in large
numbers, provide an additional rationale for their selection and
numbers.
3. Provide information on the veterinary care of the animals involved.
4. Describe the procedures for ensuring that discomfort, distress, pain,
and injury will be limited to that which is unavoidable in the conduct
of scientifically sound research. Describe the use of analgesic,
anesthetic, and tranquilizing drugs and/or comfortable restraining
devices, where appropriate, to minimize discomfort, distress, pain,
and injury.
5. Describe any method of euthanasia to be used and the reasons for its
selection. State whether this method is consistent with the
recommendations of the American Veterinary Medical Association

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-155
Field Name Instructions
(AVMA) Guidelines on Euthanasia. If not, include a scientific
justification for not following the recommendations.
For additional information, see
http://grants.nih.gov/grants/olaw/VASchecklist.pdf
Do not use the vertebrate animal section to circumvent the page limits of
the research strategy.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
For those applicants familiar with the PHS 398, please note that the
Literature Cited section of the Research Plan is now captured as
“Bibliography & References Cited” in the Other Project Information
Form.
18. Select Agent Research Select agents are hazardous biological agents and toxins that have been
identified by DHHS or USDA as having the potential to pose a severe
threat to public health and safety, to animal and plant health, or to animal
and plant products. CDC maintains a list of these agents. See
http://www.cdc.gov/od/sap/docs/salist.pdf
.
If the activities proposed in your application involve only the use of a
strain(s) of select agents which has been excluded from the list of select
agents and toxins as per 42 CFR 73.4(f)(5), the select agent requirements
do not apply. Use this section to identify the strain(s) of the select agent
that will be used and note that it has been excluded from this list. The
CDC maintains a list of exclusions at
http://www.cdc.gov/od/sap/sap/exclusion.htm
.
If the strain(s) is not currently excluded from the list of select agents and
toxins but you have applied or intend to apply to DHHS for an exclusion
from the list, use this section to indicate the status of your request or your
intent to apply for an exclusion and provide a brief justification for the
exclusion.
If any of the activities proposed in your application involve the use of
select agents at any time during the proposed project period, either at the
applicant organization or at any other performance site, address the
following three points for each site at which select agent research will
take place. Although no specific page limitation applies to this section, be
succinct.
1. Identify the select agent(s) to be used in the proposed research.
2. Provide the registration status of all entities* where select agent(s)
will be used.
• If the performance site(s) is a foreign institution, provide the
name(s) of the country or countries where select agent research
will be performed.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-156
Field Name Instructions
*An “entity” is defined in 42 CFR 73.1 as “any government
agency (Federal, State, or local), academic institution,
corporation, company, partnership, society, association, firm, sole
proprietorship, or other legal entity.”
3. Provide a description of all facilities where the select agent(s) will be
used.
• Describe the procedures that will be used to monitor possession,
use and transfer of the select agent(s).
• Describe plans for appropriate biosafety, biocontainment, and
security of the select agent(s).
• Describe the biocontainment resources available at all
performance sites.
If you are responding to a specific FOA, address any requirements
specified by the FOA.
Reviewers will assess the information provided in this Section, and any
questions associated with select agent research will need to be addressed
prior to award.
Save this file in a location you remember. Click Add Attachment,
browse to where you saved the file, select the file, and then click Open.
19. Consortium/Contractual
Arrangements
Explain the programmatic, fiscal, and administrative arrangements to be
made between the applicant organization and the consortium
organization(s). If consortium/contractual activities represent a significant
portion of the overall project, explain why the applicant organization,
rather than the ultimate performer of the activities, should be the grantee.
The signature of the Authorized Organization Representative on the
SF424 (R&R) cover form signifies that the applicant and all proposed
consortium participants understand and agree to the following statement:
The appropriate programmatic and administrative personnel of
each organization involved in this grant application are aware of
the agency’s consortium agreement policy and are prepared to
establish the necessary inter-organizational agreement(s)
consistent with that policy.
A separate statement is no longer required.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
20. Resource Sharing
Plan(s)
NIH considers the sharing of unique research resources developed
through NIH-sponsored research an important means to enhance the value
and further the advancement of the research. When resources have been
developed with NIH funds and the associated research findings published
or provided to NIH, it is important that they be made readily available for
research purposes to qualified individuals within the scientific

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-157
Field Name Instructions
community. See Supplemental Instructions Part III, 1.5 Sharing Research
Resources.
1. Data Sharing Plan: Investigators seeking $500,000 or more in direct
costs (exclusive of consortium F&A) in any year are expected to include a
brief 1-paragraph description of how final research data will be shared, or
explain why data-sharing is not possible. Specific Funding Opportunity
Announcements may require that all applications include this information
regardless of the dollar level. Applicants are encouraged to read the
specific opportunity carefully and discuss their data-sharing plan with
their program contact at the time they negotiate an agreement with the
Institute/Center (IC) staff to accept assignment of their application. See
Data-Sharing Policy or
http://grants.nih.gov/grants/guide/notice-
files/NOT-OD-03-032.html.
2. Sharing Model Organisms: Regardless of the amount requested, all
applications where the development of model organisms is anticipated are
expected to include a description of a specific plan for sharing and
distributing unique model organisms or state why such sharing is
restricted or not possible. See Sharing Model Organisms Policy, and
NIH
Guide NOT-OD-04-042.
3. Genome Wide Association Studies (GWAS): Applicants seeking
funding for a genome-wide association study are expected to provide a
plan for submission of GWAS data to the NIH-designated GWAS data
repository, or an appropriate explanation why submission to the
repository is not possible. GWAS is defined as any study of genetic
variation across the entire genome that is designed to identify genetic
associations with observable traits (such as blood pressure or weight) or
the presence or absence of a disease or condition. For further information
see Policy for Sharing of Data Obtained in NIH Supported or Conducted
Genome-Wide Association Studies, NIH Guide NOT-OD-07-088
, and
http://gwas.nih.gov/.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
21. Appendix Do not use the appendix to circumvent the page limits of the Candidate
Information and the Research Strategy or any other section of the
application for which a page limit applies. For additional information
regarding Appendix material and page limits, please refer to the NIH
Guide Notice NOT-OD-11-080,
http://grants.nih.gov/grants/guide/notice-
files/NOT-OD-11-080.html.
Only one copy of appendix material is necessary. Use the Add
Attachments button to the right of this field to complete this entry.
Use the Add Attachments button to complete this entry. A maximum of
10 PDF attachments is allowed in the Appendix. If more than 10 appendix
attachments are needed, combine the remaining information into
attachment #10. Note that this is the total number of appendix items, not

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-158
Field Name Instructions
the total number of publications. When allowed there is a limit of 3
publications that are not publicly available (see below for further details
and check the FOA for any specific instructions), though not all grant
mechanisms allow publications to be included in the appendix.
Appendix material may not appear in the assembled application in the
order attached, so it is important to use filenames for attachments that are
descriptive of the content. A summary sheet listing all of the items
included in the appendix is also encouraged but not required. When
including a summary sheet, it should be included in the first appendix
attachment. Applications that do not follow the appendix requirements
may be delayed in the review process.
New, resubmission, renewal, and revision applications may include the
following materials in the Appendix:
• Publications – No longer allowed as appendix materials
except in the circumstances noted below. Applicants may
submit up to 3 of the following types of publications:
• Manuscripts and/or abstracts accepted for publication but not
yet published: The entire article should be submitted as a PDF
attachment.
• Manuscripts and/or abstracts published, but a free, online,
publicly available journal link is not available: The entire
article should be submitted as a PDF attachment.
• Patents directly relevant to the project: The entire document
should be submitted as a PDF attachment.
• Do not include unpublished theses, or abstracts/manuscripts
submitted (but not yet accepted) for publication.)
• Surveys, questionnaires, and other data collection instruments;
clinical protocols and informed consent documents may be
submitted in the Appendix as necessary.
• For materials that cannot be submitted electronically or materials
that cannot be converted to PDF format (e.g., medical devices,
prototypes, DVDs, CDs), applicants should contact the Scientific
Review Officer for instructions following notification of
assignment of the application to a study section. Applicants are
encouraged to be as concise as possible and submit only
information essential for the review of the application.
Items that must not be included in the appendix:
• Photographs or color images of gels, micrographs, etc., are no
longer accepted as Appendix material. These images must be
included in the Research Plan PDF. However, images embedded
in publications are allowed.
•
Publications that are publicly accessible. For such publications,

