
T. Trimpe 2008 http://sciencespot.net/
Bonding Basics Review Name _____________________________
1. Complete the chart using your knowledge of atoms.
Element
Atomic Symbol
Total # of
Electrons
# of Valence
Electrons
# of Electrons
Gained or Lost
Oxidation
Number
Bromine
Lithium
Calcium
Sulfur
Boron
Silicon
Phosphorus
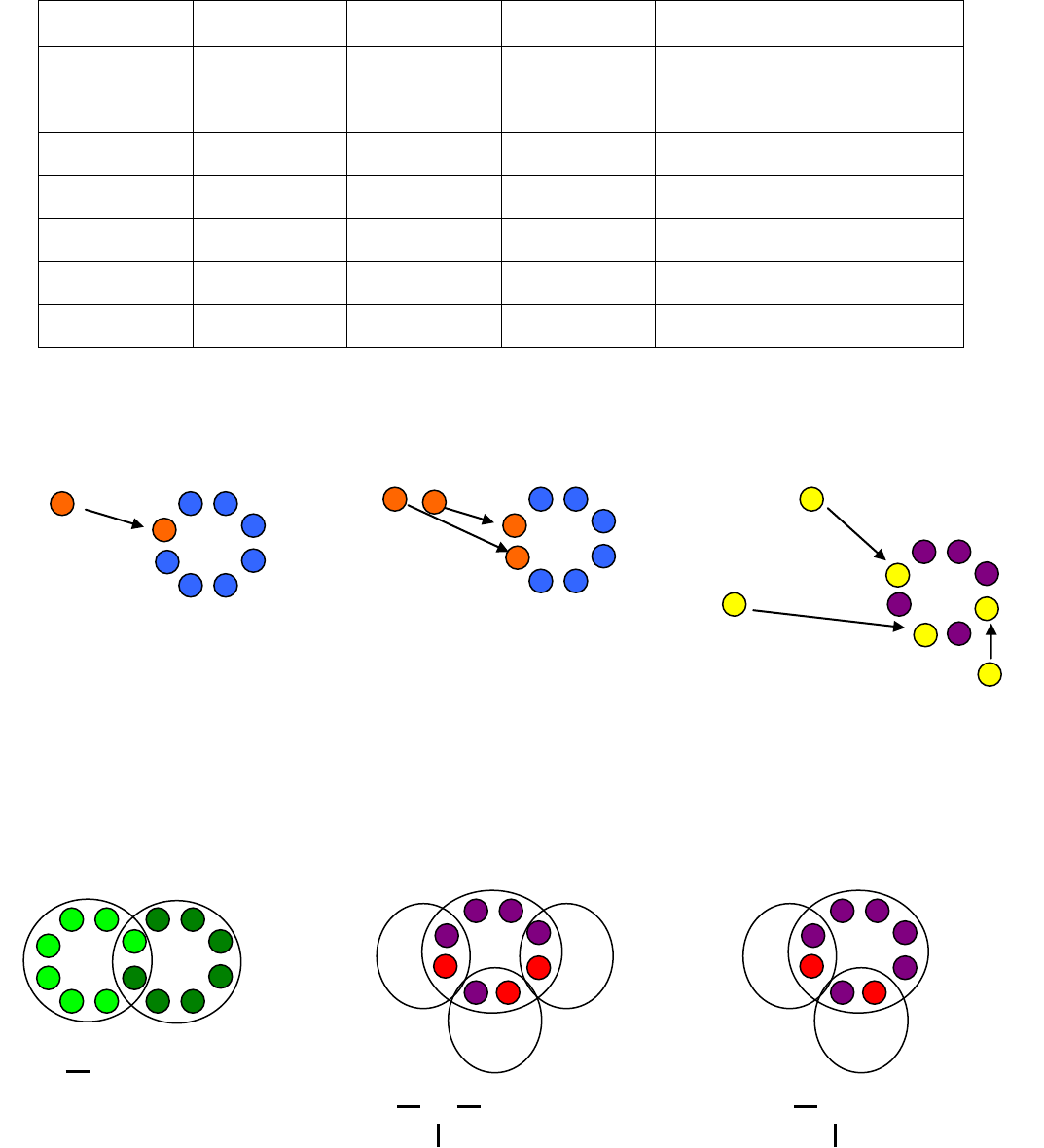
2. Ionic Bonds - Draw the Lewis structures for each atom, draw arrows to show the transfer of electrons,
write the charge for each ion, and then write the chemical formula.
(A) Potassium + Iodine (B) Magnesium + Oxygen (C) Lithium + Nitrogen
3. Covalent Bonds – Draw the Lewis structures for each atom, draw circles to show the electrons that are
shared, and then write the bond structure and chemical formula.
(A) Fluorine + Fluorine (B) 3 Hydrogen + 1 Phosphorus (C) 2 Hydrogen + 1 Sulfur
T. Trimpe 2008 http://sciencespot.net/
Bonding Basics Review ANSWER KEY
1. Complete the chart using your knowledge of atoms.
Element
Atomic Symbol
Total # of
Electrons
# of Valence
Electrons
# of Electrons
Gained or Lost
Oxidation
Number
Bromine
Br
35
7
Gain 1
1-
Lithium
Li
3
1
Lose 1
1+
Calcium
Ca
20
2
Lose 2
2+
Sulfur
S
16
6
Gain 2
2-
Boron
B
5
3
Lose 3
3+
Silicon
Si
14
4
Gain/Lose 4
4+ 4-
Phosphorus
P
15
5
Gain 3
3-
2. Ionic Bonds - Draw the Lewis structures for each atom, draw arrows to show the transfer of electrons,
write the charge for each ion, and then write the chemical formula.
(A) Potassium + Bromine (B) Magnesium + Oxygen (C) Lithium + Nitrogen
3. Covalent Bonds – Draw the Lewis structures for each atom, draw circles to show the electrons that are
shared, and then write the bond structure and chemical formula.
(A) Fluorine + Fluorine (B) 3 Hydrogen + 1 Phosphorus (C) 2 Hydrogen + 1 Sulfur
K
Br
K
1+
+ Br
1-
KBr
Mg
O
Mg
2+
+ O
2-
MgO
Li
N
Li
Li
Li
1+
+ Li
1+
+ Li
1+
+
N
3-
Li
3
N
F
F
F F F
2
H
H
P
H
H
3
P
H
P
H
H
H
H
H
S
H
H
2
S
H
S
H