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-159
Field Name Instructions
the URL or PMC submission identification numbers along with
the full reference should be included as appropriate in the
Bibliography and References cited section, the Progress Report
Publication List section, and/or the Biographical Sketch section.
3. Citizenship
One selection is required. Please select the most appropriate response from the options provided. Other
than for the K99/R00 award program, the candidate must be a citizen or non-citizen national of the United
States or its possessions and territories, or must have been lawfully admitted to the United States for
permanent residence by the time of award.
For those K award programs with a citizenship requirement, an individual who has applied for Permanent
Residence and expects to have obtained such status prior to the time award, may submit an application
recognizing that no award will be made until legal verification of permanent resident status is provided. If
a candidate’s citizenship status changes after submission of the application, the new status should be
reported in the candidate’s Personal Profile in the eRA Commons. Before an award is issued, a permanent
resident will be required to submit a notarized statement that a licensed notary has seen the candidate’s
current and valid Permanent Resident Card or some other valid verification from the U.S. Immigration
and Naturalization Service of legal admission to the U.S. as a permanent resident.
It is the responsibility of the sponsoring institution to determine and retain documentation indicating that
the individual candidate’s visa will allow him/her to reside in the proposed research training/career
development setting for the period of time necessary to complete the approved career development
program. Information may be requested by the NIH prior to issuance of an award.
Each candidate must check the applicable box, check only one:
• U.S. Citizen or non-citizen national: Check this box if the candidate is a U.S. Citizen or
Noncitizen national. Noncitizen nationals are people, who, although not citizens of the United
States, owe permanent allegiance to the United States. They generally are people born in outlying
possessions of the United States (e.g., American Samoa and Swains Island).;
• Permanent Resident of U.S.: Check this box if the candidate has been lawfully admitted for
permanent residence; i.e., is in the possession of a current and valid Permanent Resident Card or
other legal verification of such status. A notarized statement will be required as part of the pre-
award process.
• Permanent Resident of U.S. Pending: Check this box if the candidate has applied for
Permanent Residence and expects to have obtained such status prior to the time of award. A
notarized statement will be required as part of the pre-award process.
• Non-U.S. citizen with temporary U.S. visa: This box is applicable only to specific programs
that do not require U.S. citizenship or permanent residency; e.g. K99/R00. The NIH awarding
component may request verifying information as part of the pre-award process.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-160
7.6 Career Development Award Peer Review Process
The goal of NIH-supported career development programs is to help ensure that diverse pools of highly
trained scientists are available in adequate numbers and in appropriate research areas to address the
Nation’s biomedical, behavioral, and clinical research needs. Each application must be tailored to the
individual candidate.
The general process information (Overview, Streamlining, and Dual-Level Peer Review) found in Part I.6
applies to K applications as well. However, the actual review criteria and other review considerations are
different. For K applications, the scientific review group will address individual career development
award applications by considering information provided for each of the following elements in the
application:
Review Criteria:
• Candidate
• Career Development Plan/Career Goals & Objectives/Plan to Provide Mentoring
• Research Plan
• Mentor(s), Co-Mentor(s), Consultant(s), Collaborator(s)
• Environment and Institutional Commitment to the Candidate
Additional Review Criteria include the following;
• Protection of Human Subjects from Research Risk
• Inclusion of Women, Minorities, and Children in Research
• Care and Use of Vertebrate Animals in Research
• Biohazards
• Resubmission Applications
• Renewal Applications (as applicable)
Additional Review Considerations include the following:
• Training in the Responsible Conduct of Research
• Select Agents
• Resource Sharing Plans
• Budget and Period of Support
Candidates should carefully review the applicable FOA for complete information associated with the peer
review process. The FOA will describe essential information to be submitted for each of the above
elements.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-161
8. Supplemental Instructions to the SF424 (R&R) for
Preparing Institutional Ruth L. Kirschstein
National Research Service Award (NRSA)
Application
8.1 Introduction
All applicants must use the SF 424 (R&R) Application for Federal Assistance, following the instructional
information in this section. The supplemental instructions found in this section (8) are for Institutional
National Ruth L. Kirschstein National Research Service Award (NRSA) applications and include
guidance and instructional information only when there is a difference in the required information to be
submitted or there is a need for more specificity for the institutional research training program. Therefore,
these supplemental instructions must be used along with the information found in Part I.1 – I.6 (see
specific training grant review process at the end of Section 8) of this document.
These instructions apply to NIH-supported NRSA institutional research training programs (e.g., T32, T34,
T35, T90). Some training programs are funded through Requests for Applications (RFAs) and may have
special instructions.
Additionally, there are non-NRSA programs (e.g. T15, T37, D43, D71) which include research training
under different regulatory authorities, and, while some of the information may be the same, it is important
for individuals interested in those programs to carefully read the applicable Funding Opportunity
Announcement (FOA) for specific program information and special application instructions. These
training programs may have different eligibility requirements, submission dates, award provisions, and
review criteria.
It is imperative that applicants become familiar with the NIH Research Training Activity code for which
support is being requested, and applicants should carefully review the applicable FOA which contains
more specific information associated with the award mechanism and the names of individuals that may be
contacted for additional or clarifying information prior to submission of an application. Announcements
for various training programs are issued periodically in the NIH Guide for Grants and Contracts, a weekly
publication (http://grants.nih.gov/grants/guide/index.html
).
This section includes instructions to be used when applying for competing (New, Renewal, Resubmission
or Revision) institutional training grants, including both PHS Institutional Ruth L. Kirschstein National
Research Service Awards (Kirschstein-NRSA) and non-NRSA awards. The contents include substitute
both budget pages, and instructions for the Research Training Program Plan. Begin by reading the
previous Sections 4 and 5, and then follow both sets of instructions.
Prior to preparing an application, review the Funding Opportunity Announcement (FOA) to which you
are responding and consult with the appropriate PHS awarding component
identified in the FOA. Current
NIH-wide T32, T34 or T35 Kirschstein-NRSA Program Announcements (PA) are available at
(
http://grants.nih.gov/training/nrsa.htm). Note especially the eligibility requirements, submission dates,
review criteria, award provisions, and payback provisions (when applicable). PAs are also issued
periodically by the individual NIH Institutes or Centers in the
NIH Guide for Grants and Contracts. This
information is available from the appropriate PHS agency, from grantee offices of sponsored programs, or
equivalent offices.
Please note that for Kirschstein-NRSA programs that include postdoctoral trainees, the Program Director
must explain the terms of the payback service requirement to all prospective postdoctoral training

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-162
candidates. A complete description of the service payback obligation is available in the relevant NRSA
Program Announcement or the NIH Grants Policy Statement
.
8.2 Institutional Research Training Programs
Prospective applicants are encouraged to review the T Kiosk for the most current program information.
The T Kiosk includes information on NIH-wide Parent FOAs as well as IC-specific FOAs for a particular
T programs. In addition, non-NRSA training programs are described here:
http://grants.nih.gov/training/F_files_non_nrsa.htm.
8.3 Reserved
8.4 Specific Instructions for Institutional Training Grant
Applications using the SF424 (R&R) Application
Standard instructions found in Parts I.1 – I.6 should be followed with the exceptions found in this section.
Section numbers referenced below (e.g. 4.2 - 5.6) reflect those found in Part I.
8.4.1 Special Instructions for 4.2 Cover Form
Proposed Project Start and Ending Dates
The usual starting date for an institutional Kirschstein-NRSA is July 1, but there are other possible
starting dates. Consult the webpage of Standard Due Dates for Competing Applications
(http://grants.nih.gov/grants/funding/submissionschedule.htm
). Many PHS awarding components restrict
submission and review dates to once a year. Applicants are strongly encouraged to contact the appropriate
awarding component staff before submitting an application.
8.4.2 Special Instructions for 4.3 Research & Related
Project/Performance Site Locations
List all of the locations where training, program management, and the research training experiences
described in the Research Training Program Plan (8.7) will be performed. If a Project/Performance Site
will be engaged in research involving human subjects, it is the responsibility of the applicant organization
to assure that all Project/Performance Sites comply with the human subject protection regulations in
45
CFR part 46 and NIH policies for the protection of human subjects. For research involving live vertebrate
animals, the applicant organization must supply information for all training sites where animals will be
used by trainees. The applicant organization is responsible for assuring that all Project/Performance Sites
have a current Animal Welfare Assurance and comply with the PHS Policy on Humane Care and Use of
Laboratory Animals.
8.4.3 Special Instructions for 4.4 Research & Related Other Project
Information Form
Are Human Subjects Involved?
Check “Yes” if training plans include or potentially will include involvement of trainees in projects that
include human subjects as defined by 45 CFR 46. Check “Yes” even if the proposed project is exempt

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-163
from Regulations for the Protection of Human Subjects. If no activities involving human subjects are
planned, check the No box, and skip the rest of this block. This field is required.
The institution must ensure that trainees who will be involved in the design or conduct of research
involving human subjects receive training in human subjects protections. It is the institution’s
responsibility to ensure that trainees are properly supervised when working with human subjects.
In many instances, trainees supported by institutional training grants will be participating in research
supported by research project grants for which the IRB approval or a determination of exemption exists.
Existing IRB approval is sufficient for trainees, provided that the IRB determines the research would not
be substantially modified by the participation of a trainee. The appropriate grants must be identified along
with their IRB approval dates or exemption designation in Section 7 of the Research Training Program
Plan.
Note that IRB approval information for the full training grant application is not required at the time of
submission, but will be requested as Just-in-time (JIT) information prior to award. If an award is made
and the research is not exempt from requirements stipulated in 45 CFR 46, and trainees will participate in
research for which IRB review and approval does not otherwise exist, human subjects may not be
involved and trainees may not participate in research involving human subjects unless the engaged
institution has an approved FWA on file with OHRP, certification of the date of IRB approval has been
submitted to and accepted by the PHS agency, and NIH requirements for human subjects protections have
been addressed (see instructions in
Part II, Supplemental Instructions for Preparing the Human Subjects
Section of the Research Plan, and the NIH Grants Policy Statement
(http://grants.nih.gov/grants/policy/nihgps_2012/index.htm)).
These policies apply to all Performance Sites.
Are Vertebrate Animals Used?
Check “Yes” if training plans include or potentially will include trainees in projects involving the use of
live vertebrate animals at any time during the proposed project period.
Otherwise, check “No”, and skip the rest of this block. This field is required.
In many instances, trainees supported by institutional training grants will be participating in research
supported by research project grants for which the IACUC review and approval exists. This existing
IACUC approval is sufficient for trainees, provided that the research would not be substantially modified
by the participation of a trainee. The appropriate grants must be identified along with their IACUC
approval dates in Section 8 of the Research Training Program Plan.
Note that vertebrate animal approval information for the full training grant application is not required at
the time of submission, but will be requested as Just-in-time (JIT) information prior to award. If an award
is made and trainees will participate in research for which IACUC approval does not otherwise exist,
vertebrate animals may not be involved and trainees may not participate in research utilizing vertebrate
animals unless the institution has an OLAW-approved Animal Welfare Assurance, certification of the
date of IACUC approval has been submitted to and accepted by the PHS agency, and NIH requirements
for the use of vertebrate animals have been addressed.
The institution must ensure that trainees are enrolled in the institution's animal welfare training and
occupational health and safety programs for personnel who have contact with animals. It is the
institution's responsibility to ensure that trainees are properly supervised when working with live
vertebrate animals.
These policies apply to all Performance Sites.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-164
Project Summary/Abstract
Summarize the objectives, rationale and design of the research training program. Provide information
regarding the research areas and scientific disciplines encompassed by the program. Include a brief
description of the level(s) (i.e., undergraduate, predoctoral, postdoctoral, faculty) and duration of the
proposed training, the projected number of participating trainees and their anticipated levels of
experience. This section must be no longer than 30 lines of text and must follow the required font and
margin specifications.
Project Narrative
Using no more than two or three sentences, describe the relevance of this research training program to
public health. In this section, use plain language that can be understood by a general, lay audience.
Bibliography & References Cited
This item should be used only to cite references supporting the need, rationale, and approach for the
training program described in the PHS 398 Research Training Program Plan. Note that the Literature
Cited section of the Research Plan is captured in this section (unlike the placement in the PHS 398). Do
not include lists of publications of project directors, mentors or trainees in this section, as this information
will be included in the biosketches and Data Tables.
Facilities & Other Resources
Describe the facilities and resources that will be used in the proposed training program. Indicate in what
ways the applicant organization will support the program, financial or otherwise (e.g., supplementation of
stipends, protected time for mentoring, support for student activities). This could also include, for
example, space, shared laboratory facilities and equipment, funds for curriculum development, release
time for the PD/PI and participating faculty, support for additional trainees in the program, or any other
creative ways to improve the climate for the establishment and growth of the research training program.
Other Attachments
Leave blank, unless specifically requested in the FOA.
8.4.4 Special Instructions for 4.5 Senior/Key Person Profile
(Expanded) Form
Complete the Profile for the Program Director according to instructions in Section 4.5.
If multiple PD/PIs are proposed, explain in the Program Plan your rationale for how this will facilitate
program administration. If your application involves Multiple PD/PIs, follow the directions in Section 4.5
to designate the Contact PI and to assign the PD/PI role to other senior/key persons. Additionally, the
application must include a Multi-PD/PI Leadership Plan emphasizing how it will benefit the program and
the trainees. Do not submit a leadership plan if you are not submitting a Multiple PD/PI application. See
Part I Section 8.7 for information associated with Multiple Program Directors.
Complete the profiles for other senior/key persons according to instructions in Section 4.5.
The Program Director(s) (in case of multiple PD/PIs), training faculty and any other individuals whose
contributions are critical to the development, management and execution of the Research Training
Program Plan in a substantive, measurable way (whether or not salaries are reimbursed) should be
identified as senior/key persons. These would include co-Director(s), if applicable, and program staff.
Since these efforts are not project related research endeavors, they should not be identified in Other
Support information. Do not include proposed mentors and training faculty members (other than

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-165
senior/key persons) in this section. Biographical Sketches for mentors and participating faculty will be
included in the PHS 398 Research Training Program Plan Form, Participating Faculty Biosketches.
8.4.5 Special Instructions for 4.7 Research & Related Budget
This form is required for use in conjunction with the PHS 398 Training Budget for the R90 portion of
T90/R90 applications, and is the only budget form that should be used for K12 applications. Otherwise
this form should only be used when allowed or required in an FOA or IC-specific notice or
announcement. Follow instructions in Section 4.7.
8.4.6 Special Instructions for 4.6 PHS 398 Cover Page Supplement
Human Subjects
If you checked “Yes” to Human Subjects and “Yes” to Clinical Trial on the R&R Other Project
Information form, you must check either “Yes” or “No” to indicate whether plans include or potentially
include trainee participation in projects that are NIH-Defined Phase III Clinical Trials.
Inventions and Patents
Not applicable – leave blank.
Program Income
Check “No”.
Human Embryonic Stem Cells (hESC)
Check “Yes” if training plans include or potentially will include involvement of trainees in projects that
include human embryonic stem cells.
If “Yes”, list the 4-digit NIH Registration Number of the specific cell line(s) from the NIH Human
Embryonic Cell Registry, or check the box indicating that the specific stem cell line cannot be referenced
at this time. Applications proposing the use of hESC must either specify a cell line(s) from the NIH Stem
Cell Registry that will be used in the proposed research or, provide a strong justification for why an
appropriate cell line cannot be chosen from the Registry at the time of application and a certification that
one from the Registry will be used. The justification should be included in the Research Strategy section
of the application. For additional guidance, see NIH Guide Notice NOT-OD-12-111
Notice of Impending
Change in Peer Review Criteria and Submission Requirements for NIH Applications Involving Human
Embryonic Stem Cells.
Note that individual project HESC information is not required at the time of application, but will be
requested as Just-in-time (JIT) information prior to award. At that time, the NIH will require information
regarding project title, mentor and specific cell line(s) from the registry
(http://grants.nih.gov/stem_cells/registry/current.htm
) for each trainee utilizing human embryonic stem
cells in a research project. Trainees may not participate in human embryonic stem cell related research
until this information is provided.
8.5 PHS 398 Training Budget
For NRSA training grant programs, use the PHS 398 Training Budget form pages and follow the
instructions below. Refer to the relevant FOA or consult the PHS awarding component for current stipend
levels and allowable costs.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-166
For Non-NRSA training grant programs refer to the FOA for instructions regarding which Budget Form
pages to use and how to complete them.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-167
If you are requesting a budget of $500,000 direct costs or more for any year, contact the awarding
component to determine whether you must obtain prior approval before submitting the application. Some
Institutes/Centers do not require prior approval. (See Policy on the Acceptance for Review of Unsolicited
Applications That Request $500,000 or More in Direct Costs.)
PHS 398 Training Budget, Periods 1 through 5
Part A. Stipends, Tuition/Fees
Enter the number of trainees, total stipend amount and total tuition/fees for each trainee category as
appropriate. Use the current Institutional Kirschstein-NRSA stipend schedule,
(http://grants.nih.gov/training/nrsa.htm
). If a category contains different stipend levels, e.g., for varying
levels of postdoctoral experience and/or varying appointment periods, itemize in the appropriate blocks.
Enter the total stipends for all categories.
See http://grants.nih.gov/grants/guide/notice-files/NOT-OD-10-073.html for NIH policy regarding
payment of tuition and fees. Tuition at the postdoctoral level is limited to that required for specified
courses that are to be described in the Budget Justification (Part F.). Tuition and fees may be requested
only to the extent that the same resident or nonresident tuition and fees are charged to regular non-
Federally supported students and postdoctoral fellows. Where applicable, trainees should be divided into
non-degree-seeking and degree-seeking categories. Note that health insurance is not included as part of
this budget category. See the Training Related Expenses category below. Grantees should request full
needs. The formula currently in effect will be applied by the NIH awarding component at the time an
award is calculated.
Part B. Other Direct Costs
Enter the total costs for Trainee Travel, Training Related Expenses, Total Direct Costs from R&R Budget
Form (if applicable) and Consortium Training Costs (if applicable).
Trainee Travel
Some NIH awarding components pay a flat rate per trainee for trainee travel for all long-term trainees.
See the appropriate FOA and/or contact the awarding component to determine the amount provided for
travel. In the budget justification, state the purpose of any travel, giving the number of trips involved, the
destinations, and the number of trainees for whom funds are requested. PHS policy requires coach class
air travel be used. Justify foreign travel in detail, describing its importance to the training experience.
Enter the total amount requested in the Trainee Travel column.
Training Related Expenses (TRE)
Funds to defray other costs of training, such as health insurance (self-only or family), staff salaries,
consultant costs, equipment, research supplies, staff travel, etc., are requested as a lump sum based on the
amounts specified in the FOA and at
http://grants.nih.gov/grants/guide/notice-files/NOT-OD-10-073.html
for each predoctoral and postdoctoral trainee. Based on the number of trainees at the predetermined rate,
enter the total dollar figure.
Health insurance (self-only or family, as applicable) is an allowable cost that may be requested as part of
training related expenses, but only to the extent that the same health insurance fees are charged to regular
non-Federally-supported students and postdoctoral fellows. The allowable TRE amount will be awarded
as a lump sum. No further itemization or explanation is required.
The awarding Institute/Center will apply the Training Related Expenses level established for NRSA
Institutional programs for the relevant fiscal year at the time of award.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-168
Total Direct Costs from R&R Budget Form (if applicable)
Certain FOAs allow funds to cover costs for items other than those specified above. Use Research &
Related Budget Pages, Sections A through I and K, to submit those costs. Total Direct Costs from the
Research & Related Budget page should be inserted here. This line should not include any applicant
indirect costs.
Consortium Training Costs (if applicable)
If training is occurring at more than one institution, and any transfer of funds between institutions occurs,
the Training Subaward Budget Attachment Form should be used. (See Section 4.8). Total the direct costs
from the Subaward Budget Attachment Forms and insert here. The applicant institution is responsible and
accountable for any arrangements, expenditures, and submission of all required forms when more than
one institution is involved in the research training program.
Part C. Total Direct Costs Requested
Total dollar amount of other direct costs requested (automatically calculated).
Part D. Indirect Costs
Facilities and Administrative (F&A) costs under Institutional Kirschstein-NRSAs, other than those issued
to U.S., state, or local government agencies, will be awarded at 8%, excluding tuition/fees, equipment,
and sub-grants and contracts in excess of $25,000. Equipment and consortium costs are also excluded
from the F&A costs on those training grants where Training Related Expenses are not calculated and
awarded on a lump-sum basis, such as the Minority Access to Research Careers Program (MARC) or
Career Opportunities in Research (COR) Undergraduate Research Training Program. State and local
government agencies will receive the full F&A cost rate.
Indirect Cost Type: Enter “F&A”
Indirect Cost Rate (%): Enter “8”
Indirect Cost Base ($): Enter the sum of Stipends and Total Other Direct Costs requested, regardless of
whether those direct costs were listed on the PHS 398 Training Budget page or Research & Related
Budget page. Indirect costs are not paid on Tuition/Fees, equipment, and sub-grants and contracts in
excess of $25,000.
Funds Requested ($): Enter the product of Indirect Cost Rate multiplied by Indirect Cost Base.
Part E. Total Direct and Indirect Costs Requested (C+D)
The sum of Total Direct Costs Requested and Total Indirect Costs Requested will be calculated
automatically.
Part F. Budget Justification
A detailed justification is to be attached only for the first budget period, but should reflect the entire
budget period. Explain in detail the composition of any of the above items, as necessary. Itemize tuition
and individual fees. If tuition varies, (e.g., in-state, out-of-state, student status) identify these separately. If
tuition is requested for postdoctoral trainees, the specific courses must be described in the application. If
trainee travel is not paid at a flat rate per trainee by the awarding component, state the purpose of any
travel, giving the number of trips involved, the destinations, and the number of individuals for whom
funds are requested, bearing in mind that PHS policy requires coach class air travel be used. For
postdoctoral training slots, justify the stipend levels requested.
Any foreign travel must be justified in detail, describing its importance to the training experience and
considering the type of opportunities available for training, how those opportunities differ from and

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-169
complement those offered by the grantee institution, and the relationship of the proposed off-site training
experience to the career stage of the grantee.
This budget justification should apply only to funds requested on the PHS 398 Training Budget form.
When the Research & Related Budget Form is also used, two separate budget justifications are required,
each covering the costs required in the particular budget form. Combining the information into a single
upload is acceptable; however, each budget form requires a budget justification attachment so the same
budget justification will need to be included in both budget forms.
PHS 398 Training Budget, Cumulative Budget
All values on this form are calculated automatically. They present the summations of the amounts that
you have entered previously, for each of the individual budget periods. Therefore, no data entry is
allowed or required.
If any of the amounts displayed on this form appears to be incorrect, you may correct it by adjusting one
or more of the values that contribute to that total. To make any such adjustments, you will need to revisit
the appropriate budget period form(s) to enter corrected values.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-170

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-171
8.6 PHS 398 Training Subaward Budget Attachment(s) Form

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-172
This form should be used when proposing subawards to other institutions. Complete the Subaward
Budget for each contractor or collaborating institution. For NRSA programs, this is not common but is
usually encountered when a portion of the training program takes place at a site other than the grantee
institution via a collaborative or consortium arrangement. In such situations, the grantee institution is
responsible and accountable for acceptable training arrangements, expenditure of funds and the
submission of all required forms.
This form accommodates up to 30 separate subaward budgets. If you are submitting an application with
>30 subaward budgets, budgets 31 and above should be converted to PDF and included as part of Section
8.5 Part F. Budget Justification, of the parent budget (PHS 398 Training Budget). Reminder, the sum of
all subaward budgets; e.g., those attached separately and those provided as part of the budget justification,
must be included in Part B. Consortium Training Costs on the PHS 398 Training Budget.
To start the process, the applicant organization should:
• Select the Subaward Budget Attachment Form from the Optional Documents in the Grant
Application Package.
• Open the form, and click the “Select to Extract a Training Subaward Budget Attachment”
button in the middle of the form. A “SAVE” dialog box appears.
• Save the file locally using the first ten letters of the consortium organization’s name and use
“.pdf” as the file extension. (The extracted file is an Adobe PDF file.) Once you have saved the
file there is no need to extract another budget attachment. Doing so may cause you to lose any
data already stored in the saved file.
• E-mail the extracted, saved form to the consortium grantee. Note: consortium grantees must have
installed Adobe Reader before they can complete the form. The consortium grantee should
complete all the budget information as instructed in the R&R Budget form instructions in
Section
4.7. Note: Organizational DUNS and Name of Organization fields must reflect that of the
subaward/consortium grantee.
• The consortium grantee must complete the budget form and e-mail it back to the applicant
organization.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-173
• Return to the Subaward Budget Attachment Form and attach the consortium grantee’s budget to
one of the blocks provided on the form.
Submitting Subaward Budgets that are not Active for all Periods of the Prime Grant
When submitting subaward budgets that are not active for all periods of the prime grant, fill out the
subaward R&R Budget form and include only the number of periods for which the subaward is active.
The budget period start/end dates reflected in each period should reflect the corresponding prime budget
period start/end dates. This approach is the most workable solution to the limitations in existing forms
that do not allow an “empty” budget period and do not allow submission of a subaward budget with zero
effort to skip a budget period.
For example, suppose the prime has filled out a budget form with the following periods:
• period 1 Jan 1, 2010 – Dec 31, 2010
• period 2 Jan 1, 2011 – Dec 31, 2011
• period 3 Jan 1, 2012 – Dec 31, 2012
• period 4 Jan 1, 2013 – Dec 31, 2013
• period 5 Jan 1, 2014 – Dec 31, 2014
Now, suppose there is a subaward that performs in support year 1 and does not become active again until
support year 4. The subaward can fill out the first two periods of their budget form as follows:
• period 1 Jan 1, 2010 – Dec 31, 2010 (dates correspond to prime period 1)
• period 2 Jan 1, 2013 – Dec 31, 2013 (dates correspond to prime period 4)
It is not necessary that the budget period numbers between the prime and subaward match; the correlation
is reflected in the dates. Do be careful, however, that the dates exactly match what is listed for the period
in the prime budget.
Note this approach may cause a validation warning regarding the NIH $500,000 per year limit on direct
costs, therefore you should document in both the cover letter and the subaward budget justification that
the subaward is only active for specific periods of the prime. Appropriate NIH staff has access to the
cover letter and reviewers have access to the budget justification. This documentation will make the date
correlation immediately apparent and will help avoid any confusion.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-174
8.7 Research Training Program Plan Form
Before preparing the Research Training Program Plan, be sure to check the specific instructions in
the Funding Opportunity Announcement (FOA) to which you are responding. Contact the
appropriate PHS awarding component, which may have further advice or suggestions on
completing your application, including the data tables mentioned below.
Note that there are page limits for certain sections. Follow the page limits for the
Institutional Research
Training and Career Development Applicants, Including Ruth L. Kirschstein NRSA Application in the
table of page limits (Table 2.6-1), unless specified otherwise in the FOA. Please see NOT-OD-11-039 and
NOT-OD-11-076. The information provided in required data tables (see below) will not be counted
toward the page limitation. These tables should be numbered consecutively and titled as shown, even if
some are not required by the PHS awarding component to which you are applying or in the FOA to which
you are responding. Indicate by table number and title, those tables that are intentionally omitted.
Additional tables that are not required may be included in the Research Training Program Plan, however,
these tables will count as part of the page limit. Additional tables not specified in these instructions

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-175
should be identified by letter, rather than number to avoid confusion with the sequentially numbered
required tables.
The instructions for Data Tables 1-12 are located on the OER website at
http://grants.nih.gov/grants/
funding/424/index.htm#datatables. Please read the Introduction to the Data Tables before beginning to
prepare your application. This section includes important definitions that should be used consistently both
in the Data Tables and in all other parts of the application. The data tables should be included in the
application at the point indicated and should not be inserted in the narrative.
The Research Training Program Plan should include sufficient information needed for evaluation of the
program, independent of any other document (e.g., previous application). Be specific and informative,
and avoid redundancies.
Research Training Program Plan Attachments
(See also Section 2.3.2 Creating PDFs for Text Attachments
.)
Although many of the sections of this application are separate PDF attachments, page limitations
referenced in the instructions and/or funding opportunity announcement must still be followed. Agency
validations will include checks for page limits (and use of appropriate font). Some accommodation will be
made for sections that, when combined, must fit within a specified limitation.
Text attachments should be generated using word processing software and then converted to PDF using
PDF generating software. Avoid scanning text attachments to convert to PDF since that causes problems
for the agency handling the application. In addition, be sure to save files with descriptive file names.
Do not include any information in a header or footer of the attachments. A header will be system-
generated that references the name of the PD/PI. Page numbers for the footer will be system-generated in
the complete application, with all pages sequentially numbered.
Since a number of reviewers will be reviewing applications as an electronic document and not a paper
version, applicants are strongly encouraged to use only a standard, single-column format for the text.
Avoid using a two-column format since it can cause difficulties when reviewing the document
electronically.
Full-sized glossy photographs must only be included within the page limitations of the Research Training
Plan. The maximum size of images to be included should be approximately 1200 x 1500 pixels using 256
colors. Figures must be readable as printed on an 8.5 x 11 inch page at normal (100%) scale.
Investigators must use image compression such as JPEG or PNG. Do not include figures or photographs
as separate attachments either in the Appendix or elsewhere in the application.
Separate Attachments
Separate attachments have been designed for the Research Training Program Plan sections to maximize
automatic validations conducted by the eRA system. When the application is received by the agency, all
of the Research Training Program Plan sections will be concatenated in the appropriate order so that
reviewers and agency staff will see a single cohesive Research Plan.
When attaching a PDF document to the actual forms, please note you are attaching an actual document,
not just pointing to the location of an externally stored document. Therefore, if you revise the document
after it has been attached, you must delete the previous attachment and then reattach the revised document
to the application form. Use the “View Attachment” button to determine if the correct version has been
attached.
Follow page limitations as specified in Funding Opportunity Announcements.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-176
All applications and proposals for NIH funding must be self-contained within specified page limitations.
Agency validations will include checks for page limits. Note that while these computer validations will
help minimize incomplete and/or non-compliant applications, they do not replace the validations
conducted by NIH staff. Applications found not to comply with the requirements may be delayed in the
review process. Unless otherwise specified in an NIH solicitation, internet website addresses (URLs) may
not be used to provide information necessary to the review because reviewers are under no obligation to
view the internet sites. Moreover, reviewers are cautioned that they should not directly access an internet
site as it could compromise their anonymity.
Notice of Proprietary Information
Applicants are discouraged from submitting information considered proprietary unless it is deemed
essential for proper evaluation of the application. However, when the application contains information
that constitutes trade secrets, or information that is commercial or financial, or information that is
confidential or privileged, make sure you have checked “Yes” in the “Other Project Information” form.
When information in the application constitutes trade secrets or information that is commercial or
financial, or information that is confidential or privileged, it is furnished to the Government in confidence
with the understanding that the information shall be used or disclosed only for evaluation of this
application. However, if a grant is awarded as a result of or in connection with the submission of this
application, the Government shall have the right to use or disclose the information to the extent
authorized by law. This restriction does not limit the Government’s right to use the information if it is
obtained without restriction from another source.
Although the grantee institution and the PD/PI will be consulted about any such release, the PHS will
make the final determination. Any indication by the applicant that the application contains proprietary or
privileged information does not automatically shield the information from release in response to a
Freedom of Information Act (FOIA) request should the application result in an award (see 45 CFR Part
5). If an applicant fails to identify proprietary information at the time of submission as instructed in the
application guide, a significant substantive justification will be required to withhold the information if
requested under FOIA.
Begin each text section of the Research Training Program Plan with a section header (e.g.,
Introduction, Background, Program Plan, etc).
Field Name Instructions
1. Introduction to
Application (for
Resubmission or Revision
only)
Use only if Type of Application is Resubmission or Revision The
Introduction may not exceed 3 pages for resubmissions (previously known
as a revision or amendment) or 1 page for revisions (previously known as
competing supplements). See specific instructions in Section 2.7
“Resubmission” Applications and 2.8 “Revision” Applications concerning
the content of the Introduction.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-177
Field Name Instructions
2. Background The Background must fit within the page limits for Institutional Research
Training and Career Development Applicants, Including Ruth L.
Kirschstein NRSA Application in the table of page limits (Table 2.6-1),
unless specified otherwise in the FOA. Please see NOT-OD-11-039 and
NOT-OD-11-076.
Provide the rationale for the proposed research training program, relevant
background history, and the need for the research training proposed.
Indicate how the proposed program relates to current training activities at
the applicant institution.
Summarize the research training activities of the major participating
unit(s) and department(s) represented in the proposed program. Complete
and refer to the data reported in Tables 1-3:
Table 1. Membership of Participating Departments/Programs
Table 2. Participating Faculty Members
Table 3. Institutional Training Grant Support Available to Participating
Faculty Members, Departments, or Programs
Use this data to document the environment in which the proposed training
program will take place.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
3. Program Plan The Program Plan must fit within the page limit for Institutional Research
Training and Career Development Applicants, Including Ruth L.
Kirschstein NRSA Application in the table of page limits (Table 2.6-1),
unless specified otherwise in the FOA.
Please see NOT-OD-11-039 and NOT-OD-11-076.
a. Program Administration. Describe the Program Director's
qualifications for providing leadership of the program, including relevant
scientific background, current research areas, and experience in research
training. Indicate the Program Director's percent effort in the proposed
program.
Describe the administrative structure of the program and the distribution of
responsibilities within it, including the means by which the program
director will obtain continuing advice with respect to the operation of the
program.
If multiple PD/PIs are proposed, explain in this section your rationale for
how this will facilitate program administration. In addition, you must
complete the Multiple PD/PI Leadership Plan.
b. Program Faculty. Refer to the data presented in Table 2. Participating
Faculty Members and elaborate as necessary to describe each faculty

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-178
Field Name Instructions
member's research that is relevant to the program and indicate how
trainees will participate in the research. Describe the extent to which
participating faculty members cooperated, interacted, and collaborated in
the past, including joint publications and joint sponsorship of student
research. Complete and refer to data in Tables 4-6:
Table 4. Grant and Contract Support of the Participating Faculty Members
Table 5. Pre and Postdoctoral Trainees of Participating Faculty Members
Table 6. Publications of Research Completed by Trainees (or Potential
Trainees)
Use these tables to document the ability of the faculty to support the
research activities of the proposed trainees, the training record of the
faculty members, and the success of their trainees in generating
publishable research results.
For new applications, see the instructions for Table 6A or 6B, as
applicable, and list publications for trainees who are representative of
those who would be appointed if the grant is awarded. For Renewal
applications, this data constitutes part of the Progress Report (see Progress
Report below).
c. Proposed Training. Describe the proposed training program. State the
training level and number of trainees. For postdoctoral trainees, indicate
the proposed distribution by degree (e.g., M.D., Ph.D.). Describe course
work and research opportunities, the extent to which trainees will
participate directly in research, and the duration of training, i.e., usual
period of time required to complete the training offered.
Indicate how the individual disciplinary and/or departmental components
of the program are integrated and coordinated and how they will relate to
an individual trainee's experience.
For training programs that emphasize research training for clinicians,
describe the interactions with basic science departments and scientists.
Include plans for ensuring that the training of these individuals will
provide a substantive foundation for a competitive research career.
Generally, a minimum of 2 years of research training is required for all
postdoctoral trainees with health professional degrees. Describe fully any
trainee’s access to and responsibility for patients, including time
commitment.
Provide representative examples of programs for individual trainees.
Include curricula, degree requirements, didactic courses, laboratory
experiences, qualifying examinations, and other training activities, such as
seminars, journal clubs, etc. Describe how the preceptor and research
problems are chosen, how each trainee's program will be guided, and how
the trainee's performance will be monitored and evaluated. Include
detailed mentoring plans as appropriate.
If research on Human Embryonic Stem Cells (hESCs) is proposed but an
approved cell line from the NIH hESC Registry cannot be identified,

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-179
Field Name Instructions
provide a strong justification for why an appropriate cell line cannot be
chosen from the Registry at this time
d. Training Program Evaluation. Describe an evaluation plan to review
and determine the quality and effectiveness of the training program. This
should include plans to obtain feedback from current and former trainees
to help identify weaknesses in the training program and to provide
suggestions for program improvements. In addition, describe plans for
assessing trainee’s career development and progression, including
publications, degree completion, and post-training positions. Evaluation
results are to be included in renewal (competing continuation) applications
and as part of the Final Progress Report.
e. Trainee Candidates. Describe recruitment plans, including the sources
and availability of trainees; the qualifications of prospective trainees; and
the criteria and procedures by which trainees will be selected.
f. Institutional Environment and Commitment to Training. The
administration of the applicant institution as well as all participating units
and departments should include information in the application that
documents institutional support and commitment to the goals of the
research training program. The application should include a description of
support (financial and otherwise) to be provided to the proposed program.
This could include, for example, space, shared laboratory facilities and
equipment, funds for curriculum development, release time for the PD/PI
and/or participating faculty, support for additional trainees in the program,
or any other creative ways to improve the climate for the establishment
and growth of the research training program.
Admissions and Completion Records
For programs that request only predoctoral trainee support, complete Table
7A. Admissions and Completion Records for the Participating
Departments and Programs During the Past Five Years (Predoctoral
Applicants).
For programs that request only postdoctoral trainee support, complete
Table 7B. Admissions and Completion Records for the Participating
Departments and Programs During the Past Five Years (Postdoctoral
Applicants).
Programs requesting support for both predoctoral and postdoctoral trainees
need to complete both Table 7A and Table 7B. Applicant institutions do
not need to submit pre-enrollment data for individuals with disabilities and
individuals from disadvantaged backgrounds in the first two data columns
of these tables. However, all other information requested in the Table 7A
and Table 7B must be provided as instructed.
Use these tables to document the ability of the participating
departments/programs to recruit and retain predoctoral and/or postdoctoral
trainees through the completion of their training, the selectivity of the
admissions process, the success of the departments/programs in

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-180
Field Name Instructions
recruitment and retention of trainees from diverse backgrounds, and to
defend the requested number of training positions to be awarded.
Qualifications of Applicants
For programs that request only predoctoral trainee support, complete Table
8A. Qualifications of Recent Predoctoral Applicants.
For programs that request only postdoctoral trainee support, complete
Table 8B. Qualifications of Recent Postdoctoral Applicants.
Programs requesting support for both predoctoral and postdoctoral trainees
need to complete both Table 8A and Table 8B.
Use these tables to document the quality and depth of the applicant pools,
including both Kirschstein-NRSA training grant eligible and non-
Kirschstein-NRSA eligible applicants; the selectivity of the admissions
process; the competitiveness of the program; and to justify the number of
number of training positions requested.
Current Trainee Qualifications
For programs that request only predoctoral trainee support, complete Table
9A. Qualifications of the Current Predoctoral Trainees Clearly Associated
with the Training Program.
For programs that request only postdoctoral trainee support, complete
Table 9B. Qualifications of the Current Postdoctoral Trainees Clearly
Associated with the Training Program.
Programs that request both predoctoral and postdoctoral trainee support
need to complete both Table 9A and Table 9B.
Use these tables to document the number and quality of all trainees
currently enrolled in the program, and their distribution by department and
mentor; the selectivity of enrollment appointments to the training grant
over the time period represented by the current program participants; and
to defend the number of training positions to be awarded.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
4. Recruitment and
Retention Plan to Enhance
Diversity
This section must fit within the combined 25-page limit for sections 2.2-
2.4. Please see NOT-OD-11-039 and NOT-OD-11-076
.
A Recruitment and Retention Plan to Enhance Diversity is required for all
training grant activity codes except T34, T36, U2R, and all D-series
activity codes.
The NIH recognizes a unique and compelling need to promote diversity in
the biomedical, behavioral, clinical and social sciences workforce. The
NIH expects efforts to diversify the workforce to lead to the recruitment of

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-181
Field Name Instructions
the most talented researchers from all groups; to improve the quality of the
educational and training environment; to balance and broaden the
perspective in setting research priorities; to improve the ability to recruit
subjects from diverse backgrounds into clinical research protocols; and to
improve the Nation’s capacity to address and eliminate health disparities.
Accordingly the NIH continues to encourage institutions to diversify their
student and faculty populations and thus to increase the participation of
individuals currently underrepresented in the biomedical, clinical,
behavioral, and social sciences such as: individuals from underrepresented
racial and ethnic groups, individuals with disabilities, and individuals from
socially, culturally, economically, or educationally disadvantaged
backgrounds that have inhibited their ability to pursue a career in health-
related research. Institutions are encouraged to identify candidates who
will increase diversity on a national or institutional basis.
Definition of Diversity Recruitment Groups
The NIH is particularly interested in encouraging the recruitment and
retention of the following classes of candidates:
A. Individuals from racial and ethnic groups that have been shown by
the National Science Foundation to be underrepresented in health-
related sciences on a national basis (see data at
http://www.nsf.gov/statistics/showpub.cfm?TopID=2&SubID=27
and
the report Women, Minorities, and Persons with Disabilities in
Science and Engineering, 2007, p. 262). The following racial and
ethnic groups have been shown to be underrepresented in biomedical
research: African Americans, Hispanic Americas, Native Americans,
Alaskan Natives, Hawaiian Natives, and natives of the U.S. Pacific
Islands. In addition, it is recognized that under-representation can
vary from setting to setting and individuals from racial or ethnic
groups that can be convincingly demonstrated to be underrepresented
by the grantee institution should be included in the recruitment and
retention plan.
B. Individuals with disabilities, who are defined as those with a physical
or mental impairment that substantially limits one or more major life
activities.
C. Individuals from disadvantaged backgrounds who are defined as:
1. Individuals who come from a family with an annual income
below established low-income thresholds. These thresholds are
based on family size, published by the U.S. Bureau of the Census;
adjusted annually for changes in the Consumer Price Index; and
adjusted by the Secretary for use in all health professions
programs. The Secretary periodically publishes these income
levels at http://aspe.hhs.gov/poverty/index.shtml
. For individuals
from low income backgrounds, the institution must be able to
demonstrate that such candidates (a) have qualified for Federal
disadvantaged assistance; or (b) have received any of the

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-182
Field Name Instructions
following student loans: Health Professional Student Loans
(HPSL), Loans for Disadvantaged Student Program; or have
received scholarships from the U.S. Department of Health and
Human Services under the Scholarship for Individuals with
Exceptional Financial Need.
2. Individuals who come from a social, cultural, or educational
environment such as that found in certain rural or inner-city
environments that have demonstrably and recently directly
inhibited the individual from obtaining the knowledge, skills, and
abilities necessary to develop and participate in a research career.
Recruitment and retention plans related to a disadvantaged
background (C1 and C2) are most applicable to high school and
perhaps undergraduate candidates, but would be more difficult to
justify for individuals beyond that level of achievement. Under
extraordinary circumstances the PHS may, at its discretion,
consider an individual beyond the undergraduate level to be from
a disadvantaged background. Such decisions will be made on a
case-by-case basis, based on appropriate documentation.
New applications must include a description of plans to enhance
recruitment of a diverse trainee pool and may wish to include data in
support of past accomplishments. Renewal applications must include a
detailed account of experiences in recruiting individuals from
underrepresented groups during the previous funding period. Information
should be included on both successful and unsuccessful recruitment
strategies.
History and Achievements. Describe efforts to recruit trainees from
Diversity groups A, B, and C into the existing training program. For
competing continuation/renewal applications, also describe past efforts to
recruit and retain underrepresented minority students into training grant
funded positions. Refer to the data presented in Table 1 and Table 7A and
Table 7B that provided statistics on applications and admissions of these
groups in comparison to the overall trainee pool.
For Renewal applications, complete Table 10 Admissions and Completion
Records for Underrepresented Minority (URM), Trainees with Disabilities,
and Trainees from Disadvantaged Backgrounds Clearly Associated with
the Training Program. New applicants may provide this data if they wish;
however, it is not required of new applicants.
Use this data to document the success of the program in recruiting and
retaining trainees who are under-represented minorities, provide analysis
of their support, and begin to establish a record of NIH training of other
Diversity Recruitment groups.
Proposed plans. Describe steps to be taken during the proposed award
period regarding the identification, recruitment, and retention of graduate
students and postdoctorates from underrepresented groups. Consider the
success and/or failures of recruitment strategies used in the past. In

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-183
Field Name Instructions
particular, describe the specific efforts to be undertaken by the training
program and how these might relate to the recruitment efforts of the
medical school, graduate school, and/or the university at large. In most
cases, institutional efforts alone will not satisfy the requirement to recruit
individuals from underrepresented groups.
Applications without a description of diversity recruitment efforts will be
considered incomplete and may be delayed in the peer review process.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
5. Plan for Instruction in
the Responsible Conduct
of Research
Follow the page limits for Plan for Instruction in the Responsible Conduct
of Research in the table of page limits (Table 2.6-1), unless specified
otherwise in the FOA.
Please see NOT-OD-11-039.
A plan for Instruction in the Responsible Conduct of Research (RCR)
is required for all training grant activity codes except T36. Every
trainee must receive instruction in the responsible conduct of research. See
Supplemental Instructions Part III Section 1.16 for information on the NIH
Policy on Training in the Responsible Conduct of Research (RCR).
New applications must include a plan for instruction in the responsible
conduct of research. The plan should address how applicants plan to
incorporate the five instructional parts outlined in the NIH Policy on
Training in the Responsible Conduct of Research: format, subject matter,
faculty participation, duration, and frequency. In addition, the plan must
describe how participation in RCR instruction will be monitored.
In addition, Renewal applications must describe any changes in formal
instruction over the past project period and plans for the future that address
any weaknesses in the current RCR instruction. All training faculty who
served as course directors, speakers, lecturers, and/or discussion leaders
during the past project period must be named in the application.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
This is a required attachment unless the FOA specifies otherwise.
6. Progress Report
(Renewal Applications
Only)
State the period covered and briefly describe the accomplishments of the
training program. Describe any specific effects of this training program on
curriculum and/or research directions. Describe how the funds provided
under Training Related Expenses were used to benefit the program. For
each trainee supported during the period covered, indicate their
preceptor/mentor, and briefly summarize the research conducted by the

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-184
Field Name Instructions
trainee. For previous trainees appointed to the training grant and
continuing in training, provide a brief statement of their status and
progress toward completion of their training program. Refer to data
presented in Table 6 Publications of Research Completed by Trainees (or
Potential Trainees) and include in the table publications of trainees through
the time that they complete their training for all trainees currently or
previously supported by the training grant program regardless of whether
support from this training grant is cited in the publication.
Complete Table 11 Appointments to the Training Grant For Each Year of
the Past Award (Renewal Applications Only).
Use this table to document the utilization of awarded training positions. If
any trainee positions were not filled, provide an explanation.
For programs that request only predoctoral trainee support, complete Table
12A Predoctoral Trainees Supported by this Training Grant (Renewal
Applications Only).
For programs that request only postdoctoral trainee support, complete
Table 12B Postdoctoral Trainees Supported by this Training Grant
(Renewal Applications Only).
Programs requesting support (or reporting on prior support) for both
predoctoral and postdoctoral trainees need to complete both Table 12A and
Table 12B.
Use these tables to document how predoctoral training positions are used
(i.e., distribution by mentor, year in program, years of support per trainee)
and the success of the program in achieving the training objectives of the
prior award period(s) for up to 10 years. Summarize this data in the text.
If any postdoctoral trainee with a health professional degree appointed to
the grant during the most recent award period received less than 2 years of
research training, explain why. Where possible for past trainees, describe
the extent of their current involvement in research, including research
grant support and representative recent publications. Use the progress
report narrative to provide information that is not readily presented in the
required tables.
Renewal applications must include a detailed account of experiences in
recruiting individuals from underrepresented groups during the previous
funding period. Information should be included on both successful and
unsuccessful recruitment strategies.
Renewal applications must describe the type of instructions provided in the
current project period, the degree of student participation, the results of
any assessments, and other relevant information.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-185
Field Name Instructions
7. Human Subjects Complete this section if you answered “yes” to the question “are human
subjects involved?” on the R&R Other Project Information Form.
If trainee participation in research involving human subjects is solely as
part of other research projects that have received or will receive IRB
review and approval, and no portion of the Training Grant Award will be
used to support this research; provide a list of previously approved
research projects (grant number, PD/PI, and project title) and their IRB
approval dates or exemption designations. If plans are indefinite provide
an explanation and prior to trainee participation in other research project
grants, provide a list as indicated.
If the training program involves definite plans for the participation of
human subjects as defined in Supplemental Instructions Part III.3,
Human
Subjects Research Definitions and Terms, outside of research projects that
have received or will receive IRB review, follow the instructions in
Supplemental Instructions
Part II, Supplemental Instructions for Preparing
the Protection of Human Subjects Section of the Research Plan.
The appropriate grants must be identified along with their IRB approval
dates or exemption designation.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
8. Vertebrate Animals Complete this section if you answered “yes” to the question “are
Vertebrate Animals Used?” on the R&R Other Project Information Form.
If the training program involves the use of live vertebrate animals solely as
part of other research project grants, and no portion of the Training Grant
Award will be used to support the purchase, use, or husbandry of live
vertebrate animals in this research; provide a list of previously approved
research projects (grant number, PD/PI, and project title) and IACUC
approval dates. If plans are indefinite provide an explanation and prior to
trainee participation in other research project grants, provide a list as
indicated.
If the training program involves definite plans to use live vertebrate
animals outside of research projects that have received or will receive
IACUC approval, follow the instructions in Part I, 5.5, (Research Plan
Form), Vertebrate Animals
.
The appropriate grants must be identified along with their IACUC
approval dates.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-186
Field Name Instructions
9. Select Agent Research If participating faculty proposed in the training program are conducting or
plan to conduct research involving select agents in which trainees may
participate, follow the instructions in Section 5.5, (Research Plan Form),
Select Agent Research
.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
10. Multiple PD/PI
Leadership Plan
If you wish to submit a multiple PD/PI application, you must provide a
Leadership Plan. Do not submit a leadership plan if you are not submitting
a Multiple PD/PI application. For applications designating multiple
PD/PIs, all such individuals must be assigned the PD/PI role on the
Senior/Key Profile form, even those at organizations other than the
applicant organization. Refer to the instructions in Section 5.5 (Research
Plan Form), Multiple PD/PI Leadership Plan
. However, the emphasis in a
training grant multiple PD leadership plan should be on how it will benefit
the program and the trainees. A single Contact PD must be designated for
the purpose of communicating with the NIH, although other individuals
may contact the NIH on behalf of the Contact PD when necessary.
Because training programs are intended to be coherent, NIH will not
allocate the budget or training positions between multiple PDs. A single
award will be made. Multiple PD plans should include reasonable numbers
of PDs and each should be included for a specific purpose. Multiple-PD
applications should not include all mentors of the training grant as PDs,
except in unusual cases.
For background information on the Multi-PD/PI initiative, see:
http://grants.nih.gov/grants/multi_pi/index.htm
.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
11. Consortium and
Contractual Arrangements
Describe any programmatic, fiscal, or administrative arrangements
between the applicant organization and other participating organizations.
See Section 5.5, Consortium/Contractual Arrangements
for additional
guidance.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
12. Participating Faculty
Biosketches
Faculty Biosketches for participating faculty should follow the Additional
NIH and other PHS Agencies Instructions for a Biographical Sketch,
except that a personal statement is not required for participating

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-187
Field Name Instructions
faculty. These should be attached as a single document to avoid having to
upload large numbers of separate documents. However, the Biosketches of
the Program Director and other Senior/key Personnel should also be
entered as described under SF424 (R&R) Section 4.5
Senior/Key Person
Profile (Expanded) Form.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
13. Data Tables Instructions for Data Tables 1-12 mentioned above are located on the OER
website at the following URL
http://grants.nih.gov/grants/funding/424/index.htm#datatables
. These
instructions include an Introduction to the Data Tables that provides
instructions applicable to all tables, specific instructions for each table, and
Sample Data Tables. The Sample Data Tables illustrate the kind of data to
include in each table for Kirschstein-NRSA training grant applications. Be
sure to choose the Instruction and Blank Data Table set that corresponds to
the type of application you are submitting, e.g., New, Renewal, or
Revision Application, and the kind of training to be provided, e.g.,
predoctoral only, postdoctoral only, or pre and postdoctoral mixed.
Modification of these instructions for use in non-Kirschstein-NRSA
training grant applications will be included in relevant FOAs.
Save this information in a single file in a location you remember. Start
each numbered table on a new page. Bookmark the first page of each
table by its table number (Table 1, Table 2, etc.) before you upload the
file. Click Add Attachment, browse to where you saved the file, select the
file, and then click Open.
14. Letters of Support Attach appropriate letters here from all individuals confirming their roles
in the project. Letters documenting any agreements between the Program
Director(s) and senior administration officials or other institutional
officials are not required but may be included. For consultants, letters
should include rate/charge for consulting services.
The Program Director should check the FOA (particularly for non-NRSA
programs) to determine if any program-specific letters of support are
required.
Save this information in a single file in a location you remember. Click
Add Attachment, browse to where you saved the file, select the file, and
then click Open.
15. Appendix Do not use the appendix to circumvent the page limitations of the Training
Plan. All appendix material must be submitted as PDF attachments. A
summary listing all of the items included in the appendix is required, and
should be the first PDF file of the Appendix. Applications that do not

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-188
Field Name Instructions
follow the appendix requirements may be delayed in the review process.
Research publications of trainees and mentors are not normally included as
part of the Training Grant applications, but are allowed. Other types of
publications reflecting on the activities of the program as a whole may also
be included. When publications are allowed, appendix materials should be
limited to those which are not publicly available, such as:
• Manuscripts and/or abstracts accepted for publication but not yet
published.
• Published manuscripts and/or abstracts where a free, online,
publicly available journal link is not available.
• Patents directly relevant to the project.
Publications that are publicly accessible must not be included in the
appendix. For such publications, the URL or PMC submission
identification numbers along with the full reference should be included as
appropriate in the Progress Report section of the Research Training
Program Plan, and/or in the Biographical Sketch.
Do not include unpublished theses or abstracts/manuscripts submitted but
not yet accepted for publication.
Some materials other than publications that are unique to training grant
applications (but not typically included in research grant applications) may
be included as appendices. In general, the appendix may be used to
provide samples of materials that are referred to in the body of the
application, but are too cumbersome to include in the Research Training
Program Plan without disrupting the narrative flow. Examples include:
i. Syllabi for key courses, core courses and electives, including courses
in Responsible Conduct of Research, Survival Skills for Research,
etc.;
ii. Retreat, seminar series, and other program activity agendas, rosters,
and schedules;
iii. Examples of forms used to document trainee progress and monitoring
by the program;
iv. Examples of materials used in recruitment and particularly recruitment
and retention to enhance diversity of the student pool;
v. Lists of meetings attended by students and their presentations; and
vi. Student biosketches.
As a reminder, tables other than the required Data Tables 1-12, must be
incorporated into the page limit of the Research Training Program Plan.
Follow the page limits for the
Institutional Research Training and Career
Development Applicants, Including Ruth L. Kirschstein NRSA
Application in the table of page limits (Table 2.6-1), unless specified
otherwise in the FOA. These additional tables must not be included in the
appendix materials.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-189
8.8 Training Grant Peer Review Process
The goals of NIH-supported research training are to help ensure that a diverse pool of highly trained
scientists is available in adequate numbers and in appropriate research areas to address the Nation’s
biomedical, behavioral, and clinical research needs. The scientific review group will address and consider
each of criteria below in assigning the application’s overall score, weighting them as appropriate for each
application. Reviewers will first determine the quality of the proposed research training program,
including information presented in the data tables and appendix, and then consider whether the requested
number of trainee positions is appropriate for the program.
The general process information (Overview, Streamlining, and Dual-Level Peer Review) found in Part I.6
applies to training grant applications as well. However, the actual review criteria and other review
considerations are different.
Review Criteria:
• Training Program and Environment
• Training Program Director/Principal Investigator (PD/PI)
• Preceptors/Mentors
• Trainees
• Training Record
Additional Review Criteria:
• Protection of Human Subjects from Research Risk
• Inclusion of Women, Minorities and Children in Research
• Care and Use of Vertebrate Animals in Research
• Biohazards
• Resubmission Applications
• Renewal Applications
Additional Review Considerations:
• Training in the Responsible Conduct of Research
• Recruitment and Retention Plan to Enhance Diversity
• Budget and Period of Support
Applicants should carefully review the applicable FOA for complete information associated with the peer
review process. The FOA will describe essential information to be submitted for each of the above
elements.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-190
9. Supplemental Instructions to the SF 424 (R&R) for
Preparing a Multi-Project Application
9.1 Introduction
The supplemental instructions found in this Section are for Multi-Project Applications (MPA) and include
guidance and instructional information only when there is a difference in the required information to be
submitted or there is a need for more specificity for an MPA. Therefore, these supplemental instructions
must be used along with the information found in Parts I.1 – I.6 of this document.
In FY2013 NIH began to transition multi-project applications to electronic submission using the SF424
(R&R) application packages. This transition is done by activity code. As each code is added (see the
transition timeline
), all applications submitted in response to Funding Opportunity Announcements
(FOAs) for programs using that code will require electronic submission (including new, resubmission,
renewal and revision types of application). The requirement for electronic submission will be stated in
the FOA.
9.2 Multi-Project Applications
A Multi-Project Application is a single submission with multiple, interrelated components that share a
common focus or objective. A component is a distinct, reviewable part of a multi-project application for
which there is a business need to gather detailed information as defined in a particular FOA. Each
component includes the data collection identified for its specific component type. Components typically
include general information (component organization, project period, project title, etc.) performance sites,
personnel, and budget. They may also be referred to as cores or projects. The construction of the
application and naming convention will be defined by the FOA.
Although MPAs submitted electronically will use application packages that use the SF424 (R&R) and
PHS 398 forms, there are some differences in the way the multi-project application is structured. Every
application will include:
• A Single Overall Component: The Overall component describes the entire application and
provides an overview of how each of the additional components fit together.
• Other Components: Other component types (e.g., Admin Core, Project, Core) will vary by
opportunity. The component types will be specified in the FOA. Within ASSIST, only the
specified types of components will be available to choose from a drop-down menu during the
application preparation process.
• Summaries: Information automatically compiled from the data provided by the applicant in the
individual components and included in the agency assembled application (e.g., component and
categorical budget data) to help reviewers and staff work with the application. These are system-
generated summaries and will be presented in the assembled application as part of the Overall
Component. Summaries will be generated for the follow information when applicable:
Performance Sites; Human Subjects, Clinical Trials, Human Embryonic Stem Cells, and
Vertebrate Animals; Budgets; and Senior/Key Personnel, including Biosketches.
It is imperative that applicants become familiar with the FOA for the specific MPA for which support is
being requested. Before applying for an MPA, applicants should carefully review the applicable FOA
noting the special application instructions. When the instructions in the FOA differ from the instructions
given in the SF 424 (R&R) Application Guide or the ASSIST User Guide, the FOA instructions should be
followed. The FOA will specify the types of Other Components that should be used when preparing the

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-191
application, whether each component is optional or required and any restrictions on the number of times
each component can be included in a responsive application (e.g., 2-3 Projects are required). The FOA
will include specific information associated with the application and the names of individuals who may be
contacted prior to submission of an application for additional or clarifying information.
9.2.1 Collaborating with Other Organizations
Components may be developed by the applicant institution or another organization. Applicants using
ASSIST will be able to delegate application preparation responsibilities to multiple users within and
outside the applicant organization while maintaining appropriate access control and security. Refer to the
ASSIST User Guide for additional guidance.
Depending on the role of the collaborating organization in the overall project, there are two approaches to
constructing the application.
1. Collaborating Organization as the Lead in an Other Component: The applicant organization is
always identified in the Overall component. However, when an entire Other Component is led by a
different subaward/consortium organization complete, all the forms for that Other Component
(including the R&R Cover Form and Budget Form) using the subaward/consortium organization’s
information. On the Budget Form, use the Budget Type “Project” (marked on top of the form) and
the subaward/consortium organization’s DUNS and Name. ASSIST will automatically calculate the
budget information as a consortium/subaward to the Overall Component’s applicant organization.
2. Collaborating Organization as a Consortium in an Other Component: When a collaborating
organization is not the lead in an Other Component, then that organization will be represented as a
consortium/subaward. In this scenario the applicant organization is the lead on the Other
Component. Therefore, complete all the forms for the Other Component (including the R&R Cover
Form and Budget Form) using the applicant organization’s information. On the Budget Form, use
the Budget Type “Project” (marked on the top of the form) and the applicant organization’s
information. To add the subaward/consortium budget, on the “Summary” tab for the component use
the “Add Optional Form” action to add another R&R budget form. Indicate Budget Type
“Subaward/Consortium”, and complete this form using the information for the subaward/consortium
organization. ASSIST will automatically calculate this information as a subaward to the applicant
organization.
9.3 Application Submission System & Interface for
Submission Tracking (ASSIST)
ASSIST is a web-based interface for the preparation and submission of multi-project applications.
ASSIST is accessed using existing eRA Commons credentials with any of the following roles: Signing
Official (SO), Administrative Official (AO), Principal Investigator (PI), Assistant (ASST) or Account
Administrator (AA). Most applicants do not need to complete additional registrations to use the system.
ASSIST allows applicants to:
• Use current eRA Commons credentials to access ASSIST
• Populate data from established eRA Commons profiles
• Run federal-wide and agency business rule validations prior to submission
• Take advantage of automatic generation of the table of contents, headers, footers, page numbers,
etc.
PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-192
• Print/preview applications in the format used by the agency prior to submission including system-
generated data summaries
• Present to reviewers clear, color PDF images, rather than scanned versions of the application
• Avoid the hassle of preparing and shipping multiple paper copies
Applicant organizations that use system-to-system solutions will be able to use those services, in lieu of
ASSIST, for submitting multi-project applications into Grants.gov if their provider offers the service.
The ASSIST User Guide
contains detailed instructions for using ASSIST to complete the MPA
application. Instructions for the specific requirements of an MPA (e.g. types of components, page limits)
are found in the individual FOA. In addition to the guidance included in this application guide and the
ASSIST User Guide, the following resources are also available:
• Multi-Project Applications section of the Applying Electronically website
• Electronic Multi-Project Application: Annotated SF424 (R&R) Form Set
• Frequently Asked Questions
• “Tips” section at the top of each form tab in ASSIST
ASSIST uses a combination of web-based data entry screens and PDF file attachments to compile the
required data elements of an application. The information entered in the data screens is the same as the
information requested in the SF424 (R&R) and PHS 398 forms and the instructions for completing the
data elements are the same, unless noted otherwise in this section of the application guide or within the
FOA.
Table 9.3-1 ASSIST Data Forms
DATA COLLECTION OVERALL COMPONENT OTHER COMPONENT
SF424 R&R Cover Yes Yes (some data items)
PHS 398 Cover Page Supplement Yes Yes
SF424 R&R Other Project Information Yes Yes
SF424 R&R Project/Performance Sites Yes Yes
SF424 R&R Senior/Key Person Profile Yes Yes
SF424 R&R Budget No Yes
SF424 R&R Subaward Budget Attachment No Optional
PHS 398 Research Plan Yes Yes
Planned Enrollment Report No Optional
PHS398 Cumulative Inclusion Enrollment Report No Optional

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-193
Special Note on Structuring the Application: The assembled application image created for an MPA application
has a predetermined order. The Overall Component is always first and includes the system-generated data
summaries. Other component types follow in alphabetical order (e.g., Cores before Projects). Components of the
same type are grouped together in the order created in ASSIST and given sequential numbers (e.g., Core-001,
Core-002, Project-001, Project-002). Applicants do not have the ability to reorder components within ASSIST.
Therefore, some planning should be done before initiating components. For more information on MPA application
assembly, see
http://grants.nih.gov/grants/ElectronicReceipt/files/Electronic_Multi-
project_Application_Image_Assembly.pdf.
9.4 Special Instructions to the SF424 R&R for Multi-Project
Applications
Standard instructions found in Parts I.4 – I.5 apply to the Overall Component and all Other Components
with the exceptions found in this section. For each Form, follow the detailed corresponding instructions
in Part I. Sections 4 & 5 as well as the exceptions noted below.
9.4.1 Special Instructions for 4.2 Cover Form
DATA ITEM/FIELD NAME OVERALL COMPONENT OTHER COMPONENTS
General Instructions
All the SF424 (R&R) Cover form
fields are collected for the Overall
Component.
Only a subset of data fields from the Cover
Component is collected for the Other
Components. These are:
5. Applicant Information
7. Type of Applicant (optional)
11. Descriptive Title
12. Proposed Project
ASSIST: Fields are not automatically
populated. Use Expand All to expose the
fields available for data entry. Fields that are
grayed out do not need to be completed.
5. Applicant Information
This information is for the
Applicant Organization, not a
specific individual.
ASSIST: Applicant Information
DUNS and address fields are
populated from the Commons
Institutional Profile for the
organization selected when the
application was initiated and are
not editable.
The Contact email is required by
NIH for the Overall Component. If
not included, or improperly
formatted, the AOR e-mail address
will be used.
ASSIST: Fields that are grayed out do not
need to be completed.
7. Type of Applicant
This information is for the
Applicant Organization, not a
specific individual AOR or
PD/PI.
Required for Overall Component. Optional for Other Components.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-194
DATA ITEM/FIELD NAME OVERALL COMPONENT OTHER COMPONENTS
14. Project
Director/Principal
Investigator Contact
Information
ASSIST: The PD/PI information for
the Overall Component is
automatically populated from the
PD/PI section of the SF 424 (R&R)
Senior/Key Person Profile
(Expanded) form. The information
is not editable in the SF424 (R&R)
Cover form. Any changes must be
made on the Senior/Key Person
Profile form.
Not collected for Other Components.
15. Estimated Project
Funding
Required for Overall Component
ASSIST: Data must be manually
entered.
Not collected for Other Components.
9.4.2 Special Instructions for 4.3 Project/Performance Site Locations
A summary of Performance Sites for all Components will appear in the Overall section of the assembled
application image in eRA Commons and will be generated upon submission.
DATA ITEM/FIELD NAME OVERALL COMPONENT OTHER COMPONENTS
General Instruction
Include only the Primary Site in the
Overall Component.
A summary of Project/Performance
Sites in the Overall section of the
assembled application image in
eRA Commons compiled from data
collected in the other components
will be generated upon submission.
ASSIST: Summary Performance
Site information is available when
using “Preview Application.”
List all performance sites that apply
to the specific component.
DUNS Number
The DUNS is a required field for the
primary performance site in both the
Overall and Other Components.
The DUNS is a required field for the
primary performance site in both the
Overall and Other Components.
9.4.3 Special Instructions for 4.4 Other Project Information
Component
When applicable, a summary of Human Subjects, Clinical Trials, Human Embryonic Stem Cells, and
Vertebrate Animals for all Components will appear in the Overall section of the assembled application
image in eRA Commons and will be generated upon submission.
DATA ITEM
/
FIELD NAME
OVERALL COMPONENT
OTHER COMPONENTS
PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-195
DATA ITEM
/
FIELD NAME
OVERALL COMPONENT
OTHER COMPONENTS
1. Are human subjects
involved?
If activities involving human subjects are planned
at any time during the proposed project at any
performance site and/or on any Other Component,
check Yes for the Overall Component and
complete the remaining questions as instructed in
4.4 using the unique guidance below for selected
data items.
Answer only the “Are
Human Subjects
Involved?” and “Is the
Project Exempt from
Federal regulations?”
questions for Other
Components.
If yes, check appropriate
exemption number 1, 2, 3, 4,
5, 6
Complete for both the Overall and Other
Components. If the Overall Component
exemption is E4 then no other exemption number
can be set for any Other Component.
Complete for both the
Overall and Other
Components. If the
Overall Component
exemption is E4 then no
other exemption number
can be set for any Other
Component.
IRB Approval Date
Complete only for the Overall Component when
applicable.
Not collected for Other
Components.
Human Subject Assurance
Number
Complete only for the Overall Component when
applicable.
Not collected for Other
Components.
2. Are Vertebrate Animals
Used?
If activities involving vertebrate animals are
planned at any time at the Overall Component or
at any Other Component, at any performance site,
check yes on the Overall component and
complete the remaining questions as instructed in
4.4 using the unique guidance below for selected
data items. If no, skip the rest of block 2.
Answer only the “Are
Vertebrate Animals Used?”
question.
Is the IACUC review
Pending?
Complete only for the Overall Component when
applicable.
Not collected for Other
Components.
IACUC Approval Date
Complete only for the Overall Component when
applicable.
Not collected for Other
Components.
Animal Welfare Assurance
Number
Complete only for the Overall Component when
applicable.
Not collected for Other
Components.
6. Does this project
involve activities
outside of the United
States or partnerships
with international
collaborators?
Complete at both the Overall and Other
Component Levels. However, if Yes on any Other
Component, then the answer must be Yes for the
Overall Component.
Complete at both the
Overall and Other
Component Levels.
However, if Yes on any
Other Component, then
the answer must be Yes
for the Overall Component.
8. Project Narrative
Required for Overall Component. Optional for Other
Components. See
specific FOA to see if this
is required for any Other
Components.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-196
9.4.5 Special Instructions for 4.5 Senior/Key Person Profile
(Expanded)
A summary of Senior/Key Persons followed by their Biographical Sketches will appear in the Overall
section of the assembled application image in eRA Commons and will be generated upon submission.
DATA ITEM
/
FIELD NAME
OVERALL COMPONENT
OTHER COMPONENTS
General Instruction
Only list the Contact PD/PI and any Multi-
PD/PIs; all with role PD/PI on the Overall
Component. Do not list any other
Senior/Key personnel for the Overall
Component.
List all personnel who are
Senior/Key to the specific
component.
The PD/PI role should not be used
for any personnel in Other
Components.
Organization Name
Required by NIH for the Overall and Other
Components.
ASSIST: Overall Component contact PD/PI
Organization name is pre-populated from
SF 424 (R&R) organization.
Required by NIH for the Overall
and Other Components.
ASSIST: Overall Component
contact PD/PI Organization name
is pre-populated from SF 424
(R&R) organization.
Project Role
Must be PD/PI on Overall Component. Unless otherwise specified in the
FOA, in the Project
Director/Principal Investigator
section of the form, use Project
Role of “Other” with Category of
“Project Lead.”
Credential, e.g. agency
login
Must provide the eRA Commons
username for all PD/PIs.
Provide the eRA Commons
username for person identified in
the Profile – Project
Director/Principal Investigator
section of the form (typically
Project Lead).
Attach Biographical Sketch
Each Senior/Key individual, including the PD/PI, is allowed one biosketch for the
entire application. If an individual will participate on multiple components, chose
any single entry to attach the biosketch, make sure it reflects participation on
each relevant component.
The PD/PI(s) may include the biosketch in the Overall Component or any Other
Component.
Biosketches will not be repeated within each component in the application
image.
ASSIST: Summary information available when using “Preview Application.”

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-197
9.4.6 Special Instructions for 4.7 R&R Budget
The only budget information included in the Overall component is the Estimated Project Funding section
of the SF424 (R&R) Cover form. Complete a budget, including appropriate subaward budgets when
applicable, for the Other Components.
For information on preparing budgets when collaborating organizations are involved, see Section 9.2.1
.
Budget summaries will appear in the Overall section of the assembled application image in eRA
Commons, will be compiled from detailed budget data collected in the Other Components, and will be
generated upon submission.
ASSIST: Summary budget information is available when using “Preview Application.”
9.4.7 Special Instructions for 5.3 PHS 398 Cover Page Supplement
When applicable, a summary of Human Subjects, Clinical Trials, Human Embryonic Stem Cells, and
Vertebrate Animals for all Components will appear in the Overall section of the assembled application
image in eRA Commons and will be generated upon submission.
DATA ITEM
/
FIELD NAME
OVERALL COMPONENT
OTHER COMPONENTS
1. Project Director/Principal
Investigator
ASSIST: Not editable. Section is
populated from SF 424 (R&R)
Sr/Key Person Profile PD/PI section.
Any changes must be made on that
form for the Overall Component.
Not Applicable to Other
Components. Note this field reflects
the PD/PI for the entire application,
not the project lead for the Other
Component.
2. Human Subjects/Clinical
Trial?
Complete at both the Overall and
Other Component Levels. However,
if Yes on any Other Component,
then the answer must be Yes on the
Overall Component.
Complete at both the Overall and
Other Component Levels. However,
if Yes on any Other Component,
then the answer must be Yes on the
Overall Component.
Agency-Defined Phase III
Clinical Trial?
Complete at both the Overall and
Other Component Levels. However,
if Yes on any Other Component,
then the answer must be Yes on the
Overall Component.
Complete at both the Overall and
Other Component Levels. However,
if Yes on any Other Component,
then the answer must be Yes on the
Overall Component.
3. Human Embryonic Stem Cell
Does the proposed project
involve human embryonic
stem cells?
Complete at both the Overall and
Other Component Levels. However,
if Yes on any Other Component,
then the answer must be Yes on the
Overall Component.
Complete at both the Overall and
Other Component Levels. However,
if Yes on any Other Component,
then the answer must be Yes on the
Overall Component.
Cell Lines
All cell lines from Other
Components must be manually
entered in the Overall Component
cell line table.
List all cell lines that pertain to the
specific component.

PHS SF424 (R&R) Adobe Forms Version C Application Guide
Part I: Instructions for Preparing and Submitting an Application I-198
9.4.8 Special Instructions for 5.5 PHS 398 Research Plan
DATA ITEM
/
FIELD NAME
OVERALL COMPONENT
OTHER COMPONENTS
1. Introduction to Application
For Resubmission and Revision
applications, an Introduction to
Application is required on the
Overall Component, but may be
allowed for each Other Component
unless otherwise stated in the FOA.
For Resubmission and Revision
applications, an Introduction to
Application is required on the
Overall Component, but may be
allowed for each Other Component
unless otherwise stated in the FOA.
10. Multiple PD/PI Leadership
Plan
Required if more than one PD/PI is
specified on R&R Senior/Key
Person Profile form of the Overall
Component.
Not applicable.

